Purpose
To assess the results of iris suture fixation of subluxated intraocular lenses.
Design
Retrospective study.
Methods
This was a nonrandomized chart review of eyes with subluxated intraocular lenses that underwent iris suture fixation at an academic institutional care center. Seventy-two eyes of 67 consecutive patients were included. The following cases were excluded: posterior dislocations necessitating pars plana vitrectomy; secondary implantations for aphakia; and iris suture fixation at primary cataract extraction. Main outcome measures included visual acuity improvement, surgically induced astigmatism, and postsurgical complications.
Results
The mean follow-up duration was 16.64 ± 24.37 months (median = 4.03 months). All patients had preoperative monocular diplopia or unstable vision attributable to the subluxated intraocular lenses, and 40.3% of them required aphakic correction. There was an overall improvement in best-corrected visual acuity from a mean preoperative logMAR 0.35 ± 0.32 (Snellen equivalent∼20/45) to logMAR 0.21 ± 0.25 (20/32, P = .001). There was no significant change in astigmatism secondary to the surgery. The mean difference in preoperative keratometry readings was 1.6 ± 1.07 diopter (D), whereas the mean postoperative manifest refraction astigmatic error (vertexed to the corneal surface) was 1.29 ± 0.92 D ( P < .02). Re-subluxations occurred in 7 eyes during follow-up; the majority of these eyes underwent repeat fixation. Most (93.55%) intraocular lenses were stable and centered at the final follow-up. Glaucoma developed in 2 eyes postoperatively.
Conclusions
Iris suture fixation of subluxated intraocular lenses was efficacious for the eyes included in this study, and it led to long-term stability of the intraocular lenses in 93.55% of cases.
The optimal treatment of intraocular lens (IOL) subluxation remains contested, and clinical trials comparing the various methods employed are limited. The possible surgical approaches to treat IOL subluxation include IOL exchange to an anterior chamber IOL or an iris-claw IOL; scleral fixation of a posterior chamber (PC) IOL; or iris fixation (including iris suture fixation). Typically, a surgeon’s technique of choice for the treatment of subluxated IOLs depends largely on that surgeon’s experience with a certain method.
Previous studies have shown that suturing a subluxated IOL to the iris is safe and efficacious: iris suture fixation of subluxated IOLs demonstrated favorable results in pseudophakic and aphakic eyes and also in eyes that have undergone penetrating keratoplasty (PK). The largest body of evidence regarding iris suture fixation of IOLs comes from a series of post-PK eyes that was reported by our group. This type of subluxation comprises a special subset of IOL subluxations. More commonly encountered in a clinical setting are subluxations occurring in eyes that only underwent cataract surgery with a PCIOL implantation, and which do not have a history of a corneal graft procedure. The largest reported series on such a clinical setting included 44 eyes. It showed that visual acuity had improved with no surgically induced astigmatism, and a favorable safety profile.
The current study aims to assess the results of iris suture fixation of subluxated IOLs in pseudophakic eyes at a large tertiary referral center.
Methods
This retrospective chart review was approved by the institutional review board (IRB) at Johns Hopkins University and is in accordance with HIPAA regulations. Cases included in this study were consecutive instances of subluxated IOLs that were treated surgically between October 2004 and August 2013 by a single surgeon (W.J.S.) at an academic tertiary referral center. Eyes with vision limiting comorbidities such as amblyopia, corneal decompensation, and posterior pole pathologies were included. All cases included in the present study were new cases that were not reported in previous studies by our group. Exclusion criteria were: cases with combined corneal surgery and iris suture fixation, posterior dislocations necessitating pars plana vitrectomy; secondary implantations for aphakia; and iris suture fixation at primary cataract extraction. Cases with insufficient data were also excluded. A total of 72 eyes of 67 patients were included in this study.
The medical records of the study’s participants were reviewed and the following data were collected: demographic information; details on complications that occurred during cataract surgery; visual acuity and manifest refraction throughout the follow-up period; and preoperative keratometry and axial length measurements (using IOLMaster; Carl Zeiss Meditec, Dublin, California, USA). Postoperative complications such as hemorrhagic complications, cystoid macular edema (CME), new-onset choroidal neovascularization, intraocular pressure elevations or glaucoma, corneal decompensation, retinal detachment, uveitis, and re-subluxations were also recorded.
Snellen visual acuity was transformed to logMAR equivalents to facilitate statistical analysis. Visual acuity of hand motion was set at 2.3 logMAR, whereas counting fingers was set at 1.85 logMAR.
