Purpose
To determine frequency and associations of macular Bruch membrane defects in the region of macular atrophy developing after the onset of myopic choroidal neovascularization (CNV).
Design
Retrospective observational case series.
Methods
The study included all patients who were consecutively examined for high myopia (axial length ≥26.5mm) and CNV-related macular atrophy in the study period from June to July 2015. The patients underwent a comprehensive ophthalmologic examination including swept-source optical coherence tomography (OCT) of the macula. Main outcome measures were macular Bruch membrane defects.
Results
Out of 33 eyes (28 patients) with myopic CNV-related macular atrophy, 25 eyes (76%) showed macular Bruch membrane defects, which were characterized by a lack of Bruch membrane, retinal pigment epithelium, photoreceptors, and choriocapillaris. At the edges of the macular Bruch membrane defects, the ends of the Bruch membrane were upturned, and an inward protrusion of large choroidal vessels could be detected. In the center of macular Bruch membrane defects, remnants of Bruch membrane could be crumpled. In multivariate analysis, higher prevalence of secondary macular Bruch membrane defects was significantly associated with a lower prevalence of intravitreal medical therapy ( P < .001) after adjusting for larger macular atrophy area size ( P < .001) and longer interval between development of the CNV and final examination ( P = .42).
Conclusions
Macular Bruch membrane defects belong to the hallmarks of myopic CNV-related macular atrophy. Since macular Bruch membrane defects lack photoreceptors and thus represent psychophysically an absolute scotoma, they are of profound importance for visual prognosis. As incidentally observed at study end, the prevalence of macular Bruch membrane defects may be lower if a previous myopic CNV was treated by intravitreal medical therapy.
It has generally been assumed that the Bruch membrane in highly myopic eyes remained intact in the area of a myopic macular chorioretinal atrophy despite the loss of retinal pigment epithelium (RPE), closure of choriocapillaris, and loss of photoreceptors. Examining axially elongated human globes histologically revealed, however, that highly myopic eyes not only showed a widening of the physiologic Bruch membrane opening at the optic nerve head with parapapillary gamma zone (defined as the parapapillary region without Bruch membrane) but also could present with defects in the Bruch membrane in the macular region. Subsequent studies addressed the prevalence of these secondary macular Bruch membrane defects and their associations with other ocular and systemic parameters.
Macular choroidal neovascularization (CNV) is a major cause of vision impairment in patients with pathologic myopia. The natural history of myopic CNV is poor, with almost 90% of patients achieving a best-corrected visual acuity of ≤0.10 at 5 years after the onset of the CNV. The long-term decrease in vision after development of a myopic CNV has been suggested to be due to the development and enlargement of a “CNV-related macular atrophy” ( Figure 1 ). Among various lesions of myopic maculopathy, the fundus appearance of CNV-related macular atrophy is similar to so-called “patchy atrophy.” However, different from patchy atrophy, CNV-related macular atrophy is centered on the fovea and involves the central fovea ( Figure 1 ). Anti–vascular endothelial growth factor (VEGF) therapy has been established as a first-line therapy for myopic CNV based on the results of large clinical trials. However, some of the recent studies reported that the visual outcome at a follow-up longer than 4 years after application of the intravitreal medical therapy was unfavorable. Farinha and associates showed that the long-term functional outcome was dependent on the progression of CNV-related macular atrophy in eyes treated with anti-VEGF reagents as it was reported in untreated patients in earlier studies.
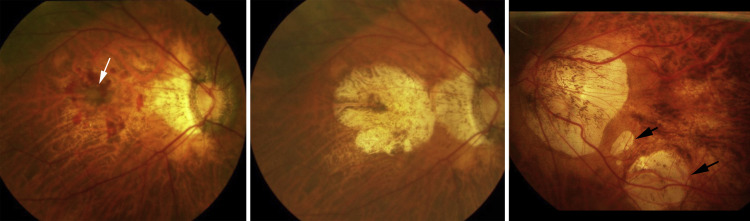
Despite its importance, the pathologic anatomy of a CNV-related macular atrophy has not fully been explored yet. Since macular Bruch membrane defects lack photoreceptors and thus psychophysically represent an absolute scotoma, they are of profound importance for visual prognosis. However, potential associations between the presence of secondary macular Bruch membrane defects and CNV-related macular atrophy have not been addressed yet. We therefore conducted the present study to examine frequency and associations of macular Bruch membrane defects in the region of CNV-related macular atrophy, which is a late complication occurring around the myopic CNV.
