Purpose
To assess microvascular blood flow of the deep retinal capillary plexus in eyes with paracentral acute middle maculopathy using optical coherence tomography (OCT) angiography.
Design
Retrospective, multicenter observational case series.
Methods
Clinical and multimodal imaging findings from 8 patients with paracentral acute middle maculopathy were reviewed and analyzed. OCT angiography scans were analyzed and processed, and vessel density was calculated.
Results
Eight patients (7 male, 1 female, aged 9–82 years) were included. OCT angiography was obtained at either the acute (4 cases) or old stage (4 cases). Scans of the deep capillary plexus showed preservation of perfusion in acute lesions and capillary attenuation in old cases. Cases of central retinal artery occlusion showed marked loss of the deep capillary plexus. The mean vessel density of the superficial capillary plexus in normal fellow eyes was 12.8 ± 1.8 mm −1 vs 12.1 ± 1.9 mm −1 in eyes with paracentral acute middle maculopathy (reduction −6.0%, P = .08). The mean vessel density of the deep capillary plexus in normal fellow eyes was 17.5 ± 1.4 mm −1 vs 14.7 ± 3.5 mm −1 in eyes with paracentral acute middle maculopathy (reduction −19.4%, P = .04). This significant difference was representative of the eyes with old lesions.
Conclusion
Paracentral acute middle maculopathy lesions correspond to preservation of perfusion in focal acute lesions and to pruning of the plexus in old cases. Cases of central retinal artery occlusion demonstrate marked hypoperfusion of the deep capillary plexus. Our study further supports an ischemic pathogenesis of this retinal vasculopathy.
Paracentral acute middle maculopathy is a recently described entity in patients presenting with an acute-onset paracentral scotoma. Spectral-domain optical coherence tomography (OCT) reveals hyperreflective band-like lesions at the level of the inner nuclear layer. Although these lesions resolve, patients are left with atrophy of the inner nuclear layer, resulting in a permanent paracentral visual field defect. Paracentral acute middle maculopathy can be idiopathic, or it can be secondary to local retinal vascular or systemic disease.
Deep retinal capillary ischemia has been proposed as a causative factor in the development of these lesions, as the intermediate and deep retinal capillary plexuses flank the inner and outer boundaries of the inner nuclear layer, respectively. Fluorescein angiography, the reference standard for visualizing the retinal vasculature, has limited depth resolution. With OCT angiography, it is possible to obtain high-resolution, depth-resolved en face images of the retinal microvasculature by calculating motion contrast in OCT B-scans acquired repeatedly at the same location. OCT angiograms are co-registered with OCT B-scans from the same location, allowing simultaneous visualization of structure and blood flow. We used OCT angiography to study the retinal capillary microvasculature at different levels of the retina in 8 cases of paracentral acute middle maculopathy occurring in various clinical scenarios.
Methods
Approval for this study was obtained from the University of California Los Angeles (UCLA) Institutional Review Board committee. Research adhered to the tenets of the Declaration of Helsinki and was conducted in accord with regulations set forth by the Health Insurance Portability and Accountability Act.
We retrospectively identified 8 patients from 3 tertiary referral centers. All were evaluated between August 2014 and July 2015 and diagnosed with paracentral acute middle maculopathy. Written informed consent was obtained for each patient. We reviewed the clinical and multimodal imaging data for each patient and obtained OCT angiography analysis. The diagnostic criteria of paracentral acute middle maculopathy included a history of acute-onset paracentral scotoma with or without decline in visual acuity and a nonprogressive course. All patients demonstrated characteristic abnormalities with spectral-domain OCT, including either the acute finding of hyperreflective, plaque-like bands within the inner nuclear layer or old lesions demonstrating thinning of the inner nuclear layer. High-resolution digital color imaging, red-free photography, spectral-domain OCT, and OCT angiography were performed at the time of presentation for each patient. Fluorescein angiography was obtained in 7 of the 8 cases.
