Intraocular Pressure: Measurement, Regulation, and Flow Relationships
Robert L. Stamper
Omar Punjabi
George Tanaka
The regulation of intraocular pressure (IOP) plays a critical role in ocular health, because derangements of IOP may lead to profound structural alterations in the eye. Very low IOP may cause refractive changes, inflammation, cataract, maculopathy, and papilledema. Acute elevations of IOP may cause corneal edema, iris sphincter paralysis, iris atrophy, lens opacities, and optic nerve damage; more gradual elevations may be responsible for slow, but profound ganglion cell loss and consequent optic nerve degeneration.
The measurement of IOP is a vital component of an ocular examination. The principles and shortcomings of the measurement method used, however, must be considered with any given numeric value of IOP. In addition, IOP readings should always be interpreted within a patient’s larger clinical context.
This chapter reviews the normal characteristics of IOP and factors causing variations. Pharmacology relevant to IOP and methods of measuring the IOP are reviewed, followed by quantitative pressure-volume-flow relations and a discussion of each type of tonometer.
Normal IOP
IOP results from a dynamic balance between aqueous humor formation and outflow, which are nearly equal under normal conditions. Aqueous formation has two components: a hydrostatic component, produced by leakage of fluid from the blood, and a secretory component, resulting from the active transport of sodium and other ions by the ciliary epithelium.1,2,3
After being produced by the ciliary processes in the posterior chamber, the aqueous circulates throughout the eye, coming into contact with the vitreous, lens, iris, and corneal endothelium. Aqueous drainage occurs by two pathways at the iridocorneal angle. Trabecular or conventional outflow, which predominates in humans, involves passage of aqueous through the trabecular meshwork and into Schlemm’s canal, collector channels, and episcleral veins. A small portion of aqueous exits the eye by the uveoscleral or unconventional pathway across the anterior ciliary muscle and iris root, into the suprachoroidal space, and out through the emissarial channels of the sclera.
The episcleral veins have an internal pressure that reflects the central venous pressure. The episcleral venous pressure (EVP) is the lower limit of IOP in an intact eye if uveoscleral outflow is ignored. An increase in the EVP makes it more difficult for the aqueous to drain and leads to increased IOP.4
Characteristics
The average IOP is thought to be about 15 mmHg.2,5 Under normal conditions, IOP is distributed evenly throughout the eye; therefore, the perfusion pressure of the retina, choroid, and ciliary body must be greater than IOP before blood can flow through these structures. The intraocular tissue pressure of about 15 mmHg is higher than elsewhere in the body, where the average is about 5 mmHg.6
The distensible uveal tissue absorbs about 2 mmHg of the IOP. The potential space between the uvea and sclera, the suprachoroidal space, therefore has a pressure about 2 mmHg lower than the IOP.7 After glaucoma-filtering surgery, the IOP may become very low and the suprachoroidal space may fill with a plasma transudate (choroidal effusion) and cause detachment of the choroid and ciliary body.