Methods
The retrospective, observational case series included 33 eyes of 28 patients (3 men) with CNV-related macular atrophy in eyes with pathologic myopia. The patients had consecutively been examined for high myopia (defined as an axial length ≥26.5 mm) and CNV-related macular atrophy in the High Myopia Clinic of the Tokyo Medical and Dental University in the study period between June 26, 2015 and July 18, 2015. The study was in agreement with the tenets of the Declaration of Helsinki and was approved by the Ethics Committee of Tokyo Medical and Dental University. All patients included in the study had been followed regularly since the onset of myopic CNV. All patients underwent a comprehensive ophthalmologic examination including refractometry, measurement of axial length (IOL Master; Carl Zeiss Meditec Co, Jena, Germany), color fundus photography, and assessment of fundus autofluorescence (TRC-50DX; Topcon, Tokyo, Japan). The mean age was 65.8 ± 10.8 years (range: 45–84 years), and mean axial length was 29.4 ± 1.2 mm (range: 27.4–32.0 mm).
The diagnosis of a CNV-related macular atrophy was based on the findings of the examination of the fundus autofluorescence. Based on fundus autofluorescence images, the area of the macular atrophy was measured using the software of the imaging system (PDT/MPS software; Topcon, Tokyo, Japan). According to Kojima and associates, the size of macular atrophy was expressed as a ratio to disc area, and was expressed as (area of macular atrophy − area of CNV)/disc area.
The mean period after clinical onset of the CNV was 96.4 ± 52.6 months (range: 24–233 months). The mean size of macular atrophy was 3.5 ± 2.2 disc areas (0.3–9.0 disc areas). Fifteen eyes (45%) had undergone therapy for myopic CNV at the onset of clinical symptoms. Nine eyes had received a single intravitreal injection of ranibizumab, 4 eyes a single intravitreal injection of bevacizumab, and 2 eyes had received 1 injection of ranibizumab and 1 injection of bevacizumab. The remaining 18 eyes (55%) had not undergone any therapy, since they had developed CNV before anti-VEGF therapies had become available (photodynamic therapy has not been available for myopic CNV in Japan).
The Bruch membrane was examined using a swept-source optical coherence tomography (OCT) (DRI-OCT; Topcon, Tokyo, Japan). As scanning protocols, 9 mm or 6 mm radial scans centered on the fovea were performed. The 96 single images were registered and averaged by the software to create a multi-averaged single image. Each image consisted of 1024 A-scans. The Bruch membrane in the area of macular atrophy was investigated by a single examiner (K.O.M.). The OCT assessment also included the examination of the RPE and photoreceptors.
The main outcome measures were the prevalence of Bruch membrane defects in the area of CNV-related macular atrophy. We also performed statistical comparisons of age, sex, axial length, and history of anti-VEGF therapies between the eyes with intact Bruch membrane over the entire area of CNV-related macular atrophy and the eyes with discontinuous Bruch membrane. The statistical analysis was performed using a commercially available statistical software program (SPSS 22.0; IBM-SPSS, Chicago, Illinois, USA). In a first step, we calculated the means and standard deviations of the parameters. In a second step, we examined associations between the parameters in a univariate manner. The Mann-Whitney U test was performed to assess the statistical significance of the difference between the groups in age, axial length, period after CNV onset, and size of macular atrophy. The Fisher exact probability test was applied to compare the frequency of macular Bruch membrane defects and a history of anti-VEGF therapy. Finally, we performed a multivariate binary regression analysis that included the presence of a macular Bruch membrane defect as a dependent variable and as independent variables all those parameters for which the P value for the univariate association with the presence of macular Bruch membrane defect was less than .10. In a step-wise manner, we then dropped those parameters that were no longer significantly associated with the presence of macular Bruch membrane defects. A P value of <.05 was considered to be statistically significant.
Results
In the OCT images, the Bruch membrane was intact and continuous over the entire area of macular atrophy in all of the 12 radial scans in 8 eyes (24%). In the remaining 25 eyes (76%), the Bruch membrane showed discontinuities in the area of macular atrophy ( Figures 2–4 ). Comparing the 25 eyes with macular Bruch membrane defects with the 8 eyes without rupture of the Bruch membrane showed no significant difference in age (67.4 ± 9.8 years vs 61.1 ± 13.0 years; P = .24), axial length (29.5 ± 1.1 mm vs 29.4 ± 1.4 mm; P = .87), and sex ( P > .99).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


