Diagnostic criteria for classic malignant glaucoma
Flattening or shallowing of the axial (central anterior chamber with peripheral flattening)
Increased or greater than expected IOP after filtering surgery
Absence of pupillary block following iridotomy or iridectomy
Anticipated response to treatment
No reduction of IOP following miotic therapy
Reduction of IOP or reversal with aqueous suppressant, mydriatic-cycloplegic and/or osmotic therapy
Generally resolved by disruption of the anterior hyaloid by laser or surgery
The term malignant glaucoma is unacceptable to some clinicians because to patients it may suggest cancer, and it is not related to the underlying pathophysiology of the disease. Instead, various other descriptions such as aqueous misdirection syndrome, ciliary block glaucoma [3, 4], and direct lens block glaucoma [5] have been proposed. Nonetheless, the term malignant glaucoma is still used often because it is readily understood by most clinicians to define the classic phakic and nonphakic forms and is based on actual pathophysiologic mechanisms (Table 15.2).
Table 15.2
Clinical manifestations of malignant glaucoma
Classic malignant glaucoma |
|---|
Nonphakic malignant glaucoma |
Aphakic |
Pseudophakic |
Malignant-like glaucoma |
Classic Malignant Glaucoma
The pathognomonic findings of classic malignant glaucoma are increased IOP or unexpected normal IOP following glaucoma surgery, central (axial) shortening of the anterior chamber, open iridotomy or iridectomy, and no evidence for choroidal effusion or hemorrhage. Historically, it has an occurrence between 0.6 and 4 % of eyes that are diagnosed with acute angle-closure glaucoma [6–8]. The chance of the onset of malignant glaucoma following surgery for angle-closure glaucoma is the greatest when the angle is not completely opened at the time of surgery, regardless of whether or not the IOP has been reduced prior to the procedure [9]. In one study, a relationship between chronic angle-closure and malignant glaucoma was reported in 10 of 14 eyes [10]. Classic malignant glaucoma typically develops after surgical peripheral iridectomy or filtering (full-thickness sclerectomy or trabeculectomy) surgery in phakic patients.
The following two clinical cases are illustrative of classic malignant glaucoma presentations.
Case 1
A 72-year-old woman presented with symptoms of acute eye pain, headache, blurred vision, nausea, and vomiting. Visual acuity was hand motions in the affected eye. The cornea had diffuse bullous edema and the anterior chamber was flat peripherally. The IOP was 56 mmHg. A diagnosis of acute angle-closure was made. The patient did not respond to standard medical therapy; the cornea was too edematous to perform laser iridotomy, pupilloplasty, or peripheral iridoplasty. A paracentesis only transiently lowered the intraocular pressure and did not appreciably clear the cornea. Surgical iridectomy was performed on an urgent basis following pretreatment with IV osmotics. Postoperatively the chamber deepened centrally and peripherally and the angle was shallow but open on gonioscopy. The vision returned to 20/80 and the cornea cleared. The lens had moderate nuclear sclerosis and cortical opacity with glaukomflecken. The intraocular pressure fell to 18 mmHg. Five days later the patient returned with complaints of mild discomfort. Visual acuity was 20/200. The cornea was clear. The anterior chamber was flat peripherally and centrally. The surgical iridectomy was patent and the intraocular pressure was 38 mmHg (Fig. 15.1). There were no suprachoroidal effusions or hemorrhages detectable by ophthalmoscopy, B-scan ultrasound, or UBM. Based upon the findings, a diagnosis of classic malignant glaucoma was made (see Table 15.3). Intensive medical therapy was instituted (see section “Medical Treatment”), and over the next several days, the chamber slowly deepened and the pressure returned to 21 mmHg. The patient subsequently underwent uncomplicated cataract extraction with posterior chamber IOL with no recurrence of malignant glaucoma. The visual acuity with correction returned to 20/25. The presence of a moderate amount of peripheral anterior synechia necessitated chronic medical therapy for secondary angle closure to maintain an IOP of 15 mmHg.
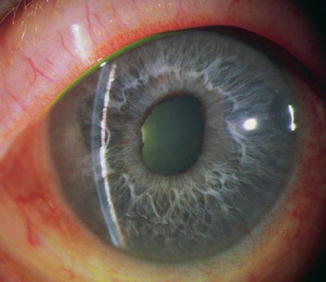