The AngioVue OCT angiography system (Optovue, Inc, Fremont, California, USA) operates at 70, 000 A-scans per second to acquire OCT angiography volumes consisting of 304 × 304 A-scans in approximately 2.6 seconds. It uses a split-spectrum amplitude decorrelation angiography software algorithm and orthogonal registration and merging of 2 consecutive scan volumes to obtain 3 × 3 mm and 6 × 6 mm OCT angiography volumes of both eyes of each patient. OCT angiograms were co-registered with OCT B-scans, to allow visualization of both retinal vasculature and structure, and were performed in both the normal and affected eyes of each patient.
The OCT angiography software was used to segment the superficial capillary plexus and deep capillary plexus in 3 × 3 mm scans. Consistently in each patient, the superficial capillary plexus slab was taken from the internal limiting membrane (offset 3 μm) to the inner plexiform layer (offset 15 μm). The deep capillary plexus slab was taken from the inner plexiform layer (offset 15 μm) to the outer plexiform layer (offset 70 μm). In cases where thinning of the inner nuclear layer caused failure of automatic segmentation, a thinner 30 μm band was manually adjusted to include the deep capillary plexus. OCT angiography analysis of the superficial and deep capillary plexus was performed independently by a trained reader to assess for capillary attenuation and pruning involving either plexus.
Quantitative analyses were performed using the publicly available software Fiji ImageJ 2.0.0-rc-29/1.49q ( http://fiji.sc/Fiji ) and GNU Image Manipulation Program GIMP 2.8.14 ( http://gimp.org ). Fiji was used to binarize and skeletonize the en face image of the superficial and deep retinal capillary plexus, showing the blood vessels as a 1-pixel-wide line. GIMP was used to count the number of black pixels and total pixels. Vessel density was then calculated as [(pixels of vessels) (3/304)]/(area in mm 2 ) in mm −1 . In cases of partial or complete projection artifact from the superficial capillary plexus, Fiji was used to binarize the superficial and deep capillary plexus images. GIMP was then used to subtract the black pixels of the superficial capillary plexus from the deep capillary plexus image. The resulting deep capillary plexus was skeletonized using Fiji software, and pixels were calculated using GIMP. Vessel density calculation was then repeated in the manner described above.
Results
Nine eyes with paracentral acute middle maculopathy (8 patients: 7 male, 1 female) were identified and enrolled in this study. Clinical characteristics are summarized in Table 1 . Patient ages ranged from 9 to 82 years (mean 49.5 years, median 60). Isolated band-like hyperreflective lesions in the middle retinal layers consistent with acute paracentral middle maculopathy lesions were observed with spectral-domain OCT imaging in 5 patients at baseline. Three patients presented with patchy thinning of the inner nuclear layer on spectral-domain OCT, consistent with old paracentral acute middle maculopathy. Retinal vascular etiologies leading to paracentral acute middle maculopathy included central retinal artery occlusion (CRAO, n = 4), branch retinal artery occlusion (BRAO, n = 2), suction eye injury (n = 1), and sickle cell retinopathy (n = 1).
