Purpose
To evaluate potential accumulation of fluid in the outer choroid in eyes with central serous chorioretinopathy.
Design
Retrospective observational case series.
Methods
Patients in 2 community-based retinal practices were evaluated for hyporeflective areas in the outer choroid consistent with collections of fluid using enhanced depth imaging optical coherence tomography. Eligible patients were examined over the preceding 2 years, had a history of central serous chorioretinopathy, and did not have a history of choroidal neovascularization or photodynamic therapy.
Results
In the New York group there were 131 eyes of 70 patients who had a mean age of 56.3 (± 12.5) years, and 88 (67.2%) had hyporeflective regions consistent with posterior loculation of fluid in the macular region. In the Minnesota data set there were 91 eyes of 48 patients who had a mean age of 47.9 (± 9.9) years and hyporeflective regions consistent with posterior loculation of fluid was present in 59 (64.8%). In the entire group the mean subfoveal choroidal thickness of those without loculated fluid was 344 μm, as compared with 498 μm with loculated fluid ( P < .001). The areas of loculated fluid were hyporeflective, were larger topographically than the large choroidal vessels, had an angular inner border, and did not have a bounding vascular wall.
Conclusions
Posterior loculation of fluid is a common finding in central serous chorioretinopathy, but it has a different pattern and distribution than do collections of fluid in the outer choroid and suprachoroidal space as seen in other forms of choroidal effusion.
Uveal effusion may be caused by hypotony, ocular inflammation, infection, trauma, nanophthalmos, medications, vascular malformations, or circulatory abnormalities. In these conditions excessive leakage from the choroidal vasculature, driven by increased flow, pressure, or decreased vascular competence, leads to the accumulation of choroidal fluid, typically in the equatorial and pre-equatorial regions of the eye. While the accumulation of fluid can occur anywhere in the thickness of the choroid, prominent collections accumulate in the outer choroid, typically in the suprachoroidal space, a region where there is a sparse collection of linking fibers. Central serous chorioretinopathy is a condition presumed to occur secondary to choroidal hyperpermeability as first proposed by Gass. Multifocal areas of hyperpermeability in the posterior pole were found decades later when angiography using indocyanine green became possible. The idea was that the hyperpermeability caused the choroid hydrostatic pressure to increase, leading to detachments of the retinal pigment epithelium (RPE) and hyperbaric defects in the RPE monolayer, seen as “leaks” under the retina. If the pressure increased within the choroid in central serous chorioretinopathy, one would expect the choroid to be thickened. Indeed, thickening of the choroid was found using enhanced depth imaging (EDI) optical coherence tomography (OCT). However, the nature of the thickening of the choroid remains undefined; it is possible that fluid accumulates in the posterior choroid in central serous chorioretinopathy as it does in other forms of uveal effusion. Evaluation of the nature of any possible accumulation may give further clues as to the pathophysiology of the disease and suggest additional modalities of treatment.
Methods
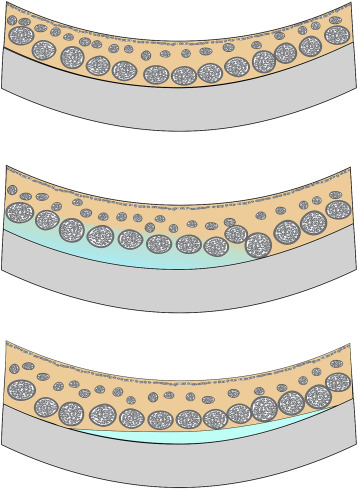
This retrospective study was approved by the Western Institutional Review Board (Puyallup, Washington, USA) and complied with the Health Insurance Portability and Accountability Act of 1996. The patients in this study were evaluated in a community-based retinal practice in New York during the past 2 years by one of the authors (R.F.S.) and from a community-based retinal practice in Minnesota (by author E.H.R.). Patients with a diagnosis of central serous chorioretinopathy were included whether or not they had active disease at the time of study entry. The diagnosis of central serous chorioretinopathy was based on the appearance of subretinal fluid caused by leaks from the level of the retinal pigment epithelium as diagnosed by fluorescein angiography in the absence of accelerated hypertension, inflammation, or infiltration. Patients with choroidal neovascularization or a history of photodynamic therapy or those with ocular media problems limiting fundus imaging were excluded. The patients had macular OCT evaluations using the Heidelberg Spectralis HRA+OCT (Heidelberg Engineering, Heidelberg, Germany) to include