Fig. 15.1
Classic malignant glaucoma with central and peripheral flat chambers after a surgical peripheral iridectomy (Reproduced with permission from Cyrlin [70])
Table 15.3
Differential diagnosis of malignant glaucoma
Pupillary block | Choroidal effusion | Suprachoroidal hemorrhage | Malignant glaucoma |
|---|---|---|---|
Anterior chamber (AC) flat or shallow | AC flat or shallow | AC flat or shallow | AC flat or shallow |
Intraocular pressure (IOP) normal or elevated | IOP subnormal or low | IOP normal or elevated | IOP normal or elevated |
Fundus appears normal | Large, smooth, light brown choroidal elevations | Dark brown or dark red choroidal elevations | No choroidal elevation |
No suprachoroidal fluid | Straw-colored fluid present | Light red or dark red blood | No suprachoroidal fluid |
Choroidal drainage does not cure | Drainage does cure | Drainage does cure | Does not cure |
Iridotomy/iridectomy does cure | Does not cure | Does not cure | Does not cure |
Does not have patent iridotomy/iridectomy | Has patent iridotomy/iridectomy | Has patent iridotomy/iridectomy | Has patent iridotomy/iridectomy |
Onset early or late postoperatively | Onset first 5 days postoperatively, occasionally later | Onset at surgery or first 5 days postoperatively, rarely later | At surgery or first 5 days postoperatively, but sometimes weeks to months postoperatively |
Case 2
A 38-year-old woman with a history of uveitis and glaucoma developed a secluded pupil with pupillary block and iris bombé. This was relieved by surgical sector iridectomy. The IOP was initially managed medically but subsequently required trabeculectomy. On the third postoperative day, the bleb collapsed and the anterior chamber flattened both peripherally and centrally with an IOP of 24 mmHg (Fig. 15.2). A diagnosis of classic malignant glaucoma was made based on the presentation and the diagnostic findings (see Table 15.3). The patient did not respond to intensive medical therapy (see section on “Medical Treatment”) and vitrectomy was performed (see section on “Surgical Therapy”). Postoperatively the chamber deepened and the pressure was controlled on medical therapy.
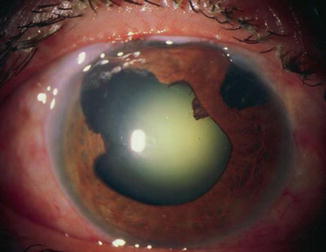

Fig. 15.2
Classic malignant glaucoma with central and peripheral flat chambers after sector iridectomy and subsequent trabeculectomy (Reproduced with permission from Cyrlin [70])
Elevated IOP and shallowing of the chamber can occur during the surgery, immediately afterward, after the discontinuation of cycloplegics [9], after the initiation of miotics [9, 11], or quite some time later [11–15]. If one eye develops malignant glaucoma after surgery, the contralateral eye will probably do likewise. Although the IOP may in some cases be in the teens to low twenties following filtration surgery, it may at times be 40–60 mmHg or greater. Miotics are ineffective in lowering IOP and instead may further increase the pressure. Conventional filtering surgery in patients who have had previous peripheral iridectomy does not reverse the problem.
Nonphakic Malignant Glaucoma
Malignant glaucoma in aphakia is a term for eyes with persistence of preexisting malignant glaucoma after cataract surgery as well as in patients with malignant glaucoma occurring following routine cataract surgery. Reports of nonphakic malignant glaucoma have increased during the past few decades. It has been noted after cataract surgery in eyes with [16] or without [7, 16] preexisting glaucoma. It has been described in eyes undergoing anterior chamber intraocular lens implantation at the time of cataract extraction [17]. Pseudophakic malignant glaucoma should be considered after surgery in patients with axial shallowing of the anterior chamber and in eyes with a shallow chamber and pressures in the teens or higher after a combined phacotrabeculectomy with lens implant [18]. Malignant glaucoma has also been reported following Nd:YAG laser capsulotomy [19], diode laser cyclocoagulation [20], and aqueous tube shunt surgery [21].
Other Malignant Glaucoma Syndromes
Numerous clinical entities share some or all the findings of classic phakic and nonphakic malignant glaucoma. Malignant glaucoma, malignant-like glaucoma [2], and other entities defined by their presumed pathophysiologic mechanisms have been described by various authors. An extensive review of case reports in the literature has been presented by Luntz and Rosenblatt [22].
Table 15.4 lists the circumstances when a clinical picture consistent with classic malignant glaucoma, but not necessarily occurring after typical incisional surgery, has been reported.
Table 15.4
Clinical picture consistent with classic malignant glaucoma
After laser treatment for glaucoma |
After laser iridectomy: |
Narrow angle [2] |
Acute angle closure [23] |
After Nd:YAG laser cyclophotocoagulation [28] |
After miotics |
With prior glaucoma surgery: |
Iridectomy [29] |
Filtration [11] |
Without prior surgery [30] |
After trauma [5] |
Associated with retinal disease |
After retinal detachment surgery [33] |
After central vein occlusion [34] |
Associated with inflammation [5] |
Associated with infection |
Fungal keratomycosis [35] |
Nocardia asteroidis [36] |
The presence of diagnostic criteria and the response to treatment as described earlier distinguish between malignant glaucoma and malignant-like glaucoma. The need for a patent iridectomy or iridotomy before the diagnosis is made cannot be emphasized enough.
Pathogenesis
The clinical findings in malignant glaucoma and its various presentations previously have been thought to be primarily the end result of aqueous misdirection or posterior aqueous diversion (Fig. 15.3a, b). Shaffer recognized early the effects of aqueous accumulation in the vitreous and its possible function in the pathogenesis of classic malignant glaucoma [37]. By definition, malignant glaucoma does not result from pupillary block and is not relieved by laser iridotomy or surgical iridectomy. Mechanisms that involve various contributions of the lens or zonules, ciliary processes, anterior hyaloid face, or vitreous body have been proposed.
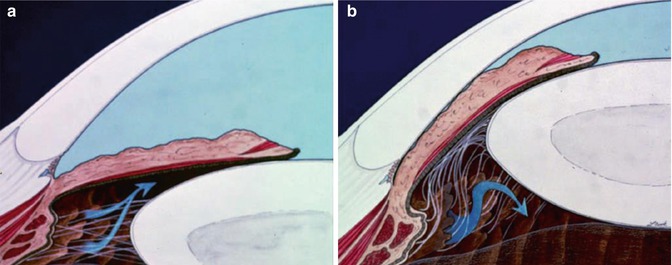