| Case | Sex | Age (y) | Affected Eye | BCVA | Ophthalmologic Diagnosis | Systemic Disease |
|---|---|---|---|---|---|---|
| 1 | Male | 66 | OD | 20/50 | CRAO | HTN; type II DM; hypothyroidism |
| 2 | Male | 72 | OD | 20/20 -1 | BRAO | HTN; atrial fibrillation; mitral valve repair; pacemaker |
| 3 | Male | 31 | OS | 20/20 -1 | BRAO | Transposition of the great vessels (status post repair); head trauma 2 days prior |
| 4 | Male | 63 | OS | 20/30 | CRAO | Left middle cerebral artery infarction; HTN |
| 5 | Female | 57 | OS | 20/300 | CRAO | Internal carotid artery dissection; cerebral vascular occlusion coinciding with ocular event; meningioma; hyperlipidemia |
| 6 | Male | 82 | OS | CF at 4 ft | CRAO, NPDR | HTN; type II DM |
| 7 | Male | 16 | OU | 20/20 | SCR | Sickle cell disease (hemoglobin type SC) |
| 8 | Male | 9 | OD | 20/20 -1 | Trauma | Alpha-thalassemia trait |
OCT angiography was obtained at the acute stage of the lesion in 4 cases and at the old stage in 4 cases. The superficial capillary plexus demonstrated minimally attenuated perfusion in affected eyes. Focal acute cases (n = 2, Cases 2 and 3) demonstrated patent perfusion of the deep capillary plexus within the lesions, whereas focal old cases (n = 2, Cases 7 and 8) showed pruning of the deep capillary plexus within the lesions. One month follow-up analysis was obtained for Case 2 and demonstrated normal vascular density of the deep capillary plexus. Four cases of diffuse paracentral acute middle maculopathy associated with CRAO (Cases 1 and 4 with acute lesions, and Cases 5 and 6 with old lesions) demonstrated profound to complete absence of the deep capillary plexus with significant projection artifact from the superficial capillary plexus.
Quantitative analysis was carried out in all cases. Because the patient in Case 7 was affected bilaterally, and thus had no control fellow eye, Case 7 was excluded from statistical analysis. Table 2 includes the vessel density measurements for each eye (n = 9), and Table 3 shows a summary of the statistical analysis that was carried out comparing the patients with unilateral disease (n = 7). The mean vessel density of the superficial capillary plexus in normal fellow eyes was 12.8 ± 1.8 mm −1 vs a mean of 12.1 ± 1.9 mm −1 in eyes with paracentral acute middle maculopathy (reduction −6.0%, P = .33). The mean vessel density of the deep capillary plexus in normal fellow eyes was 17.5 ± 1.4 mm −1 vs a mean of 14.7 ± 3.5 mm −1 in eyes with paracentral acute middle maculopathy (reduction −19.4%, P = .04).
| Case | Affected Eye | Acute vs Chronic PAMM | Vessel Density (mm −1 ) | |||
|---|---|---|---|---|---|---|
| Superficial Capillary Plexus | Deep Capillary Plexus | |||||
| Normal Eye | PAMM Eye | Normal Eye | PAMM Eye | |||
| 1 | OD | Acute | 12.2 | 11.8 | 17.1 | 15.7 |
| 2 | OD | Acute | 12.9 | 12.5 | 17.6 | 17.8 |
| 3 | OS | Acute | 14.9 | 14.4 | 19.3 | 18.7 |
| 4 | OS | Acute | 9.7 | 11.7 | 16.4 | 13.9 |
| 5 | OS | Chronic | 13.1 | 10.9 | 16.8 | 11.0 |
| 6 | OS | Chronic | 12.2 | 9.1 | 15.8 | 9.2 |
| 7 | OD | Chronic | N/A | 15.0 | N/A | 21.3 |
| OS | Chronic | N/A | 13.1 | N/A | 19.4 | |
| 8 | OD | Chronic | 14.9 | 14.5 | 19.3 | 16.7 |
| Variable | Mean ± SD Vessel Density N = 7 | Mean ± SD Vessel Density in Acute Lesions N = 4 | Mean ± SD Vessel Density in Chronic Lesions N = 3 |
