Purpose
To compare the effectiveness of intravitreal injection of aflibercept and ranibizumab for patients with polypoidal choroidal vasculopathy (PCV).
Design
Retrospective, interventional case series.
Methods
Ninety-eight treatment-naïve PCV eyes were included. The eyes had received intravitreal aflibercept or ranibizumab injections. All patients were treated using an initial series of 3 monthly loading injections, followed by further injections as required. The visual and anatomic outcomes of treatment were evaluated after 12 months.
Results
The mean best-corrected visual acuity (BCVA) in the aflibercept-treated group (38 eyes), expressed as the logarithm of the minimal angle of resolution (logMAR), had decreased from 0.63 ± 0.49 to 0.44 ± 0.37 after 12 months of treatment ( P = .012). Similarly, in the ranibizumab-treated group (60 eyes), the BCVA had decreased from 0.66 ± 0.43 to 0.49 ± 0.36 ( P = .018). The central foveal thickness had decreased in the aflibercept-treated group from 396 ± 167 μm to 212 ± 144 μm, and in the ranibizumab-treated group from 402 ± 198 μm to 240 ± 183 μm ( P < .001 in both cases). However, there was no significant difference between the groups with regard to either BCVA improvement or decrease in central foveal thickness. Polyp regression was significantly more frequent in the aflibercept-treated group, occurring in 39.5% of eyes, than in the ranibizumab-treated group (21.6% of eyes; P = .007).
Conclusions
In PCV patients, the visual acuity improvement achieved after 12 months of intravitreal aflibercept did not differ significantly from that achieved using intravitreal ranibizumab. However, aflibercept treatment more often led to polyp regression than did treatment using ranibizumab.
Polypoidal choroidal vasculopathy (PCV) is a clinical condition that is generally classified as a subtype of neovascular age-related macular degeneration (nAMD). Hallmarks of the disease include a characteristic branching vascular network and polypoidal choroidal vascular lesions. The prevalence of PCV in newly diagnosed nAMD has been reported as approximately 23%-55% in Asian patients, which is higher than in white patients.
Currently, there are several effective treatment methods for PCV. Photodynamic therapy (PDT) with verteporfin or intravitreal injection of anti–vascular endothelial growth factor (VEGF) agents have been used to treat the condition, and encouraging results have been reported in both cases.
Recently, aflibercept (Eylea; Bayer HealthCare, Berlin, Germany) has been introduced as a new anti-VEGF agent to treat nAMD. The drug is a soluble, decoy receptor-fusion protein; it consists of the binding domains of VEGF receptors 1 and 2 fused to the Fc portion of human immunoglobulin G1. This structure allows the drug to bind all isoforms of VEGF-A, VEGF-B, and placental growth factor (PlGF). Intravitreal aflibercept injections dosed monthly or every 2 months after 3 initial monthly doses demonstrated similar therapeutic effect compared with monthly intravitreal ranibizumab injections.
To date, there have been several investigations into the therapeutic effect of aflibercept in PCV; the reports have shown promising results and realistic prospects for PCV treatment using aflibercept monotherapy. However, there have been no reports specifically focusing on the differences between the 2 anti-VEGF agents (ranibizumab and aflibercept) in the treatment of PCV. Therefore, the current study was conducted in order to determine the differences between aflibercept and ranibizumab in terms of therapeutic efficacy in the treatment of PCV.
Methods
We performed a computerized search for patients who had been diagnosed with PCV and treated using anti-VEGF (ranibizumab or aflibercept) injections between May 2013 and September 2014. We then reviewed the patients’ medical records. All patients had been examined and treated at the Retina Center of Kim’s Eye Hospital, Konyang University College of Medicine. This study was approved by the Institutional Review Board of Kim’s Eye Hospital, Konyang University College of Medicine. The study conformed to the tenets of the Declaration of Helsinki (IRB No. A-2015-022).
Subjects
The following inclusion criteria were used: (1) age >50 years; (2) confirmation of PCV using both fluorescein angiography (FA) and indocyanine angiography (ICGA), performed using a confocal laser scanning system (HRA-2; Heidelberg Engineering, Dossenheim, Germany) at the first visit; (3) no previous treatment prior to diagnosis; (4) treatment using only 1 kind of anti-VEGF agent (aflibercept or ranibizumab); and (5) minimum follow-up period of 12 months. We only included patients whose ICGA revealed the characteristic polypoidal structures at the border of the branching choroidal vascular network. In cases where subretinal hemorrhage interfered with identification of the underlying pathologic features, diagnosis was based on the results of the follow-up examinations, including those from ICGA.