EDI OCT scans over a 30 × 5 degree region with 100 images averaged using 7 sections in New York. In Minnesota a number of scan variations were done with the EDI technique using the same OCT instrument. The scan width varied between 15 and 30 degrees. The section through the fovea was used to measure the choroidal thickness, which was the distance between the outer edge of the reflection caused by the retinal pigment epithelium/Bruch membrane complex to the inner border of the sclera. Some patients were also imaged using a prototype swept source OCT instrument (Atlantis OCT; Topcon Medial Systems, Oakland, New Jersey, USA), which has a light source with center wavelength of 1050 nm and a bandwidth of 100 nm; the instrument scans at 100 000 A-scans per second and, depending on the scan protocol, can average images. The choroid was evaluated for the presence of hyporeflective extravascular spaces greater than 250 μm in smallest diameter. These were presumed to represent posterior loculations of fluid. These were subclassified as interdigitated, in which the dark extravascular spaces extended between and behind the larger choroidal vessels in the posterior choroid ( Figure 1 ). A second subtype was termed dissociated and represented isolated collections of fluid posterior to the larger choroidal vessels. Data were analyzed with descriptive statistics and to evaluate relationships between the vitreous anatomic findings and age-generalized estimating equations were used. A P value less than or equal to .05 was considered significant. The IBM SPSS version 21 statistical package was used (IBM Corporation, Armonk, New York, USA).
Results
The general appearances of the hyporeflective areas, thought to be consistent with loculated fluid in the choroid, are shown in Figures 2–4 . In the New York group there were 70 patients, with a mean age of 56.3 (standard deviation ± 12.5, interquartile range [IQR] 47–66.3) years; there were 54 male patients (77.1%) ( Table ). Of the 70 patients, 17 (24.3%) had a history of using corticosteroids in any form, with 7 (9.9%) having used oral administration. The 131 eyes had a mean visual acuity of 0.2 logMAR (20/32 Snellen equivalent); a pigment epithelial detachment was present in 25 eyes (19.1%) and subretinal fluid was present in 44 (33.6%). The mean subfoveal choroidal thickness was 451 (± 146, IQR: 336–527) μm. Hyporeflective zones, presumed to represent posterior loculation of subretinal fluid, were present in 88 eyes (67.2%). The interdigitated pattern was seen in 87 and the dissociated form in 4, all of which, except for 1, also had areas of interdigitated fluid. The thickness of the choroid in eyes with no loculation was 349 μm as compared to 501 μm in those with loculation, a difference that was highly significant ( P < .001). Posterior loculation of fluid was not more common in patients with active disease as manifested by retinal pigment epithelial detachment ( P = .3), subretinal fluid ( P = .08), and use of corticosteroids ( P = .16), but was more common in male patients ( P = .006, all tests were χ 2 analyses). Using generalized estimating equation analysis, the only significant predictor of the presence of posterior loculation of fluid in the choroid was the subfoveal choroidal thickness; once subfoveal choroidal thickness was entered into the model, sex was no longer significant.
| Entity | Value | |
|---|---|---|
| New York group | ||
| Patients | 70 | |
| Age in years | 56.3 (± 12.5) | |
| Number male | 54 (77.1%) | |
| Eyes evaluated | 131 | |
| Mean visual acuity | 0.2 logMAR (20/32 Snellen equivalent) | |
| Pigment epithelial detachment | 25 (19.1%) | |
| Subretinal fluid | 44 (33.6%) | |
| Hyporeflective zone of presumed posterior loculation of fluid | 88 (67.2%) | |
| Subfoveal choroidal thickness in eyes with no presumed loculation of fluid | 349 μm | |
| Subfoveal choroidal thickness in eyes with presumed loculation of fluid | 501 μm | P < .001 |
| Minnesota group | ||
| Patients | 48 | |
| Age in years | 47.9 (± 9.9) | |
| Number of males | 31 (64.6%) | |
| Eyes evaluated | 91 | |
| Mean visual acuity | 0.153 logMAR (20/28 Snellen equivalent) | |
| Pigment epithelial detachment | 18 (19.8%) | |
| Subretinal fluid | 54 (59.3%) | |
| Hyporeflective zone of presumed posterior loculation of fluid | 59 (64.8%) | |
| Subfoveal choroidal thickness in eyes with no presumed loculation of fluid | 337 μm | |
| Subfoveal choroidal thickness in eyes with presumed loculation of fluid | 405 μm | P < .001 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


