Purpose
To evaluate the performance of ganglion cell layer/inner plexiform layer (GCL/IPL) measurements with spectral-domain optical coherence tomography (Cirrus HD-OCT) for detection of early glaucoma and to compare results to retinal nerve fiber layer (RNFL) measurements.
Design
Cross-sectional prospective diagnostic study.
Methods
We enrolled 99 subjects, including 59 eyes with glaucoma (47 subjects) (mean deviation >−6.0 dB) and 91 normal eyes (52 subjects). Patients underwent biometry and peripapillary and macular OCT imaging. Performance of the GCL/IPL and RNFL algorithms was evaluated with area under receiver operating characteristic curves (AUC), likelihood ratios, and sensitivities/specificities adjusting for covariates. Combination of best parameters was explored.
Results
Average (SD) mean deviation in the glaucoma group was −2.5 (1.9) dB. On multivariate analyses, age ( P < 0.001) and axial length ( P = 0.03) predicted GC/IPL measurements in normal subjects. No significant correlation was found between average or regional GC/IPL thickness and respective outer retina (OR) thickness measurements ( P > 0.05). Average RNFL thickness performed better than average GCL/IPL measurements for detection of glaucoma (AUC = 0.964 vs 0.937; P = 0.04). The best regional measures from each algorithm (inferior quadrant RNFL vs minimum GCL/IPL) had comparable performances ( P = 0.78). Entering the GC/IPL to OR ratio into prediction models did not enhance the performance of the GCL/IPL measures. Combining the best parameters from each algorithm improved detection of glaucoma ( P = 0.04).
Conclusions
Regional GCL/IPL measures derived from Cirrus HD-OCT performed as well as regional RNFL outcomes for detection of early glaucoma. Using the GC/IPL to OR ratio did not enhance the performance of GCL/IPL parameters. Combining the best measures from the 2 algorithms improved detection of glaucoma.
The hallmark of glaucoma is loss of the retinal ganglion cell axons that leads to a typical optic neuropathy. Qualitative and quantitative evaluation of the optic nerve head and the retinal nerve fiber layer (RNFL) has been used to detect glaucomatous damage. There is evidence that macular ganglion cell loss can be demonstrated in early experimental glaucoma. In addition, recent publications suggest that evidence of glaucomatous damage can be observed in the inner retina or ganglion cell complex (GCC), ie, the combined thickness of the RNFL, the ganglion cell layer (GCL), and the inner plexiform layer (IPL), early during the disease process by means of spectral domain optical coherence tomography (SD-OCT). A recent study showed that GCC loss could be detected in eyes with preperimetric glaucoma. It has been suggested that the macular GCL thickness may be the most relevant parameter to measure in glaucoma. However, given the resolution of the current generation of SD-OCTs, accurate measurement of the GCL alone is not practical. Hence, some SD-OCT manufacturers have developed segmentation algorithms for calculating the GCC thickness or the combined thickness of the GCL and IPL. The software versions 6.0 or higher of Cirrus High-Definition OCT (Cirrus HD-OCT, Carl Zeiss Meditec, Dublin, California, USA) now provide a ganglion cell analysis in which GCL/IPL thickness measurements and various graphic and statistical analyses are provided. There is preliminary evidence that such GCL/IPL measurements may perform as well as RNFL measures for detection of early glaucoma. Prior studies have reported age and axial length to be the main factors affecting variability of the GCL/IPL or GCC measurements. A recent study found that the GCC/total retinal thickness (TRT) ratio performed better than all other RNFL or macular parameters for detection of glaucoma. This finding, if confirmed, suggests that the thickness of the inner retinal tissues is related to the density and thickness of the outer retinal cells and, therefore, the ratio of the inner to outer retinal (OR) layers may be a useful parameter to include in algorithms used for detection of glaucoma. The goal of the current study is to explore factors affecting the GCL/IPL thickness in a group of normal eyes from the University of California, Los Angeles (UCLA), OCT Imaging Study, so as to evaluate the performance of the newly defined GCL/IPL measurements for detection of early glaucoma and to compare the findings to RNFL thickness measurements. We also hypothesized that the outer retina thickness may predict the GCL/IPL thickness, so including the OR thickness or the GCL/IPL/OR ratio in the prediction models using macular measurements may improve their performance.