|---|---|---|---|
| Superficial capillary plexus normal eye (mm −1 ) | 12.8 ± 1.8 | 12.4 ± 2.1 | 13.4 ± 1.4 |
| Superficial capillary plexus PAMM eye (mm −1 ) | 12.1 ± 1.9 | 12.6 ± 1.2 | 11.5 ± 2.7 |
| Superficial capillary plexus percent reduction (%) | −6.0 ± 14.9 ( P = .33 a ) | +2.1 ± 10.9 | −16.9 ± 13.4 |
| Deep capillary plexus normal eye (mm −1 ) | 17.5 ± 1.4 | 17.6 ± 1.2 | 17.3 ± 1.8 |
| Deep capillary plexus PAMM eye (mm −1 ) | 14.7 ± 3.5 | 16.5 ± 2.1 | 12.3 ± 3.9 |
| Deep capillary plexus percent reduction (%) | −19.4 ± 20.1 ( P = .04 a ) | −6.8 ± 7.4 | −36.3 ± 19.7 |
Cases 1 through 4 were scanned at the acute stage of the paracentral acute middle maculopathy lesion (n = 4), and Cases 5 through 8 were scanned at the old stage of the lesion (n = 4). Focal acute lesions (n = 2, Cases 2 and 3) demonstrated preservation of perfusion of the deep capillary plexus (18.4 mm −1 ) compared with the normal fellow eyes (18.2 mm −1 ), whereas eyes with focal old lesions (n = 1, Case 8) demonstrated attenuation of the deep capillary plexus (16.7 mm −1 ) compared with the normal fellow eye (19.3 mm −1 ) ( Table 3 ). Localized vascular density analysis was also performed and compared between normal eyes and eyes with focal acute lesions. In Case 2, 33% of the 3 × 3 mm cube was involved, and the deep capillary density measured 6.3 mm −1 in the affected eye vs 6.5 mm −1 for the corresponding area in the unaffected eye. In Case 3, 25.6% of the 3 × 3 mm cube was involved, and capillary density in the affected area measured 4.8 mm −1 vs 5.2 mm −1 in the corresponding area of the unaffected, contralateral eye. This analysis confirmed the preservation of perfusion within the deep retinal capillary plexus for acute focal lesions.
The 4 cases of CRAO with both acute (n = 2) and old (n = 2) paracentral acute middle maculopathy lesions showed partial or complete loss of the deep capillary plexus with overlying projection artifact from the superficial capillary plexus (Cases 1, 4, 5, and 6). The degree of projection artifact was obtained by recalculating the capillary density and subtracting the density of the superficial capillary plexus from that of the deep capillary plexus. A summary of calculated vessel density of the resulting deep capillary plexus is shown in Table 4 . The mean vessel density of the deep plexus after subtraction in normal fellow eyes was 11.4 mm −1 vs 7.5 mm −1 in eyes with paracentral acute middle maculopathy and CRAO (reduction −41.2%) despite a relatively similar superficial vessel density in both groups.
| Case | PAMM Eye | Acute vs Chronic PAMM | Vessel Density of the Deep Capillary Plexus | |||||
|---|---|---|---|---|---|---|---|---|
| Normal Eye | PAMM Eye | |||||||
| Original (mm -1 ) | After Subtraction a (mm -1 ) | PR (%) | Original (mm -1 ) | After Subtraction a (mm -1 ) | PR (%) | |||
| 1 | OD | Acute | 17.1 | 11.8 | −36.6 | 15.7 | 10.0 | −44.5 |
| 4 | OS | Acute | 16.4 | 11.8 | −32.2 | 13.9 | 8.0 | −54.4 |
| 5 | OS | Chronic | 16.7 | 9.2 | −58.0 | 11.0 | 4.4 | −86.1 |
| 6 | OS | Chronic | 15.8 | 12.8 | −20.5 | 9.2 | 7.7 | −17.2 |
a Subtraction of the superficial capillary plexus projection artifact from the deep capillary plexus.
Representative cases (2, 3, 6, and 8) are described in detail below with corresponding figures.