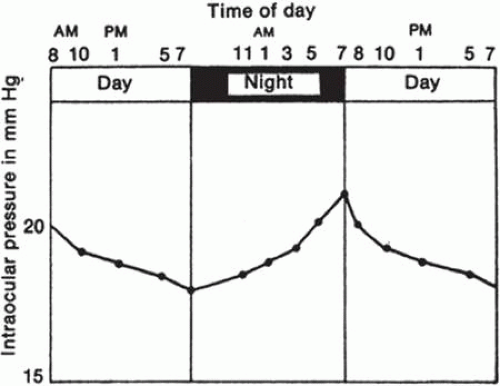
The normal IOP is pulsatile, reflecting its vascular origin (Fig. 1).8 The pulses follow the arterial pulses, and a diagnosis of cardiac arrhythmia can actually be made from a continuous measurement of the IOP.9 The amplitude of the pulse is generally 2 to 3 mmHg, but may be higher if there is a large arterial pulse pressure, such as in hypertension or aortic regurgitation.8 Some have postulated that a low intraocular pulse amplitude is an indicator of reduced blood flow to the ciliary body.10 This can be most readily seen in carotid occlusive disease; whether a reduced IOP pulse amplitude is specific enough to truly indicate a reduced ocular blood flow remains to be demonstrated.11
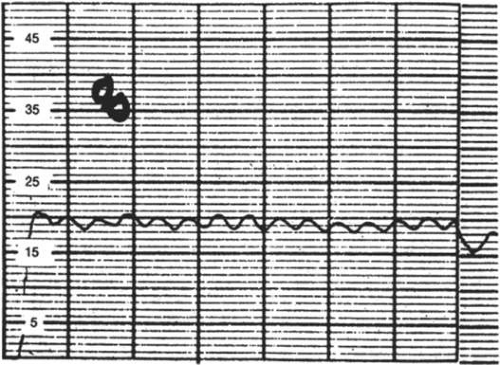 FIGURE 1. The intraocular pressure is pulsatile in nature, having a normal magnitude of 2 mmHg, as shown in this tracing made with a pneumotonometer. |
The IOP is a dynamic function like heart rate and blood pressure and is influenced by many factors. A single measurement of IOP does not necessarily reflect the average pressure in that hour, day, or week.
Diurnal Variation
IOP in humans follows a diurnal rhythm.4,8 Most commonly, the pressure is highest in the early morning (around 7 AM), and lowest in the evening (around 5 PM; Fig. 2), but considerable individual variation exists regarding the time of day or night when the peak and nadir occur.12,13,14 The IOP usually varies <5 mmHg from peak to nadir, although differences as large as 15 mmHg have been found. Some people with elevated IOP are found to have large diurnal peaks and troughs15 and can show variations of as much as 8 mmHg from hour to hour.16 IOP rises in the supine position with larger increases in glaucoma patients; one study suggests that supine IOP predicts peak IOP better than any sitting measurement in the office.17 Nocturnal peaks of IOP occur more frequently with aging possibly related to the supine position.18 Irregular sleep patterns alter the usual diurnal pattern of IOP.19
The diurnal pattern of IOP has been correlated with circadian rhythms of other functions, such as blood pressure, body temperature, and adrenal corticosteroid secretion. In many subjects, the IOP rises several hours after an increase in plasma adrenal corticosteroids.20,21 This pattern may be due to changes in both aqueous secretion and outflow resistance.22,23
The existence of diurnal variation implies that a single random IOP measurement does not adequately represent average pressure over a 24-hour period. Peak IOP can only be obtained by around the clock measurements or with measurements taken at different times of the day. In glaucoma patients, normal IOPs recorded during office hours can give a false clinical impression of adequate pressure control.24,25,26 Phelps et al. argued that a single office pressure measurement was likely, though not certain, to be representative of the peak diurnal pressure.27 Zeimer, however, using data from Phelps et al., calculated that the 95% confidence interval for such a prediction was ± 6.6 mmHg, an uncertainty of more than 13 mmHg.14 Jonas et al. determined that a single in office IOP measurement had a 75% chance of missing the peak IOP.28 Sequential IOP measurements in the office may be helpful in determining both the range and peaks of IOP.29 A small (approximately 1.5 mmHg) seasonal variation in IOP has been described, with the highest pressures recorded in winter and the lowest in summer.26,30
Influencing Factors
The IOP is subject to numerous short- and long-term influences (Tables 1 and 2). The IOP undergoes rhythmic oscillations other than those associated with the arterial pulse. It varies with the respiratory cycle, presumably through changes in venous pressure. Vasomotor variations are associated with waves having a periodicity of 3 to 8 per second (Traube-Hering waves). Although each of these variations produces changes of only 1 to 2 mmHg, the difference between the highest and lowest pressure over a 1-minute period can be as much as 8 mmHg.8
TABLE 7-1. Factors Raising Intraocular Pressure | |
|---|---|
|
TABLE 7-2. Factors Lowering Intraocular Pressure | |
