Purpose
To describe a possible causal association between the position of perforating scleral vessels and the position of lacquer cracks in eyes with pathologic myopia.
Design
Retrospective case series.
Methods
Medical records and multimodal imaging results, including confocal scanning laser ophthalmoscopy and spectral-domain optical coherence tomography, were reviewed from patients with lacquer cracks secondary to pathologic myopia who presented between 2010 and 2014 to 2 institutions. Main outcome measures were the prevalence of perforating scleral vessels at the site of the lacquer crack, the position of the lacquer crack within the macula, and the relationships between perforating scleral vessels and retinal-choroidal structures.
Results
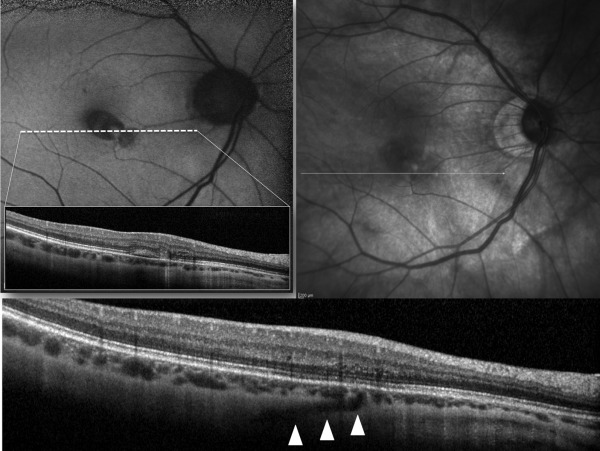
A total of 35 eyes of 30 patients with lacquer cracks were included. The average number of lacquer cracks was 1.2 ± 0.5/eye and in 37 out of 45 lacquer cracks (82%) retrobulbar vessels were found to perforate the sclera at the site of the lacquer crack. Lacquer cracks were more prevalent in the central macula (51%) than in the nasal (19%), temporal (14%), inferior (11%), and superior macula (5%) ( P = .001). Transverse en face images through the area of lacquer cracks were available for 8 cases and clearly depicted the perforating vessel’s course through the sclera and its termination in the choroid, directly beneath the lacquer cracks.
Conclusions
Perforating scleral vessels are often present beneath the site at which lacquer cracks form in pathologic myopia. We hypothesize that scleral expansion at the location of these perforating vessels may play a role in the formation of lacquer cracks.
High myopia is a major cause of visual impairment and has rapidly increased in prevalence over the past 50 years. Eyes with high axial length are described as having pathologic myopia when they manifest degenerative changes of the sclera, choroid, and retina. In pathologic myopia, progressive posterior segment elongation and deformation may lead to the development of macular lesions, including posterior staphyloma, lacquer cracks, myopic choroidal neovascularization (CNV), myopic macular retinoschisis, and myopic chorioretinal atrophy.
Lacquer cracks are mechanical breaks in the Bruch membrane–retinal pigment epithelium (RPE)–choriocapillaris complex owing to excessive axial elongation and represent a hallmark feature of pathologic myopia. Lacquer cracks are a risk factor for CNV, a severe complication of advanced pathologic myopia. Their presence implies a guarded prognosis for the retention of central vision. Myopic stretch lines and lacquer cracks may appear similar; however, it is important to differentiate these 2 entities, as their pathogenesis and visual prognosis can be significantly different.
Publications describing the histopathologic features of lacquer cracks are scarce. Moreover, based on a PubMed search, it appears that no prior study has investigated in vivo (by means of multimodal imaging) the changes in the human sclera in areas of lacquer crack occurrence. Diminution of choroidal and scleral thicknesses are frequently associated with pathologic myopia, and for this reason it is often possible to evaluate the complete depth of these structures using spectral-domain optical coherence tomography (SD OCT) technology. Moreover, it is easier to visualize deeper retrobulbar structures such as vessels perforating the sclera (eg, posterior ciliary vessels) in myopic eyes than in emmetropic eyes using SD OCT.
In this study we use multimodal imaging to demonstrate a possible causal association between the position of perforating scleral vessels and lacquer cracks in eyes with pathologic myopia.
Methods
This is a retrospective observational case series of patients from 2 institutions (Department of Ophthalmology of the University Scientific Institute San Raffaele in Milan and Vitreous Retina Macula Consultants of New York) who presented between January 2010 and December 2014 with lacquer cracks secondary to pathologic myopia in at least 1 eye. This retrospective study followed the tenets of the Declaration of Helsinki for research involving human subjects. Informed consent was obtained from all subjects. Institutional Review Board (IRB) approval was obtained for this retrospective study and the retrospective review of patient information, from both sites (Scientific Institute San Raffaele IRB, and Western IRB). The study was carried out in compliance with local and national IRB guidelines. Health Insurance Portability and Accountability Act (HIPAA) compliance was also maintained.