Case 2
A 72-year-old man ( Figure 1 ) presented with a 3-day history of seeing a “red line” across his right eye. Past medical history was notable for hypertension, atrial fibrillation, mitral valve repair, and pacemaker placement. Best-corrected visual acuity (BCVA) was 20/20-1 in the right eye and 20/20-2 in the left eye. Fundus examination showed a patch of retinal whitening along the superior arcade and a normal retinal examination of the left eye. Spectral-domain OCT ( Figure 1 , Bottom row) showed numerous dense hyperreflective foci of the inner nuclear layer in band-like patches throughout the superior macula, consistent with paracentral acute middle maculopathy. An occult BRAO was suspected.
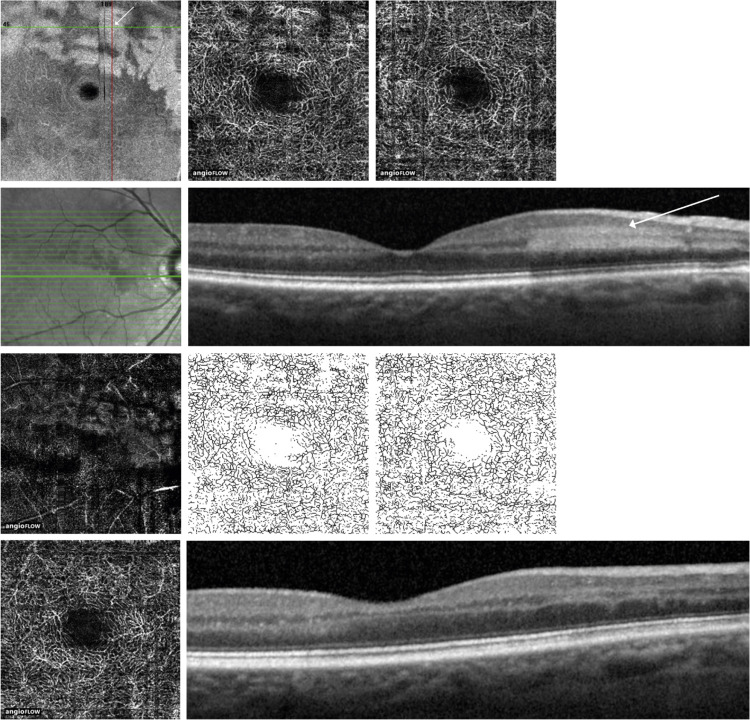
On en face OCT angiography, the superficial capillary plexus of the affected right eye appeared to be within normal limits. The deep capillary plexus ( Figure 1 , First row, second from left) demonstrated patent structure with careful analysis of the en face images, and patent flow and perfusion with careful study of the OCT angiograms, corresponding to the hyperreflective paracentral acute middle maculopathy lesions with adjacent areas of hyporeflectivity, likely due to a contrast effect of the adjacent bright hyperreflective plaques. Vessel density was calculated for the superficial and deep retinal capillary plexus. There was minimal difference in vessel density between each eye in the superficial capillary plexus (12.9 mm −1 in normal left eye vs 12.5 mm −1 in affected right eye, reduction −3.1%) and the deep capillary plexus (17.6 mm −1 in normal left eye vs 17.8 mm −1 in affected right eye, difference +0.8%).
The patient returned after 1 month and noted that the paracentral scotomas were present but improved. The OCT showed persistent hyperreflective lesions in the inner nuclear layer with corresponding thinning. Repeat OCT angiography did not show any reduction of the vascularity of the deep capillary plexus based on quantitative capillary density comparisons between the affected and unaffected areas.