Fig. 15.3
(a) Normal pathway of aqueous passage from the posterior to anterior chamber (arrows). (b) Aqueous misdirection posteriorly to the vitreous (arrows) in malignant glaucoma (Illustrations courtesy of B. Thomas Hutchinson, MD. Reproduced with permission from Cyrlin [70])
The term ciliary block has been used by some clinicians to emphasize the presumed role of the ciliary processes in blocking the forward flow of aqueous in many cases [3, 4]. Swollen, anteriorly displaced ciliary processes may be pressed up against the lens or against abnormally forward vitreous in phakic patients, or the ciliary processes may be touching or adherent to vitreous in aphakic patients [8]. Anterior displacement of the lens has also been attributed to ciliary muscle contraction [5] and loose zonules [38, 39]. Swelling, spasm, or displacement of the ciliary processes could then be enhanced by miotics or inflammation. Direct lens block, as described by Levene [3], would result from anterior displacement of the lens with shallowing of both the peripheral and the central anterior chamber. Emphasizing posterior aqueous diversion, ciliary block, or direct lens block as the primary mechanisms in malignant glaucoma underestimates the role of the anterior hyaloid and vitreous, which are probably of greater significance.
One of the great puzzles about malignant glaucoma is that laboratory studies suggest that at physiological rates of aqueous production, malignant glaucoma should not occur. Epstein and colleagues have proposed that resistance to the forward flow of aqueous could be further exacerbated by a diminution of anterior hyaloid surface area resulting from apposition to the peripheral lens or ciliary body. Malignant glaucoma would result from a persistent expansion of the vitreous volume in an eye with a relatively impermeable and diminished anterior hyaloid surface rather than from isolated pools of trapped aqueous in the vitreous cavity (Fig. 15.4) [40, 41]. Quigley has hypothesized a vicious circle of elevated pressure, vitreous compaction and dehydration, resulting in a further reduction in fluid conductivity, and shallowing of the chamber [42].
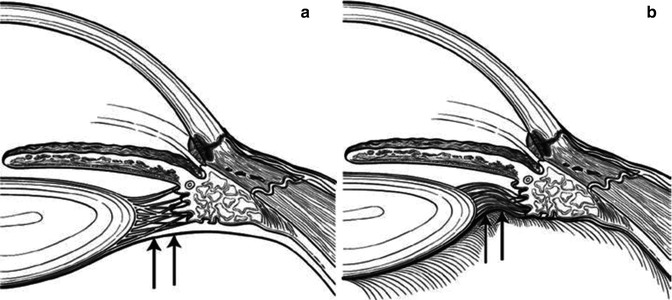

Fig. 15.4




Expansion of the vitreous volume in an eye with a relatively impermeable and diminished anterior hyaloid surface. (a) Expansion of the vitreous volume results in movement of the peripheral anterior hyaloid into apposition with the posterior ciliary body. (b) Vitreous expansion decreases the hyaloid face available for fluid flow
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