Methods
This study was approved by the institutional review board at the UCLA and was carried out in accordance with the Declaration of Helsinki. We prospectively recruited 59 eyes of 47 glaucoma subjects and 91 eyes of 52 normal subjects between December 2010 and October 2012. Patients with clinical diagnoses of open-angle glaucoma made by an attending ophthalmologist were prospectively identified and invited to be enrolled in the study if they met the following criteria: age ≥30 years, open angles, visual acuity ≥20/80, visual field mean deviation (MD) ≥−6 dB, refractive error ≤8.0 diopters (D) and astigmatism ≤3 D. Eyes with evidence of retinal or neurologic diseases or prior glaucoma surgery were excluded. All patients had at least 1 prior visual field examination before being enrolled in the study. Normal subjects were recruited by advertising on UCLA’s campus, placing fliers in the clinics, and soliciting spouses and friends of patients seen at the Glaucoma Clinic, Jules Stein Eye Institute. The enrolled normal subjects were required to have open angles, corrected visual acuity of 20/25 or better, and normal eye examinations, including normal visual fields, and not having definitive evidence of glaucomatous damage at the level of the optic nerve head (see below).
All subjects underwent a thorough eye examination on the day of imaging, including visual acuity, automated refraction, measurement of intraocular pressure (IOP), gonioscopy, slit-lamp exam, dilated fundus exam, and standard achromatic perimetry (SAP) or short-wavelength automated perimetry (SWAP) fields. Axial length and keratometry were measured by IOLMaster (Carl Zeiss Meditec), and those test results were considered reliable if the signal to noise ratio for individual scans was above 3. Central corneal thickness measurements were measured with a DGH 55 Pachmate (DGH Technology, Exton, Pennsylvania, USA). The device’s output is the average of 10 reliable measurements of the central corneal thickness. Stereoscopic disc photographs and Optic Disc and Macular Cubes 200 × 200 (Cirrus HD-OCT) were carried out after pupillary dilation. All the images were reviewed afterward by one of the investigators, and images with signal strength <7, lost data on the peripapillary ring, obvious motion artifact, or incorrect segmentation were excluded.
The Optic Disc Cube 200 × 200 consists of 40,000 axial scans (in a 6 × 6 × 2 mm cube) centered on the optic disc. Average RNFL thickness and RNFL thickness in quadrants and clock-hour sectors on a measurement circle 3.46 mm in diameter are calculated, and their deviation from a normative database is provided in a color-coded scheme. RNFL pseudocolor thickness maps and deviation maps for the 6 × 6 mm area are also provided. The Macular Cube 200 × 200 algorithm measures 40,000 axial scans (in a 6 × 6 × 2 mm cube) centered on the fovea. The ganglion cell analysis available on the Cirrus software version 6.0 (or higher) measures the combined thickness of the GCL and IPL in a 4.8 × 4.0 mm oval with a longer horizontal axis. The GCL/IPL layer is thickest in this area, based on postmortem and in vivo measurements. Similar to other Cirrus printouts, it provides GCL/IPL measurements in 6 wedge-shaped sectors after excluding the central foveolar region (1 mm in diameter) along with a pseudocolor scheme for the GCL/IPL thickness. A deviation map also flags abnormally thin areas within the oval area as yellow ( P < 5%) or red ( P < 1%) superpixels.
Eyes with evidence of reproducible visual field loss consistent with glaucoma on SAP or SWAP visual fields, regardless of IOP or disc appearance, were considered to have glaucoma. Swedish Interactive Thresholding Algorithm standard testing strategy was used for both SAP and SWAP tests. Only eyes with reliable visual fields (false-positive rate of 15% or less) were included. Fixation loss and false-negative error rates were not used as criteria for reliability for fields obtained on the day of enrollment. An abnormal SAP or SWAP visual field was defined as presence of a glaucoma hemifield test outside normal limits and the presence of ≥4 abnormal test locations on a pattern deviation plot, with P < 5% both confirmed at least once. These criteria have been shown to be highly specific and have demonstrated reasonable sensitivity for detection of early glaucomatous visual field loss. The visual fields were reviewed to exclude lid or lens artifacts.