The postoperative manifest refraction astigmatic error reflects the corneal astigmatism and the IOL-induced astigmatism. In order to assess the surgical induction of astigmatism, manifest refraction at the final follow-up visit was converted to a cross-cylinder notation and then vertexed to the corneal surface assuming a vertex distance of 13.75 mm. The vertexed astigmatic error was compared to the difference in preoperative keratometry values (ie, corneal astigmatism) obtained by IOLMaster.
Surgery
Eyes with decreased vision owing to IOL subluxation, severe instability of the IOL, or subjective patient complaints (debilitating glare or monocular diplopia) were eligible for fixation surgery. Eyes with minor subluxations and stable vision over time were not operated on. All surgeries were performed under retrobulbar anesthesia with marcaine (0.75%; Hospira, Lake Forest, Illinois, USA), lidocaine (4%; Hospira) mixed in equal volumes, and hyaluronidase (Halozyme Therapeutics, San Diego, California, USA). Pharmacologic dilation was avoided as the pupil was dilated using retrobulbar injection. This facilitated later pupillary capture of the IOL optic.
The surgical procedure itself was similar to that described before by our group ( Figures 1 and 2 ; Supplementary Material available at AJO.com). Briefly, each surgical procedure began by creating a limbal paracentesis at 3 o’clock and a 5-mm clear corneal incision at 9 o’clock. Miochol (Bausch and Lomb, Bridgewater, NJ, USA) was injected intracamerally, followed by preservative-free lidocaine (0.5%). Healon GV (Abbott Medical Optics, Santa Ana, CA, USA) was injected intracamerally. Then, a sclerotomy was made at the 4 o’clock position and 3 mm posterior to the limbus using a microvitreoretinal (MVR) blade. A Barraquer sweep was then introduced through the sclerotomy to engage the subluxated IOL or the IOL-bag complex, to achieve iris capture of the IOL optic. Single-piece IOLs, plate haptic IOLs, IOLs with square-edged haptics, IOLs encased in a Soemmering ring, and IOLs with deformed haptics were folded or cut by intraocular scissors and then removed through the main corneal incision (n = 61). Eyes with explanted IOLs were then implanted with a 3-piece MA50BM lens (Alcon, Fort Worth, Texas, USA), as described elsewhere. Once an iris-capture of the IOL optic was achieved, the Barraquer sweep was introduced again to provide posterior support for the optic. A 10-0 prolene suture on a CTC-6 needle (Ethicon, New Jersey, USA) was then passed through the cornea and the mid-peripheral iris proximal to the haptic, and then again through the iris distal to the haptic and out through the cornea again. The ends of the suture were removed through another limbal paracentesis using a Lester hook and were then sutured in at least 4 throws (modified McCannel suture). This was repeated once more in the second haptic to achieve a 2-point fixation. After verifying the adequacy of the sutures, the optic was gently pushed posteriorly through the pupil. The viscoelastic device was removed from the anterior chamber and exchanged with balanced saline solution. Pars plana core vitrectomy was performed through the sclerotomy in select cases where vitreous prolapsed to the anterior chamber. The main corneal and scleral incisions were sutured using 10-0 Vicryl suture (Ethicon, Somerville, NJ, USA). The incisions were tested for leakage with Weck-Cells (Beaver Visitec, Waltham, Massachusetts, USA). At the end of the surgical procedure, subconjunctival dexamethasone and appropriate antibiotics were administered.
