Purpose
To evaluate refractive error in infants who underwent intravitreal bevacizumab injection for treatment of threshold retinopathy of prematurity (ROP).
Design
Retrospective nonrandomized interventional comparative study.
Methods
The study group included all infants who consecutively received a single intravitreal bevacizumab (0.375 mg or 0.625 mg) injection for therapy of threshold ROP in fundus zone I or zone II. The control group included infants who had previously undergone retinal argon laser therapy of ROP. The follow-up examination included refractometry under cycloplegic conditions.
Results
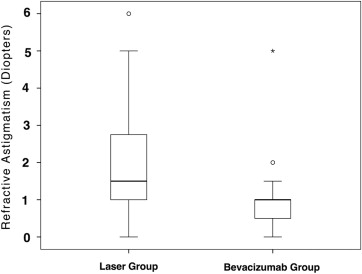
The study group included 12 children (23 eyes; mean birth weight: 622 ± 153 g; gestational age: 25.2 ± 1.6 weeks) and the control group included 13 children (26 eyes; birth weight: 717 ± 197 g; gestational age: 25.3 ± 1.8 weeks). Both groups did not differ significantly in birth age and weight and follow-up. At the end of follow-up at 11.4 ± 2.3 months after birth, refractive error was less myopic in the study group than in the control group (−1.04 ± 4.24 diopters [median: 0 diopters] vs −4.41 ± 5.50 diopters [median: −5.50 diopters]; P = .02). Prevalence of moderate myopia (17% ± 8% vs 54% ± 10%; P = .02; OR: 0.18 [95% CI: 0.05, 0.68]) and high myopia (9% ± 6% vs 42% ± 10%; P = .01; OR: 0.13 [95% CI: 0.03, 0.67]) was significantly lower in the bevacizumab group. Refractive astigmatism was significantly lower in the study group (−1.0 ± 1.04 diopters vs 1.82 ± 1.41 diopters; P = .03). In multivariate analysis, myopic refractive error and astigmatism were significantly associated with laser therapy vs bevacizumab therapy ( P = .04 and P = .02, respectively).
Conclusions
In a 1-year follow-up, a single intravitreal bevacizumab injection as compared to conventional retinal laser coagulation was helpful for therapy of ROP and led to less myopization and less astigmatism.
Retinopathy of prematurity (ROP) is a retinal ischemic disease in prematurely born infants, and it is characterized by retinal and retinovitreal neovascularization, which eventually leads to tractional retinal detachment. Since vascular endothelial growth factor (VEGF) plays an important role in the pathogenesis of ROP, recent studies have shown the feasibility of the intravitreal use of VEGF inhibitors for the therapy of ROP. In a recent randomized controlled trial, Mintz-Hittner and associates demonstrated that intravitreal bevacizumab monotherapy as compared with conventional laser therapy showed in infants with stage 3+ ROP a significant benefit for zone I disease but not zone II disease. Interestingly, the peripheral retinal vessels continued to develop after treatment with intravitreal bevacizumab, whereas conventional laser therapy led to a permanent destruction of the peripheral retina. It has remained unclear whether the intravitreal bevacizumab therapy as compared to the peripheral retinal laser coagulation leads to the same amount of axial myopization. We therefore conducted this study to assess the refractive error of young children who had undergone intravitreal bevacizumab therapy and compared the refractive error of children who had previously undergone laser therapy.
Methods
The retrospective nonrandomized interventional comparative study included all children who were treated with an intravitreal injection of bevacizumab (Avastin; Genentech Inc, San Francisco, California, USA) for ROP threshold disease in posterior zone II or zone I or for prethreshold ROP in zone I. The study was a retrospective analysis of clinical data obtained during routine care of the children. The Ethics Committee II of the Medical Faculty Mannheim of the Ruprecht-Karls-University Heidelberg approved the study (# 2012-613R-MA). The diagnosis of ROP was based on the revised guidelines of the International Committee for the Classification of Retinopathy of Prematurity. The criterion for therapy of ROP was the presence of a threshold disease, which was defined as disease stage with a 50% likelihood of progressing to retinal detachment. Threshold disease was considered to be present when stage 3 ROP was present in either zone I or zone II, with at least 5 continuous or 8 total clock hours of disease, and the presence of plus disease. Starting in 2008, we discussed with the parents of all affected children the possibility of an off-label intravitreal injection of bevacizumab as an alternative to conventional standard laser treatment performed according to the international guidelines of the Early Treatment for Retinopathy of Prematurity Cooperative Group. The children of all parents who preferred the intravitreal bevacizumab therapy received that treatment and were included in our study. Informed consent clearly describing the experimental nature of the intravitreal bevacizumab injection was obtained from the parents of all children in the study.