Inclusion criteria were: (1) diagnosis of pathologic myopia defined by a refractive error (spherical equivalent) >-8.00 diopters (D) or an axial length >26.5 mm, accompanied by characteristic degenerative changes of the sclera, choroid, and retina; (2) presence of lacquer cracks; and (3) availability of dense raster SD OCT B-scans (less than or equal to 30 μm intervals between subsequent B scan) encompassing the area of lacquer cracks. Lacquer cracks were defined as lesions that exhibited the following clinical characteristics: (1) linear or stellate morphology (not punctate) (this permitted exclusion of lesions secondary to multifocal choroiditis and punctate inner choroidopathy); (2) absence of hyperpigmented margins; (3) not localized to sites of choroidal excavation. Multimodal imaging defining criteria for lacquer cracks included lesions that met the above clinical criteria and also were hyperreflective on infrared reflectance (IR), hyperfluorescent on fluorescein angiography (FA), or hypofluorescent on fundus autofluorescence (FAF).
Exclusion criteria were as follows: (1) presence of CNV in the study eye; (2) any previous treatment in the study eye including treatment with intravitreal injection of anti–vascular endothelial growth factor agents, photodynamic therapy, or thermal laser photocoagulation; (3) history of ocular inflammation in the study eye; (4) significant media opacities; (5) any other retinal disease (such as retinal vein occlusion, diabetic retinopathy, angioid streaks, or macular dystrophy) in the study eye. Myopic stretch lines were excluded on the basis of multimodal imaging criteria because they typically appear hyperautofluorescent on FAF and hypofluorescent on FA (in contrast to hypoautofluorescent on FAF and hyperfluorescent on FA for lacquer cracks).
A review of paper and electronic medical records was performed during which demographic data, best-corrected visual acuity (BCVA), and clinical findings were recorded. Multimodal imaging data were reviewed and included combinations of color fundus photography (Topcon Medical Systems, Oakland, New Jersey, USA) and confocal scanning laser ophthalmoscopy (cSLO; Heidelberg Engineering, Heidelberg, Germany). MultiColor (Heidelberg Engineering), IR, FAF, FA, indocyanine green angiography (ICGA), and SD OCT (Spectralis SD-OCT [Heidelberg Engineering], ± enhanced depth imaging [EDI] and transverse en face images through the area of lacquer cracks) were also reviewed.
The cSLO and SD OCT images were viewed and analyzed with the contained Heidelberg Eye Explorer software (Spectralis Acquisition and Viewing Modules; version 5.6.1.0, Heidelberg Engineering) by 2 experienced retinal physicians (G.Q., K.B.F.). Presence of perforating vessels at the site of lacquer crack occurrence (verified by the 2 experienced retinal physicians by analysis of adjacent SD OCT scans) and their localization and relationships to retinal-choroidal structures were recorded. Choroidal and scleral thicknesses underlying lacquer cracks were also measured using EDI images. Choroidal thickness was defined as the distance between the outer border of the RPE-Bruch membrane complex and the choroid-sclera junction. Scleral thickness was defined as the distance between the choroid-sclera junction and the outer border of the sclera.
Statistical calculations were performed with GraphPad Prism version 6.0 (GraphPad Software, San Diego, California, USA). Analyses included qualitative descriptions of clinical findings, descriptive statistics for demographic data (reported as mean ± standard deviation), and a calculation of the prevalence of perforating vessels at the site of lacquer crack occurrence (reported with 95% confidence intervals). The differences in quantitative parameters between lacquer cracks, with and without perforating vessels, were analyzed using the nonparametric Mann-Whitney U test. The frequency of lacquer cracks at each position in the macula was expressed as a percent proportion of the total number of lacquer cracks. Comparisons were performed using χ 2 test. All variables were tested for normal distributions, according to the Kolmogorov-Smirnov test. The chosen level of statistical significance was P < .05. All tests were 2-sided and a P value less than .05 was considered significant.
Results
Thirty-five eyes from 30 patients (12 patients from the Department of Ophthalmology of the University Scientific Institute San Raffaele in Milan and 18 patients form the Vitreous Retina Macula Consultants of New York) met the inclusion criteria. The study population consisted of 13 male and 17 female subjects with a mean age of 59.9 ± 14.8 years (range, 34–78 years). Twenty-eight patients were white and 2 were Asian. Twenty-five fellow eyes were excluded from the analysis either because of the absence of lacquer cracks (14 eyes) or because of CNV (11 eyes). Mean BCVA in the study eye was 20/59 (Snellen). Mean axial length in the study eye was 30.9 ± 1.3 mm. Mean refractive error in the study eye was –11.5 ± 1.8 D.
A total of 45 lacquer cracks were imaged using dense raster SD OCT B-scans (1.2 ± 0.5 lacquer cracks/eye). The frequency of the position of lacquer cracks, as represented by the 1 mm central diameter macula ETDRS grid, was (1) central macula (51%), (2) nasal macula (19%), (3) temporal macula (14%), (4) inferior macula (11%), and (5) superior macula (5%). The frequency of lacquer crack occurrence at each position was significantly different ( P = .0001).