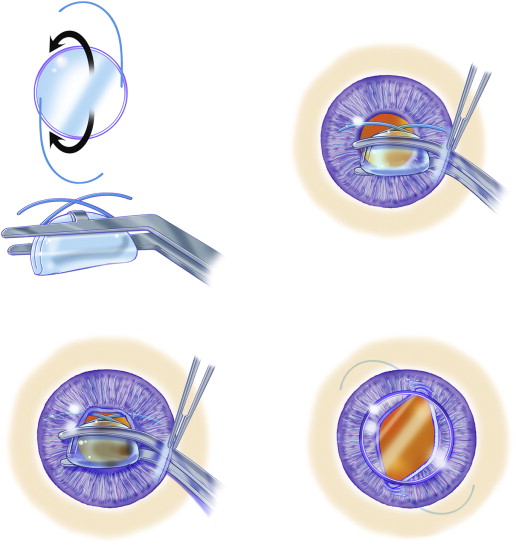
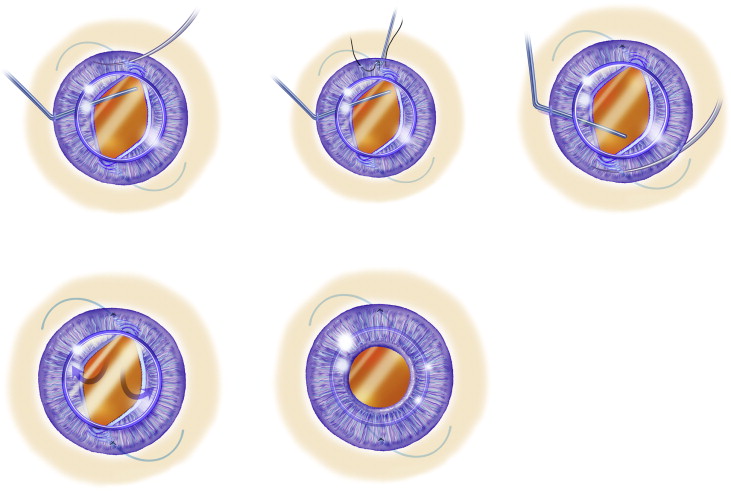
Statistical Analysis
Paired t tests were used to assess the mean change in best-corrected visual acuity (BCVA) within subgroups; in manifest refraction; and surgical induction of astigmatism. The difference in mean BCVA between eyes with or without a recurrent subluxation was assessed using an unpaired t test without assuming equal variances. When considering only those eyes that received more than 30 days of follow-up, a Wilcoxon rank-sum test was used. Fisher exact tests were used for all odds ratio tests. The Kaplan-Meier estimator was used to estimate time until re-subluxation, and Cox proportional-hazard models were used to estimate the unadjusted associations between predictors and time until re-subluxation (22 predictors were assessed). A P value of less than .05 was considered statistically significant, and all tests were 2-sided and based on complete-case analysis. All analyses were performed using R statistical software v3.0.2 (with packages rms v4.0-0 and survival v2.37-4).
Results
The mean age at fixation surgery was 76 ± 12.43 years. The mean time elapsed since cataract surgery was 9.42 ± 6.62 years. In eyes with pseudoexfoliation (PXF) syndrome, the mean time elapsed was 10.7 ± 6.28 ( P = .16 for the comparison of non-PXF to PXF eyes). Mean follow-up duration was 16.64 ± 24.37 months (median = 4.03 months). Sixty-two eyes had a follow-up duration of greater than 30 days.
Twelve eyes had a documented complication during the original cataract surgery. Another 7 eyes had undergone neodymium–yttrium-aluminum-garnet capsulotomy sometime between cataract extraction and fixation surgery, while 12 other eyes had a Soemmering ring. A capsular tension ring was not implanted in any of the eyes included in this study. PXF was diagnosed in 33 eyes. A history of ocular trauma was present in 12 eyes. Phimosis of the anterior capsulotomy was present in 4 eyes. Eighteen eyes were highly myopic (axial length >26 mm), and the overall preoperative mean axial length was 24.85 ± 1.66 mm. Seven eyes had a history of a previous failed fixation surgery.
All included eyes had monocular diplopia, or unstable and fluctuating vision owing to the movement of the subluxated IOLs. Improvement of BCVA was evaluated only in eyes with a follow-up duration of 30 days or more (n = 62). The mean preoperative BCVA was 0.35 ± 0.32 logMAR (approximately 20/45 Snellen equivalent), but 25 of the eyes (40.3%) required aphakic correction to reach this visual acuity. The mean postoperative BCVA was 0.21 ± 0.25 (approximately 20/32 Snellen equivalent, P = .001).
Preoperative and postoperative manifest refraction was available for 59 patients. The mean preoperative spherical equivalent (SE) was 0.91 ± 5.08 diopters (D). The mean postoperative SE was −0.88 ± 1.91 D ( P < .01).
Information on preoperative keratometry and postoperative manifest refraction was available for 61 eyes. Sixty of the 61 eyes had follow-up data beyond 30 days. For these 60 eyes, the preoperative mean difference in keratometry readings was 1.6 ± 1.07 D. The mean postoperative manifest refraction astigmatic error (vertexed to the corneal surface) was 1.29 ± 0.92 D ( P < .02, Figure 3 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