The intravitreal bevacizumab injection was performed under sterile conditions in the pediatric intensive care unit for prematurely born infants. After topical anesthesia of the cornea and conjunctiva by repeated application of oxybuprocaine hydrochloride eye drops and after inducing a slight systemic sedation of the children, the external eye and the surrounding skin were disinfected with a povidone-iodine 5% ophthalmic solution (Betadine; Alcon Inc, Fort Worth, Texas, USA) and the eyes were draped. A lid speculum was inserted. Using a 30-gauge needle, bevacizumab (Avastin) was intravitreally injected at 1.5 mm posterior to the limbus in the temporal inferior quadrant. Because of the relatively small volume injected, an anterior chamber paracentesis was not performed. A reflux of fluid or vitreous was avoided by passing the 30-gauge needle through the sclera in an oblique direction. After the injection, we applied a steroid-antibiotic eye drop combination 4 times per day and atropine 0.5% eye drops twice a day for 1 week. The surgical procedure was performed by 1 of 3 surgeons (B.C.H., F.C.S., W.J.). The study group was compared with a control group of children who had previously undergone retinal argon laser therapy of ROP in the same center. The indications for therapy were the same as for intravitreal bevacizumab injection. Control group and study group did not differ significantly in birth weight, gestational age, and length of follow-up.
The follow-up examinations were carried out by ophthalmologists experienced in diagnosing the disease. The follow-up scheme included daily visits after the injection or the laser therapy until a regression of the neovascularization was detected and the plus disease disappeared. Afterwards, 2 visits per week followed until the neovascularization had completely regressed. Weekly visits ensued up to a gestational age of 42 weeks. At a follow-up examination at about 12 months after birth, all infants were screened for refractive disorders, development of the anterior segment, and the retinal situation. Refractive errors were determined by cycloplegic retinoscopy and automatic refractometry (Autorefractor Portable Retinomax 3; Nikon Corp, Tokyo, Japan) approximately 30 minutes after instilling 1% cyclopentolate hydrochloride (twice with a delay of 10 minutes). For purposes of this study, moderate myopia was defined as a myopic refractive error of ≥−5 diopters (D) and high myopia was defined as a myopic refractive error of ≥−8 D. The description of the posterior pole examination was as follows: normal, abnormally straightened temporal retinal vessels, macular ectopia, retinal fold, partial or complete retinal detachment.
Statistical analysis was performed using a commercially available statistical software package (SPSS for Windows, version 20.0; IBM-SPSS Inc, Chicago, Illinois, USA). Continuous data were presented as mean ± standard deviation. Prevalence was presented as mean ± standard error. The normal distribution of parameters was tested using the Kolmogorov-Smirnov test. χ 2 tests were used to compare proportions. Odds ratios (OR) were presented and their 95% confidence intervals (CI) were described. All P values were 2-sided and were considered statistically significant when less than .05.