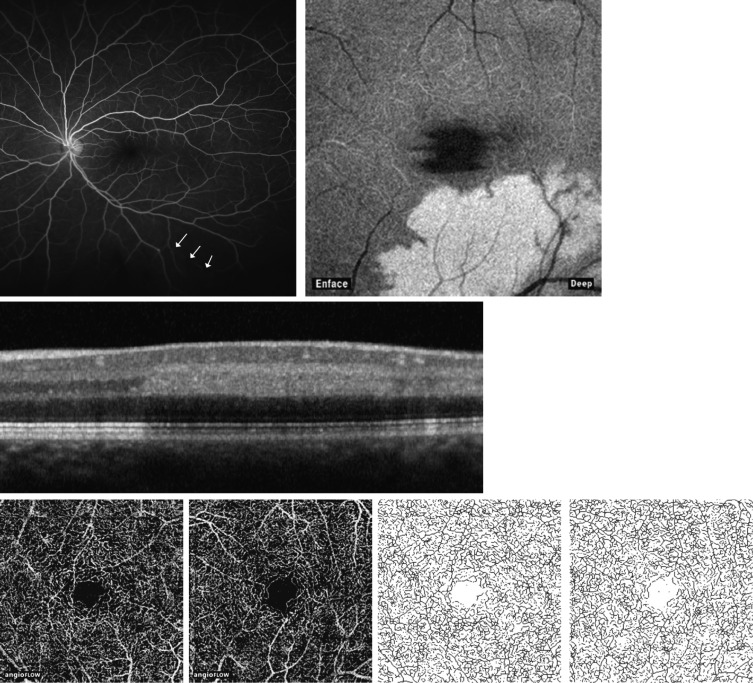
Case 3
A 31-year-old man ( Figure 2 ) presented with acute loss of vision in the superior visual field of his left eye after left-sided head trauma while intoxicated with alcohol. Past medical history was notable for a transposition of the great vessels, surgically repaired when he was an infant. BCVA was 20/20 in the right eye and 20/20-1 in the left eye. Ophthalmoscopic examination of the right eye was unremarkable except for 1 dot-blot hemorrhage inferiorly Ophthalmoscopic examination of the left eye revealed attenuation of the inferotemporal retinal arteriole consistent with a BRAO. Corresponding fluorescein angiography ( Figure 2 ) confirmed a peripheral filling defect of the inferotemporal retinal arteriole. Spectral-domain OCT ( Figure 2 ) revealed a band of hyperreflectivity at the level of the inner nuclear layer consistent with paracentral acute middle maculopathy. OCT angiography (Third row, second from left) revealed patent flow and perfusion in the deep capillary plexus of the left eye, corresponding to the paracentral acute middle maculopathy lesion on spectral-domain OCT. Vessel density was calculated for the superficial capillary plexus and deep capillary plexus and results for each eye were compared. There was minimal difference in the superficial capillary plexus (14.9 mm −1 in the normal right eye vs 14.4 mm −1 in the affected left eye, reduction −3.4%) and in the deep capillary plexus (19.3 mm −1 in the normal right eye vs 18.7 mm −1 in the affected left eye, reduction −3.1%).
Case 6
An 82-year-old man ( Figure 3 ) presented with sudden loss of vision in the left eye. Past medical history was notable for hypertension, hyperlipidemia, and type 2 diabetes. He reported a temporal headache on the left side but denied any other symptoms of giant cell arteritis. BCVA was 20/30 in the right eye and count fingers at 4 feet in the left eye. Ophthalmoscopic examination of the right eye was within normal limits except for a flat benign choroidal nevus. Ophthalmoscopic examination of the left eye revealed attenuated retinal vessels in the absence of embolic material, associated with cotton-wool spots and a cherry-red spot in the macula. Fluorescein angiography revealed delayed retinal vascular flow. Spectral-domain OCT ( Figure 3 , Top left) showed diffuse hyperreflective band-like lesions at the level of the inner nuclear layer in the left eye, consistent with an acute CRAO associated with paracentral acute middle maculopathy. Erythrocyte sedimentation rate was within normal limits (26 mm/hr), but C-reactive protein was elevated to 3.2 mg/L, raising suspicion for a diagnosis of giant cell arteritis. The patient was admitted, given a 5-day course of intravenous solumedrol, and discharged home on a prednisone taper. A temporal artery biopsy on the left side showed no evidence of giant cell arteritis on pathology review.




