Purpose
To evaluate the long-term efficacy of ciliary neurotrophic factor delivered via an intraocular encapsulated cell implant for the treatment of retinitis pigmentosa.
Design
Long-term follow-up of a multicenter, sham-controlled study.
Methods
Thirty-six patients at 3 CNTF4 sites were randomly assigned to receive a high- or low-dose implant in 1 eye and sham surgery in the fellow eye. The primary endpoint (change in visual field sensitivity at 12 months) had been reported previously. Here we measure long-term visual acuity, visual field, and optical coherence tomography (OCT) outcomes in 24 patients either retaining or explanting the device at 24 months relative to sham-treated eyes.
Results
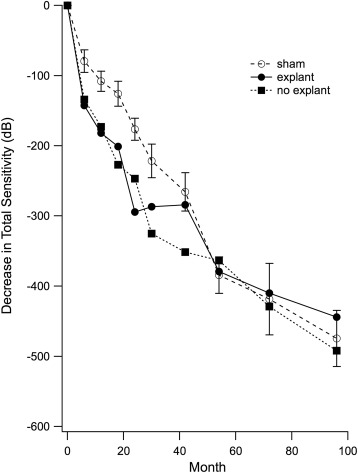
Eyes retaining the implant showed significantly greater visual field loss from baseline than either explanted eyes or sham eyes through 42 months. By 60 months and continuing through 96 months, visual field loss was comparable among sham-treated eyes, eyes retaining the implant, and explanted eyes, as was visual acuity and OCT macular volume.
Conclusions
Over the short term, ciliary neurotrophic factor released continuously from an intravitreal implant led to loss of total visual field sensitivity that was greater than the natural progression in the sham-treated eye. This additional loss of sensitivity related to the active implant was reversible when the implant was removed. Over the long term (60–96 months), there was no evidence of efficacy for visual acuity, visual field sensitivity, or OCT measures of retinal structure.
A major challenge in the treatment of retinitis pigmentosa (RP) is the safe and effective delivery of therapeutic macromolecules to the retina. One potential approach to this challenge is an intraocular encapsulated-cell implant to enable controlled, continuous, and long-term delivery of therapeutic macromolecules, including neurotrophic factors. Ciliary neurotrophic factor is a member of the interleukin-6 (IL-6) family of cytokines and interacts with a tripartite receptor complex composed of ciliary neurotrophic factor receptor α plus 2 signal-transducing transmembrane units, leukemia inhibitory factor receptor β and glycoprotein 130 (gp130). Ciliary neurotrophic factor receptor α is located on Müller glial membranes and on rod and cone photoreceptors. Ciliary neurotrophic factor has been shown to rescue photoreceptors after a single intraocular injection in a variety of animal models of RP and with maintained exposure through gene therapy in mouse models. However, retinal degeneration models treated with ciliary neurotrophic factor may also show suppression of retinal function as measured with the electroretinogram, suggesting that dosage and duration of exposure can be critical.
A therapeutic approach to treatment with ciliary neurotrophic factor in patients with RP was established through sustained-release delivery with intraocular encapsulated cell technology implants (NT-501; Neurotech USA, Cumberland, Rhode Island, USA). After showing safety and efficacy in animal models, the ciliary neurotrophic factor implants passed appropriate milestones in a phase 1 human clinical study of RP. Thus, 2 phase 2 studies (CNTF3 and CNTF4) were designed to evaluate the effect of ciliary neurotrophic factor on retinal structure and function in patients with RP. CNTF3 was motivated by preliminary evidence of visual acuity improvement in the phase 1 trial. The primary endpoint was change in best-corrected acuity at 12 months. As reported previously, no significant changes in visual acuity were observed in ciliary neurotrophic factor or sham-treated eyes, effectively ruling out any short-term benefit to visual acuity from the ciliary neurotrophic factor implants.
CNTF4 sought to determine whether sustained release of ciliary neurotrophic factor could slow the progression of visual field loss in patients with RP and visual acuity of 20/63 or better. Patients (n = 68) were randomized to either high- or low-dose NT-501 implants in 2:1 ratio in 1 eye, and a sham treatment in the fellow eye. The high dose was selected based on the dose-response effect of ciliary neurotrophic factor in the rcd1 dog model of retinal degeneration and was the maximum effective dose. The low dose was 50% of the minimum effective dose in the dog model. The primary outcome at 12 months was the change in total visual field sensitivity, as determined by the sum of the thresholds for the 76 locations with the Humphrey visual field 30-2 test. As reported previously, eyes treated with the high-dose implant showed a greater loss of total visual field sensitivity compared with sham, and this decrease was reversible with implant removal. Ciliary neurotrophic factor also resulted in a dose-dependent increase in retinal thickness. Ancillary studies with adaptive-optics scanning laser ophthalmoscopy showed cone preservation in ciliary neurotrophic factor–treated eyes compared with the sham-treated fellow eyes in a subset of CTNF4 participants.