|---|---|
|
The IOP is relatively immune to physiologic changes in arterial blood pressure. Large swings in blood pressure cause the IOP to vary in the same direction, however.31,32 Ligating one carotid artery causes a drop in IOP on that side, presumably due to a decrease in aqueous secretion, although the effect diminishes over several days.33 External carotid compression has a similar result, and measurements of the magnitude of the pressure drop and the rate of recovery have been used to estimate carotid blood flow.34 Interestingly, when one carotid artery is ligated or compressed, the IOP in the contralateral eye rises, possibly due to the increased blood flow and pressure in the contralateral carotid artery.32
Obstruction of the venous return from the eye or head raises the venous pressure in the episcleral system and thus raises the IOP. The IOP rises about 0.8 mmHg for each 1-mmHg rise in episcleral venous pressure (EVP).35 Conditions raising EVP include external pressure on the jugular vein, compression of the superior vena cava by a tumor, cavernous sinus thrombosis, arteriovenous fistula in the cavernous sinus (transmitting arterial pressure directly to the venous system), and any change in the orbital apex (e.g., neoplastic, inflammatory, dysthyroid) that compromises the venous drainage of the eye. A tight collar or necktie may also compress the jugular vein and raise the IOP.36 However, this effect may be marked only in eyes with pre-existing glaucoma and may not last long.37,38
The Valsalva maneuver, in which expiration is forced against a closed glottis producing no air flow, is a related condition that increases venous pressure and hence IOP.39 Increased intrathoracic pressure collapses the superior vena cava, obstructing venous inflow from the head. Coughing and straining with defecation produce similar changes. Straining at weight lifting will raise the IOP, which is further increased by holding one’s breath at the same time.40 Some evidence exists to implicate increased episcleral venous pressure as contributing to the pressure rise with weight lifting type exercise.41 Increased IOP has been associated with blowing a high-resistance musical wind instrument, such as a trumpet.42,43 The Valsalva maneuver will also narrow the anterior chamber angle and thicken the ciliary body, which may precipitate angle closure in susceptible eyes.44
Changes in position affect the IOP. There is a 2- to 4-mmHg increase in IOP when changing from the sitting or upright position to the supine position.45,46,47 The elevation of IOP in the supine position explains some but not all of the nocturnal elevation seen in both healthy and glaucomatous eyes.48,49 In glaucomatous eyes, the peak nocturnal IOP can be best estimated by the daytime supine IOP even though the supine position does not account for most of the nocturnal rise.50 Light does not interfere with the nocturnal, sleep associated rise in IOP.51 Placing the head below the level of the heart in Trendelenburg position may cause a considerable temporary increase in the IOP.52,53 Inverted posture may rapidly lead to IOPs of more than 30 mmHg.54 These changes parallel an increase in the venous pressure in the head, but whether this is the mechanism for the increased IOP is not known. Some patients with glaucoma may have a much larger change in IOP with position than normal individuals.55
External pressure on the eye increases the IOP, at least initially. The eye cannot expand significantly to accommodate the fluid displaced by indenting the globe and the IOP rises. The external pressure, however, accelerates the rate of aqueous outflow and the displaced volume eventually leaves the eye.1 If the external pressure is released, the decreased intraocular volume yields an IOP that is lower than the initial IOP before applying external pressure. This sequence of events forms the basis by which repeated tonometry decreases IOP and is why some surgeons massage the eye before intraocular surgery.56,57 Measurement of outflow facility in an eye during tonography is also based on the decrease in IOP after external pressure.