The RNFL and GCL/IPL thickness measurements from the Optic Disc and Macular 200 × 200 cubes were exported to a personal computer. Factors potentially affecting the GCL/IPL thickness in normal control subjects were explored with scatter plots, univariate regression, and 2-by-2 tables. We used spline methods and examined scatter plots to determine whether the relationship between thickness measurements and the continuous predictors were linear. Average RNFL thickness, RNFL measurements in quadrants, RNFL thickness in 12 clock-hour sectors, and average, minimum, and regional GCL/IPL measurements were compared in the glaucoma and control groups.
Binary multivariate logistic regression analyses (backward stepwise) were carried out to adjust for the influence of covariates affecting the GC/IPL or RNFL measurements and correcting for potential correlation of the 2 eyes of the same subjects. Covariates were kept in the models if the P value was <0.15. Average and regional RNFL and GC/IPL measurements were explored individually for discrimination between patients with glaucoma and subjects without glaucoma. The sensitivity, specificity and accuracy (accuracy = [sensitivity + specificity]/2) and areas under receiver operating characteristic (ROC) curves (AUCs), the coefficients for the partial AUCs, and their corresponding standard errors are reported.
The multivariate logistic regressions were adjusted for age, SD-OCT signal strength, disc size, and axial length. This regression was the basis of the adjusted ROC curves where the optimal threshold separating the 2 groups was allowed to vary depending on the covariates. We also explored potential interactions between diagnosis (normal vs glaucoma) and the other covariates in the regression models. We investigated which of the above factors had an influence on thickness and computed the adjusted sensitivity, specificity, accuracy, and AUCs under this model. In addition to the total AUCs, we used partial areas under the ROC curves to compare performance of various parameters at high specificity (such as 95%). Positive and negative likelihood ratios (LRs) were calculated for global and regional findings on the SD-OCT printouts to compare the performance of various parameters as a function of the normative database of the Cirrus HD-OCT. The software Stata (v 12.1, StataCorp, College Station, Texas, USA) was used for all data and statistical analyses.
Results
A total of 150 eyes of 99 subjects (59 eyes of 47 subjects with early perimetric glaucoma; and 91 control eyes belonging to 52 subjects) were enrolled. Table 1 describes the demographic characteristics of the study sample in detail. The control subjects were younger and had shorter axial length on average ( P < 0.001 for both). On univariate analyses, the only factors found to be associated with thinner average RNFL thickness in the control group were older age ( P = 0.01), longer axial length ( P = 0.012), and potentially smaller disc area ( P = 0.073), whereas the SD-OCT signal strength ( P = 0.305) and spherical equivalent ( P = 0.344) did not show any association with the average RNFL thickness. On multivariate analyses, older age (beta = −0.43 μ per year; P = 0.003); longer axial length (beta = −2.11 μ per mm; P = 0.018); and smaller disc area (beta = 1.19 μ per 0.1 mm 2 ; P = 0.056) were associated with thinner average RNFL measurements in normal eyes.