The exclusion criteria were the following: (1) concomitant PDT treatment; (2) use of both aflibercept and ranibizumab (in cases of switching from ranibizumab to aflibercept, or vice versa); (3) concurrent ocular diseases such as diabetic retinopathy, high myopia (spherical equivalent of >6 diopters), vein or artery occlusion, or epiretinal membrane; and (4) the presence of any other ocular disease that may have affected visual acuity. No limits on visual acuity were set as either inclusion or exclusion criteria.
Patient Assessment and Outcome Measures
The primary outcome was mean change in best-corrected visual acuity (BCVA) from baseline at 3, 6, 9, and 12 months. For statistical analysis, the Snellen BCVA was converted to a logarithm of the minimal angle of resolution (logMAR). The secondary outcome was mean change in central foveal thickness from baseline at 3, 6, 9, and 12 months; this was measured using spectral-domain optical coherence tomography (SD OCT; Spectralis; Heidelberg Engineering). Central foveal thickness was defined as the vertical distance from the hyperreflective line of the Bruch membrane to the inner limiting membrane on fovea-centered SD OCT images (consisting of either 19 or 31 horizontal lines [6 × 6-mm area]). Furthermore, the percentage of patients who had gained or lost >3 lines of vision, measured from baseline, was recorded, as was the rate of polyp regression.
ICGA was used to measure various baseline characteristics, as well as to identify polyp regression. The areas of individual polyps, excluding the hypofluorescent halo, were measured manually using the software that had been provided with the HRA-2 hardware. These areas were then summed, and thus the total polyp area was derived. The area of the abnormal branching vascular network was also measured on the basis of the ICGA images. The greatest linear dimension (GLD) comprised the entire PCV vascular lesion, including the polypoidal lesion, the vessels of the branching vascular network, and any type 2 choroidal neovascularization (CNV). Pigment epithelial detachments (PED) without underlying vascular components were not included in the GLD measurement. The incidence of choroidal vascular hyperpermeability, defined as multifocal hyperfluorescence seen in the middle and late ICGA phase, was also evaluated.
All measurements were obtained by 2 retinal specialists (H.J.C. and H.S.K.), who worked independently using the Heidelberg Eye Explorer software (v. 5.6.4.0; Heidelberg Engineering) and who had been masked to patient information.
Intravitreal Anti-VEGF Treatment
All patients were given a loading dose of 3 intravitreal anti-VEGF (ranibizumab [0.5 mg/0.05 mL] or aflibercept [2 mg/0.05 mL]) injections with a 1-month interval between treatments. After the loading injections had been administered, additional injections were performed on an “as-needed” basis if any of the following conditions were observed: (1) visual deterioration of >2 lines (>0.2 logMAR); (2) OCT evidence of persistent fluid or hemorrhage involving the macula for at least 1 month following the previous injection; (3) evidence of an active PCV lesion found on FA, ICGA, or OCT. All patients were followed up for 12 months on a monthly basis after initial treatment. Follow-up examinations, including BCVA measurement, fundus photography, and SD OCT, were performed 3, 6, 9, and 12 months after the initial treatment visit. Additional FA, ICGA, and SD OCT examinations were performed whenever PCV recurrence was suspected. In previous studies, aflibercept injections have been delivered every 8 weeks during the maintenance phase ; in keeping with this practice, we injected additional aflibercept at 2-month intervals, while ranibizumab continued to be injected at 1-month intervals following the loading injections.
Statistical Analysis
SPSS software version 13.0 (SPSS Inc, Chicago, Illinois, USA) was used for all statistical analyses. Frequencies were compared between the groups using either the χ 2 test or Fisher exact test. Continuous variables were compared using unpaired t tests. A P value <.05 was considered statistically significant.
Results
Baseline Characteristics
A total of 141 eyes (129 patients) had been diagnosed with PCV at our institution during the study period; of these, 43 eyes were excluded for the following reasons: follow-up loss within 12 months (29 eyes), concomitant PDT treatment (6 eyes), treatment using both ranibizumab and aflibercept (8 eyes). Ultimately, a total of 98 PCV-positive eyes in 90 patients (63 men, 27 women) were enrolled in the study. All patients were South Korean, and the mean age of the entire study group was 63.7 ± 9.5 years. Table 1 outlines the clinical details of the patients included in the study.