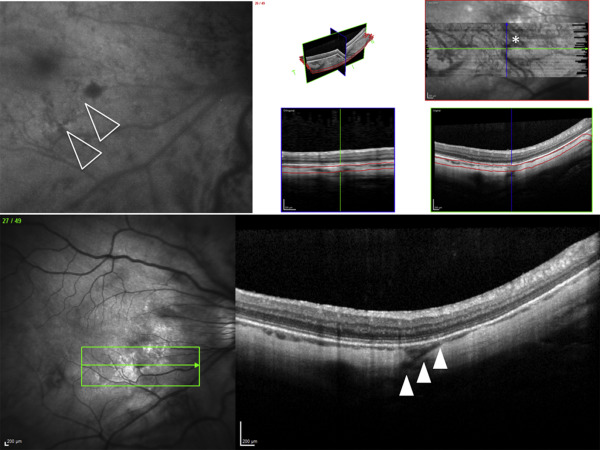
Thirty-seven out of 45 lacquer cracks imaged by dense raster SD OCT (± EDI) B-scans showed structures of linear hyporeflectivity that were consistent with retrobulbar vessels perforating the sclera beneath the site of lacquer cracks (as visualized by multimodal cSLO imaging) ( Figures 1–3 and Supplemental Figures 1 and 2 , available at AJO.com ). The calculated prevalence of these structures was 82% (71%–93%, 95% CI). Perforating vessels joined the choroid either beneath the terminal part of the lacquer cracks (22 out of 37 lacquer cracks, 60%) or beneath the central part of the lacquer cracks (15 out of 37 lacquer cracks, 40%). In 8 lacquer cracks, transverse en face images through the area of lacquer cracks were available. En face images clearly demonstrated the course of perforating vessels through the thickness of the sclera and showed their emergence in the choroid just beneath the lacquer cracks ( Figures 1 and 2 ). In 8 out of 45 lacquer cracks it was not possible to detect perforating vessels beneath the lacquer cracks (despite careful review of dense raster B-scans). Of note, 3 eyes demonstrated a single lacquer crack without associated perforating vessels, while lacquer cracks with and without perforating vessels were found in the same eye in 5 cases. A subretinal hemorrhage was seen at the site of the lacquer crack in 14 out of 45 eyes. Lacquer cracks were not detected at the site of subretinal hemorrhage in 2 cases.
Comparisons of choroidal and scleral thicknesses at sites of lacquer cracks, between lesions with perforating vessels and those without, were not significant (choroidal thickness: 50.3 ± 25.7 μm and 51.5 ± 17.5 μm, respectively, P = .5; scleral thickness: 266.1 ± 69.1 μm and 263.8 ± 97.7 μm, respectively, P = .9). There was no difference in scleral thickness under lacquer cracks compared to adjacent areas without lacquer cracks (data not shown).
Illustrative Case
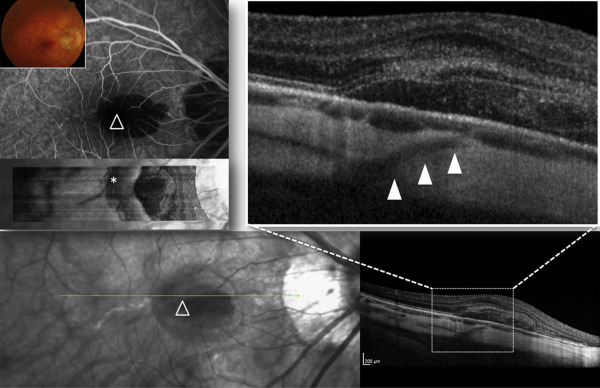
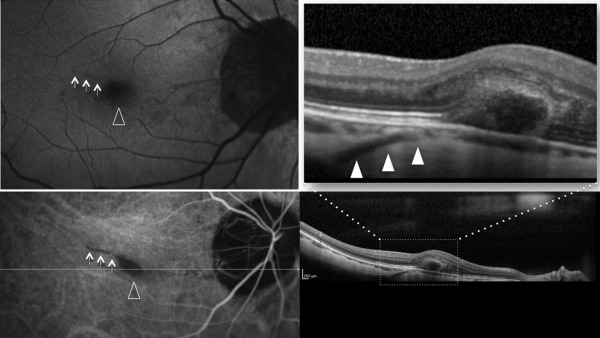
A 44-year-old woman with pathologic myopia presented with sudden visual loss in the right eye (OD). BCVA was 20/125 OD and 20/25 in the left eye (OS). Ophthalmoscopy showed a macular hemorrhage OD. SD OCT B-scans demonstrated a linear hyporeflective structure that was consistent with a retrobulbar vessel traversing the sclera. The perforating vessel was seen to terminate in the choroid beneath the inferonasal border of the subretinal hemorrhage ( Figure 4 ). The perforating vessel was also clearly visible in the tracked (Spectralis real-time eye tracking) SD OCT line scan performed 3 months before the development of the macular hemorrhage ( Figure 5 ). At that time, no IR or FAF abnormalities were evident. Interestingly, 3 months later, after resolution of the subretinal hemorrhage, the tracked (Spectralis real-time eye tracking) SD OCT line scan revealed the perforating vessel joining the choroid just beneath the inferonasal terminal part of the lacquer crack (visualized as a hypofluorescent line on FAF) ( Figure 6 ).