| Variables | Normal Subjects | Glaucoma Patients | P value |
|---|---|---|---|
| Number of eyes (patients) | 91 (52) | 59 (47) | |
| Age (mean ± SD, years) | 58.6 (±9.2) | 66.1 (±6.0) | <0.001 a |
| Gender (female/male) | 58/33 | 36/23 | 0.737 b |
| Race | 0.620 b | ||
| White | 39 (75.0%) | 36 (76.6%) | |
| African American | 4 (7.7%) | 6 (12.8%) | |
| Hispanic | 3 (5.8%) | 2 (4.3%) | |
| Asian | 6 (11.5%) | 3 (6.4%) | |
| LogMAR visual acuity (mean ± SD) | 0.0 (±0.1) | 0.1 (±0.1) | 0.061 a |
| Refractive error (diopters, mean ± SD) | −0.4 (±2.4) | −1.2 (±2.6) | 0.154 a |
| IOP (mm Hg, mean ± SD) | 14.7 (±2.9) | 14.1 (±3.8) | 0.314 a |
| Central corneal thickness (μm, mean ± SD) | 556.1 (±38.9) | 548.8 (±35.5) | 0.323 a |
| Keratometry (diopters, mean ± SD) | 44.2 (±1.5) | 43.9 (±1.4) | 0.260 a |
| Lens status | <0.001 b | ||
| Phakic | 91 (100%) | 42 (71.2%) | |
| Pseudophakic | 0 (0%) | 17 (28.8%) | |
| Axial length (mm, mean ± SD) | 23.8 (±1.1) | 24.7 (±1.2) | <0.001 a |
| Signal strength macular cube (mean ± SD) | 8.7 (±1.0) | 8.3 (±0.8) | 0.010 c |
| Signal strength optic disc cube (mean ± SD) | 8.8 (±1.0) | 8.0 (±0.8) | <0.001 c |
| Disc area (mm 2 , mean ± SD) | 1.73 (±0.3) | 1.73 (±0.4) | 0.922 a |
| Mean deviation (dB, mean ± SD) | −0.1 (±1.2) | −2.5 (±1.9) | <0.001 c |
| Pattern standard deviation (dB, mean ± SD) | 1.6 (±0.4) | 4.5 (±2.2) | <0.001 c |
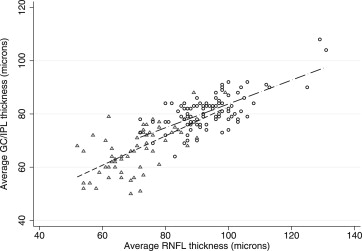
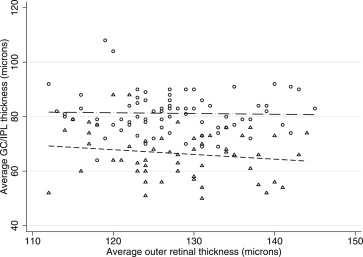
Thinner global GC/IPL measurements in control subjects were associated with advancing age ( P = 0.001), flatter keratometry ( P = 0.058), longer axial length ( P = 0.101), and male gender ( P = 0.149) on univariate analyses. Signal strength did not show a significant association with average GC/IPL thickness ( P = 0.339). On multivariate analyses, only age (beta = −0.28 μ per year; P = 0.001) and axial length (beta = −1.36 μ per mm; P = 0.03) predicted average GC/IPL thickness measurements in normal subjects. Interaction of age and axial length was not significant in multivariate analyses ( P = 0.140). A lower average GC/IPL thickness predicted a worse MD in the normal control group (beta =1.45 dB per micron, P = 0.017), but the association disappeared after correcting for age and axial length. The average GC/IPL thickness correlated significantly with the average RNFL thickness in both the normal and the glaucoma groups (r = 0.693 and = 0.628, respectively; P < 0.001 for both) ( Fig. 1 ), whereas the association with the disc area was not statistically significant (r = 0.121, P = 0.252 in the control group and r = −0.020, P = 0.876 in the glaucoma group). No significant association was found between the average or regional GC/IPL thickness and average or regional outer retina thickness measurements (r = −0.029, P = 0.801 for normal subjects and r = −0.156, P = 0.251 for glaucoma eyes for global measurements; P > 0.05 for all regional associations) ( Fig. 2 ).
Performance of the Two Algorithms for Detection of Glaucoma
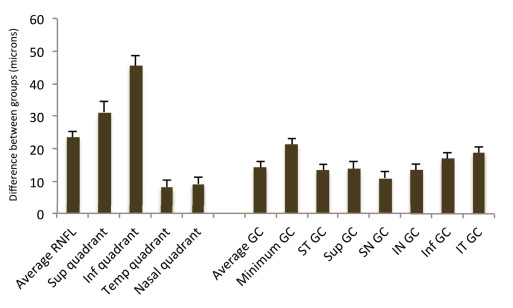