CNTF3 and CNTF4 showed that long-term intraocular delivery of ciliary neurotrophic factor could be safely achieved through intraocular implants. They also demonstrated that there was no demonstrable benefit to the patients at 12 months. However, ciliary neurotrophic factor showed clear biological activity as indicated by the dose-dependent increase in macular thickness and a decrease in visual field sensitivity in the treated eye. These studies did not address, however, long-term consequences of intraocular ciliary neurotrophic factor. Ciliary neurotrophic factor downregulates phototransduction and reduces electroretinogram (ERG) amplitude, consistent with reduced visual field sensitivity at 12 months. What is not known, however, is whether this downregulation over the short term is associated with preservation of photoreceptors over the long term, as in mouse models of RP. The goal of the present study, therefore, was to determine the functional and structural status of patients many years after the ciliary neurotrophic factor treatment.
Methods
Patients were followed for 24 months in the CNTF4 trial (registered as NCT00447980 on clinicaltrials.gov ). There was an optional registry study in which the patients were followed for an additional 12 months. Three of the CNTF4 study sites continued to follow 24 patients out of the 36 enrolled for durations up to 8 years (mean duration of follow-up = 80 months). These included 8 patients with the low-dose device and 16 patients with the high-dose device. The original trial design approved by the United States Food and Drug Administration (FDA) specified that implants be removed at 24 months. After the initiation of the trial, the FDA recommended that patients retain the implants at the end of the study (avoiding a second surgery). Since all patients had consented to have their implants removed at the end of the study, they were informed of study results to date, given an updated risk/benefit assessment, and offered a choice either to keep the implant in place (n = 14) or to have the implant removed (n = 10). Institutional review boards for each participating site approved the study and subjects signed written informed consent forms prior to participation in the study.
Best-corrected visual acuity (BCVA) was measured by an electronic visual acuity tester (EVA) using the ETDRS protocol. Visual field sensitivity was measured with the 30-2 grid using the Humphrey visual field analyzer. Visual field sensitivity was measured twice per eye on each of 3 baseline visits. Eligibility for enrollment was determined by the results of baseline 1. The average value of the 2 baseline 2 and 2 baseline 3 examinations (4 total), each representing the sum of actual thresholds for all 76 locations, provided the baseline visual field sensitivity. During the CNTF4 trial, 4 visual field sensitivity measurements per eye were taken for each subsequent visit and the average of the 4 visual field sensitivity sums was used to assess the change from baseline. On follow-up visits beyond 30 months, visual fields were typically obtained without replicates. Retinal thickness and morphology were evaluated by optical coherence tomography (OCT). During the CNTF4 trial, the fast macular thickness map protocol, a 7-mm horizontal line scan, and 6-mm vertical line scan were obtained with the Stratus OCT (Carl Zeiss Meditec, Inc, Dublin, California, USA). On follow-up visits beyond 30 months, spectral-domain (SD) OCTs were obtained with the Heidelberg Spectralis HRA+OCT (Heidelberg Engineering, Heidelberg, Germany).
Results
A summary of outcome data on the visit approximately 80 months post implant surgery is shown in the Table . Mean logMAR BCVA in the ciliary neurotrophic factor–treated eye at follow-up was 0.26 (20/32). This was not significantly different ( P = .96) from the mean logMAR BCVA in the sham eye of 0.25 (20/32).
| Implant | Sham | Difference (95% CI, Significance) | |
|---|---|---|---|
| Best-corrected visual acuity (logMAR), mean (SD) | 0.26 (SD: 0.23) | 0.25 (SD: 0.32) | 0.004 (−0.16 to 0.15, P = .96) |
| Change in visual field sensitivity (dB), mean (SD) | −468 (SD: 252) | −475 (SD: 229) | 7.0 (−33 to 70, P = .40) |
| Total macular volume (mm 3 ), mean (SD) | 7.23 (SD: 0.87) | 7.15 (SD: 0.88) | 0.08 (−0.12 to 0.29, P = .41) |
Changes in total visual field sensitivity on the final visit were comparable ( P = .88) for those receiving the low-dose device and those receiving the high-dose device, so doses were combined for subsequent analyses. On the final visit at an average of 80 months post implantation, the total sensitivity loss in explanted eyes (−378 dB) was not significantly different ( P = .29) from the total loss in eyes retaining the device (−492 dB). Combining low- and high-dose eyes, and explant vs no explant, the average difference from baseline on the final visit for treated eyes (−468 dB) was 7.0 dB less (95% confidence interval = −33 to 70, P = .40) than for sham eyes (−475 dB).
Changes from baseline in total visual field sensitivity are shown for each visit in the Figure . All patients (n = 24) were followed to 30 months. For extended follow-up, 5 had their final visit at 66–78 months and 19 were tested at 96 months. Sham eyes (open circles) showed an average decline in total sensitivity of 475 dB over 96 months (59 dB/year). Eyes retaining the implant (solid squares) showed greater loss in total visual field sensitivity through month 42 (351 dB vs 266 dB in sham). By month 54, total loss in eyes retaining the implant was comparable to that in sham eyes and remained so through month 96. In eyes where the ciliary neurotrophic factor device was explanted at 24 months (solid circles), there was a plateau in loss between 24 and 40 months. Beyond 42 months, total visual field loss in explanted eyes was comparable to sham eyes.