Results
The study group consisted of 23 eyes of 12 children (7 boys, 5 girls) with a mean ± standard deviation birth weight of 622 ± 153 g (median: 590 g; range: 450-1115 g) and a mean gestational birth age of 25.2 ± 1.6 weeks (median: 24.9 weeks; range: 23-29 weeks). Of the 12 children, 3 showed an acute posterior stage of ROP and fulfilled the criteria for treatment according to the guidelines of the Early Treatment for Retinopathy of Prematurity Cooperative Group. The remaining 9 children showed an ROP stage 3+ with neovascularization in 5 adjacent sectors of 30 degrees width or 8 nonadherent sectors and fulfilled the same criteria for therapy as did the children of the control group. In the first child of our series with aggressive posterior ROP in both eyes, 1 eye with clear optic media received a primary laser treatment and the other eye with a persistent tunica vasculosa lentis underwent an intravitreal injection of bevacizumab. Since the eye with laser therapy continued to develop retinovitreal neovascularization, it received an intravitreal injection of bevacizumab 4 weeks after laser therapy. That eye was not included either in the bevacizumab group or in the laser group. All other infants received a single injection of bevacizumab without any laser therapy prior to, or after, the bevacizumab application. Nine children (treated by B.C.H. and F.C.S.) received a dosage of 0.375 mg bevacizumab. We arrived at the dosage of 0.375 mg bevacizumab by taking one-fourth of the dosage given to an adult eye (about 1.5 mg) and by considering that the eye of an infant makes up about one-fourth of the volume of an emmetropic adult eye. Three children (treated by W.J.) received a dosage of 0.625 mg bevacizumab. The follow-up examination was performed at 11.1 ± 3.1 months of corrected age (median: 10.5 months; range: 7.0-23.0 months). The reason for the different dosages of bevacizumab was that the injection was performed by different surgeons.
The control group included 26 eyes of 13 children (7 boys, 6 girls) with a median birth weight of 717 ± 197 g (median: 690 g; range: 440-1090 g) and a mean gestational birth age of 25.3 ± 1.8 weeks (median: 25.0 weeks; range: 23-28 weeks). The follow-up examination was performed at 11.7 ± 1.6 months (median: 11.5 months; range: 10-14.0 months) of corrected age. Study group and control group did not differ significantly in birth weight ( P = .07), gestational age at birth ( P = .77), or length of follow-up ( P = .43).
All eyes of the study group showed a regression of plus disease within 2 to 6 days after the intravitreal injection; a decrease in pupillary rigidity; a resolution of any tunica vasculosa lentis, if present prior to the injection; and a complete regression of the retinal neovascularization within 2 to 3 weeks. In none of the children was a second intravitreal injection of bevacizumab necessary. None of the eyes of the study group showed signs of an injury to the lens or retina, retinal detachment or vitreous hemorrhage, or intraocular inflammation during the follow-up period. One eye of the laser group developed a partial retinal detachment (stage 4b). The infant who received a primary laser treatment in the right eye and an intravitreal bevacizumab injection 4 weeks later developed a retinal fold and macular ectopia. The left eye, which had received intravitreal bevacizumab only, showed a regression of the tunica vasculosa lentis and of all retinal neovascularizations without any retinal fold or any other morphologic abnormality. At 29 months of corrected age, the refractive error was −10.25 D in the right eye and −8.00 D in the left eye.
At the end of the follow-up period, mean refractive error was significantly less myopic in the study group than in the control group (−1.04 ± 4.24 D [median: 0 D; range: −12.5 to +4.63 D] vs −4.41 ± 5.50 D [median: −5.50 D; range: −14.0 to +4.38 D]; P = .02) ( Figure 1 ). In multivariate analysis with refractive error as dependent parameter and study group, birth weight, sex, and gestational birth age as independent parameters showed that myopic refractive error was significantly associated with laser therapy vs bevacizumab therapy ( P = .04; regression coefficient: 3.11 [95% confidence interval: 0.11, 6.11]), while birth weight ( P = .55), sex ( P = .22), and gestational age at birth ( P = .80) were not significantly associated with refractive error. In a binary regression analysis with laser therapy vs bevacizumab therapy as dependent variable and refractive error, birth weight, sex, and gestational birth age as independent parameters revealed that laser therapy was significantly associated with myopic refractive error ( P = .04; OR: 1.15 [95% CI: 1.01,1.31]), while birth weight ( P = .08), sex ( P = .29), and gestational age at birth ( P = .33) were not significantly associated with the type of therapy. The prevalence of moderate myopia (17% ± 8% [95% CI: 1,34] vs 54% ± 10% [95% CI: 33,74]) and of high myopia (9% ± 6% [95% CI: 0,21] vs 42% ± 10% [95% CI: 22,63]) was significantly ( P = .02; OR: 0.18 [95% CI: 0.05, 0.68] and P = .01; OR: 0.13 [95% CI: 0.03, 0.67], respectively) lower in the bevacizumab group than in the laser group.