Table 2 compares the global and regional/sectoral thickness measurements for RNFL and GC/IPL between the 2 groups, and Figure 3 shows the distribution of the difference between glaucoma and control groups for the average and regional GC/IPL and RNFL thickness measurements. All potentially significant covariates (ie, parameters with P < 0.15 on univariate analyses or factors that make biological sense, such as disc area) were included in the final stepwise multivariate analyses along with each global or regional RNFL or GC/IPL predictor and logit scores were calculated for each outcome to calculate AUCs. The final logistic models included age, signal strength, axial length, and disc size as covariates. The outer retina thickness or the ratio of GC/IPL to outer retina thickness was not a significant covariate in any of the global or regional multivariate models ( P > 0.6 for all). The AUCs, sensitivities/specificities, and accuracies for the global and regional GC/IPL and RNFL thickness measures are presented in Table 3 . As is evident in Figures 4 and 5 , the AUC for the average RNFL thickness was superior to average GC/IPL measurements (AUC = 0.964; 95% CI 0.936-0.991 vs 0.937; 95% CI: 0.895-0.980; P = 0.04). The partial AUC (pAUC) difference between the average RNFL and average GC/IPL thickness measurements with the cutoff point placed at 95% approached statistical significance ( P = 0.057) ( Fig. 4 ). The performance of the minimum and inferotemporal GC/IPL thickness (the best performing regional GC/IPL measures) was similar to that of the inferior quadrant RNFL thickness, the best performing regional RNFL outcome (AUCs = 0.962 [95% CI: 0.915-0.985] vs 0.955 [95% CI: 0.906-0.981] vs 0.977 [95% CI: 0.943-0.996], P = 0.71 and = 0.23, respectively; P for pAUC difference of minimum and inferotemporal GC/IPL vs the inferior quadrant RNFL = 0.77 and 0.82, respectively). When sensitivity and specificities were evaluated, the sensitivity for the minimum GC/IPL thickness or inferotemporal GC/IPL sector (81.3%, 95% CI: 70.9%-91.7% and 84.9%, 95% CI: 75.5%-94.3%, respectively) were comparable to but lower than that of the inferior quadrant RNFL (89.8%, 95% CI: 82.0-97.6%) when compared at high specificity (≥95%). As mentioned above, the ROCs and sensitivities/specificities were all adjusted for the confounding factors listed above. We also hypothesized that the adding the best regional GC/IPL parameter to the best regional RNFL parameter could potentially improve discrimination of glaucomatous from normal eyes although, given the very good performance of both parameters, a ceiling effect would be expected. We found that combining the two best GC/IPL and RNFL parameters actually improved the performance of the SD-OCT. The pseudo-R 2 for the logistic models improved from 0.655 to 0.756, and the partial AUC coefficient with the cutoff point at 95% specificity was actually better for the model including the combined parameters vs the model including only the inferior quadrant (difference in observed pAUC coefficients: 0.005, 95% CI: 0.0002-0.010; P = 0.041) ( Fig. 6 ).
| OCT Parameters | Normal Group | Glaucoma Subjects | P value |
|---|---|---|---|
| GC/IPL parameters | |||
| Average GC/IPL (mean ± SE) | 81.1 (±1.0) | 66.5 (±1.3) | <0.001 |
| Minimum GC/IPL (mean ± SE) | 79.2 (±0.9) | 57.8 (±1.4) | <0.001 |
| Superotemporal GC/IPL (mean ± SE) | 80.5 (±0.9) | 66.9 (±1.4) | <0.001 |
| Superior GC/IPL (mean ± SE) | 81.8 (±1.0) | 67.9 (±1.7) | <0.001 |
| Superonasal GC/IPL (mean ± SE) | 82.3 (±1.1) | 71.2 (±1.8) | <0.001 |