| Aflibercept-Treated Group (38 Eyes) | Ranibizumab-Treated Group (60 Eyes) | P Value | |
|---|---|---|---|
| Age, y; mean ± SD | 65.37 ± 9.24 | 63.22 ± 10.44 | .563 a |
| Sex | .881 b | ||
| Male | 26 (68.4%) | 41 (68.3%) | |
| Female | 12 (31.6%) | 19 (31.7%) | |
| Baseline BCVA (logMAR) Snellen equivalent | 0.63 ± 0.49 (20/85) | 0.66 ± 0.43 (20/91) | .625 a |
| Baseline central foveal thickness, μm; mean ± SD | 396 ± 167 | 402 ± 198 | .358 a |
| PCV location, n (%) | .382 c | ||
| Macular | 32 (84.2%) | 53 (88.3%) | |
| Peripapillary | 5 (13.2%) | 5 (8.4%) | |
| Periphery | 1 (2.6%) | 2 (3.3%) | |
| GLD on ICGA, μm; mean ± SD | 3117 ± 923 | 2965 ± 1011 | .329 a |
| Size of branching vascular network, mm 2 ; mean ± SD | 2.05 ± 1.81 | 2.48 ± 1.64 | .336 a |
| Size of largest polyp, μm; mean ± SD | 425 ± 166 | 411 ± 196 | .328 a |
| Total polyp area, mm 2 ; mean ± SD | 0.41 ± 0.11 | 0.38 ± 0.17 | .727 a |
| Pattern of polypoidal lesion, n (%) | .466 b | ||
| Solitary polyp | 27 (71.1%) | 47 (78.3%) | |
| Cluster of polyps | 11 (28.9%) | 13 (21.7%) | |
| Subretinal hemorrhage, n (%) | 13 (34.2%) | 22 (36.7%) | .815 b |
| Pigment epithelial detachment, n (%) | 20 (52.6%) | 29 (48.3%) | .343 b |
| Concurrent type 2 CNV, n (%) | 7 (18.4%) | 12 (20.0%) | .439 b |
| Choroidal vascular hyperpermeability, n (%) | 15 (39.5%) | 25 (41.6%) | .552 b |
| Subfoveal choroidal thickness, μm; mean ± SD | 258 ± 93 | 265 ± 101 | .772 a |
At baseline, there were no significant differences between the aflibercept-treated eyes (38 eyes) and ranibizumab-treated eyes (60 eyes) with regard to BCVA, central foveal thickness, GLD size, size of branching vascular network, size of largest polyp, total polyp area, incidence of subretinal hemorrhage, incidence of PED, incidence of choroidal vascular hyperpermeability, or subfoveal choroidal thickness ( Table 1 ). There were no complications associated with the injections, such as endophthalmitis, traumatic lens injury, or retinal detachment. None of the patients developed any systemic complications, such as cerebrovascular accident, after the injections.
Visual Outcome
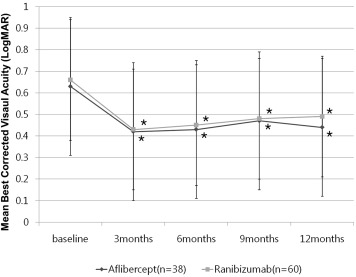
In both groups, the mean BCVA had significantly improved from baseline after 3, 6, 9, and 12 months ( Figure 1 ). Specifically, in the aflibercept-treated group, the mean BCVA (logMAR [Snellen equivalent]) had improved from 0.63 ± 0.49 (20/85) to 0.44 ± 0.37 (20/55) after 12 months ( P = .012). Similarly, in the ranibizumab-treated group, the BCVA had improved from 0.66 ± 0.43 (20/91) to 0.49 ± 0.36 (20/61) after 12 months of treatment ( P = .018). However, there was no significant difference between the groups in terms of BCVA improvement ( Table 2 ). The mean injection numbers did not differ significantly between the groups, either (4.94 ± 1.34 in the aflibercept-treated group and 5.52 ± 1.91 in the ranibizumab-treated group; P = .152; Table 2 ).
| Aflibercept (38 Eyes) | Ranibizumab (60 Eyes) | P Value | |
|---|---|---|---|
| LogMAR at 12 months, mean ± SD (Snellen equivalent) | 0.44 ± 0.37 (20/55) | 0.49 ± 0.36 (20/61) | .828 a |
| Mean logMAR change from baseline | −0.19 | −0.17 | .771 a |
| BCVA changes, n (%) | |||
| Improved ≥logMAR 0.3 | 12 (31.6%) | 16 (26.7%) | .253 b |
| Worsened ≥logMAR 0.3 | 9 (23.6%) | 12 (20.0%) | .852 b |
| Mean central foveal thickness changes from baseline, μm | −184 | −162 | .742 a |
| Polyp regression, n (%) | |||
| Regression | 15 (39.5%) | 13 (21.6%) | .007 b |
| Partial regression | 12 (31.6%) | 10 (16.7%) | |
| Persistence | 11 (28.9%) | 37 (61.7%) | |
| Number of injections, mean ± SD | 4.94 ± 1.34 | 5.52 ± 1.91 | .152 a |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