| Inferonasal GC/IPL (mean ± SE) | 80.6 (±1.0) | 67.2 (±1.7) | <0.001 |
| Inferior GC/IPL (mean ± SE) | 79.9 (±1.0) | 63.1 (±1.4) | <0.001 |
| Inferotemporal GC/IPL (mean ± SE) | 81.7 (±0.9) | 62.9 (±1.4) | <0.001 |
| RNFL parameters | |||
| Average RNFL (mean ± SE) | 94.0 (±1.1) | 70.5 (±1.3) | <0.001 |
| Superior quadrant (mean ± SE) | 115.7 (±1.7) | 84.3 (±2.4) | <0.001 |
| Nasal quadrant (mean ± SE) | 72.9 (±1.2) | 63.6 (±1.2) | <0.001 |
| Inferior quadrant (mean ± SE) | 124.0 (±1.9) | 78.6 (±2.1) | <0.001 |
| Temporal quadrant (mean ± SE) | 63.5 (±1.2) | 55.5 (±1.5) | <0.001 |
| 1 o’clock sector (mean ± SE) | 104.8 (±2.3) | 79.7 (±2.7) | <0.001 |
| 2 o’clock sector (mean ± SE) | 89.7 (±1.9) | 76.5 (±3.7) | <0.001 |
| 3 o’clock sector (mean ± SE) | 61.7 (±1.2) | 58.9 (±1.3) | 0.021 |
| 4 o’clock sector (mean ± SE) | 66.5 (±1.3) | 58.6 (±1.3) | <0.001 |
| 5 o’clock sector (mean ± SE) | 100.9 (±2.4) | 74.7 (±2.3) | <0.001 |
| 6 o’clock sector (mean ± SE) | 136.6 (±2.8) | 86.3 (±3.2) | <0.001 |
| 7 o’clock sector (mean ± SE) | 134.3 (±2.7) | 74.9 (±3.4) | <0.001 |
| 8 o’clock sector (mean ± SE) | 62.7 (±1.5) | 55.2 (±1.8) | 0.008 |
| 9 o’clock sector (mean ± SE) | 51.4 (±1.1) | 49.8 (±1.4) | 0.417 |
| 10 o’clock sector (mean ± SE) | 76.8 (±1.6) | 61.5 (±2.2) | <0.001 |
| 11 o’clock sector (mean ± SE) | 124.8 (±2.3) | 89.7 (±3.1) | <0.001 |
| 12 o’clock sector (mean ± SE) | 117.8 (±2.8) | 83.6 (±3.0) | <0.001 |
| AUC (95% CI) | Sensitivity | Specificity | Accuracy | AUC (95% CI) | Sensitivity | Specificity | Accuracy | ||
|---|---|---|---|---|---|---|---|---|---|
| RNFL parameters | |||||||||
| Average RNFL | 0.964 (0.936-0.991) | 88.1% | 91.2% | 90.0% | |||||
| Superior quadrant | 0.936 (0.898-0.974) | 84.8% | 86.8% | 86.0% | Nasal quadrant | 0.889 (0.837-0.942) | 74.6% | 85.7% | 81.3% |
| Inferior quadrant | 0.976 (0.952-0.999) | 93.2% | 91.2% | 92.0% | Temporal quadrant | 0.880 (0.826-0.933) | 71.2% | 82.4% | 78.0% |
| 1 o’clock sector | 0.896 (0.844-0.947) | 81.4% | 83.5% | 82.4% | 2 o’clock sector | 0.875 (0.815-0.936) | 93.2% | 80.2% | 86.7% |
| 3 o’clock sector | 0.866 (0.809-0.923) | 83.1% | 80.2% | 81.6% | 4 o’clock sector | 0.877 (0.821-0.932) | 88.1% | 75.8% | 82.0% |
| 5 o’clock sector | 0.906 (0.858-0.953) | 91.5% | 75.8% | 83.7% | 6 o’clock sector | 0.952 (0.917-0.987) | 91.5% | 86.8% | 89.2% |
| 7 o’clock sector | 0.964 (0.933-0.995) | 93.2% | 93.4% | 93.3% | 8 o’clock sector | 0.867 (0.810-0.923) | 96.6% | 62.6% | 79.6% |
| 9 o’clock sector | 0.861 (0.802-0.920) | 81.4% | 82.4% | 81.9% | 10 o’clock sector | 0.898 (0.847-0.949) | 91.5% | 76.9% | 84.2% |
| 11 o’clock sector | 0.919 (0.876-0.961) | 88.1% | 82.4% | 85.3% | 12 o’clock sector | 0.907 (0.860-0.955) | 81.4% | 90.1% | 85.7% |
| GC/IPL parameters | |||||||||
| Average GC/IPL | 0.937 (0.889- 0.972) | 86.4% | 87.9% | 87.3% | Minimum GC/IPL | 0.962 (0.915-0.985) | 91.5% | 94.5% | 93.3% |
| Superior GC/IPL | 0.906 (0.848-0.948) | 76.3% | 86.8% | 85.0% | Inferior GC/IPL | 0.938 (0.889-0.972) | 83.1% | 91.2% | 88.0% |
| Inferonasal GC/IPL | 0.891 (0.833-0.938) | 72.9% | 82.4% | 78.7% | Superotemporal GC/IPL | 0.926 (0.873-0.963) | 83.3% | 74.6% | 89.0% |
| Inferotemporal GC/IPL | 0.955 (0.906-0.981) | 86.4% | 94.5% | 91.3% | Superonasal GC/IPL | 0.871 (0.809-0.922) | 71.2% | 83.5% | 78.7% |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