Forced eyelid closure is a form of external pressure on the eye. Blinking causes a pressure rise of 5 to 10 mmHg, and forced blinking has resulted in recorded IOPs as high as 90 mmHg.58 “Squeezing the eyelids during either Goldmann tonometry or Tonopen readings can cause an average elevation of IOP of about 2 mmHg in normals and 4 mmHg in glaucoma eyes.59,60 Some glaucoma eyes can have as much as an 8 mmHg rise.54 Voluntary lid fissure widening, as is common during tonometry, is associated with an increased IOP of about 2 mmHg, whereas pressure on the eyelid caused by holding the eyelids open may result in much larger increases.61 Closed vitrectomy can subject an eye to high IOPs as well as wide fluctuations of the IOP.62
Attempts to move the globe in a direction opposite to a mechanically restricted extraocular muscle raises the IOP. It is thought that this pressure rise is due to compression of the eye between the restricted muscle and the contracting muscle. This occurs, for example, when upgaze is attempted and the inferior rectus muscle is entrapped by an orbital floor fracture. With dysthyroid involvement of the inferior rectus muscle, the IOP on attempted upgaze may rise 3 to 10 mmHg.63,64 IOP in normal individuals does not rise more than 2 mmHg with upgaze. Therefore, this phenomenon can be a helpful diagnostic sign when recognized. If unrecognized in a thyroid patient, it can cause an overestimation of resting IOP.
A similar increase in IOP is seen when the extraocular muscles co-contract, as in Duane’s syndrome, or after aberrant regeneration of a previously injured third nerve.4 IOP may increase after intravenous succinylcholine, a muscle-depolarizing agent commonly used by anesthesiologists to paralyze the skeletal muscles. Succinylcholine is generally thought to raise IOP by causing tonic extraocular muscle contraction; therefore, clinicians have avoided its use in patients with open eye injuries to avoid extrusion of global contents.65,66,44,45 Kelly et al.,67 however, have shown that succinylcholine raises IOP even when the extraocular muscles are detached from the globe. They attribute the ocular hypertensive effect of succinylcholine mainly to its cycloplegic action and to a lesser degree on its effect on EVP and choroidal blood volume. Nondepolarizing muscle relaxants, such as pancuronium or atracurium, do not raise the IOP.68,69
Exercise can decrease the IOP, perhaps because of the acidosis produced by short-term physical exertion, although this mechanism has been disputed.70,71 Even brief physical exertion can cause a mild lowering of IOP, possibly through sympathetic constriction of choroidal vessels and reduction in uveal volume.72 Continuous strenuous exercise can significantly reduce IOP, probably through increased plasma osmolarity.73 Physical exertion of longer duration appears to have less effect on the IOP but exercise conditioning has been reported to lead to a small decrease in baseline IOP.74 This effect may be more pronounced in patients with higher baseline IOPs.75
Blunt ocular trauma usually has a biphasic effect on IOP: there is initially a transient rise, followed by a more prolonged decline until the injury resolves.1 After trauma, inflammatory debris or swelling of the trabecular beams may obstruct aqueous outflow. Prostaglandins may also play a role. In rabbits, topical application of prostaglandin-E2 causes a rise in IOP, which is prevented by pretreatment with prostaglandin synthetase inhibitors such as aspirin and indomethacin.76 The role of humoral factors is supported by the observation of consensual responses in the contralateral eye after ocular trauma and topical prostaglandin application.77 The late, more prolonged decrease in IOP after trauma is probably due to the decrease in aqueous secretion usually seen in inflammatory conditions.
The level of accommodation has a small effect on the IOP. Accommodative effort decreases the IOP, whereas relaxation of accommodation returns the IOP to baseline values.78,79 These changes parallel the larger effects of parasympathomimetic and parasympatholytic agents. Viscoelastic materials injected into the eye during surgery can also cause significant elevation of IOP.80,81
The IOP is depressed by general anesthesia, irrespective of the type of anesthetic.63,82 Possible mechanisms include muscle relaxation, decreased blood pressure, increased blood carbon dioxide levels, and a direct central effect of the anesthetic agent.76,83 One important exception is ketamine, which may transiently raise IOP when given intravenously.60 Retrobulbar anesthesia causes a considerable drop in IOP due to inhibition of the ciliary body and loss of extraocular muscle tone.1 A large volume of anesthetic instilled into the retro- or peribulbar space may initially compress the globe and raise IOP, however. Similarly, a retrobulbar hemorrhage induced by the needle for retrobulbar anesthesia may compress the globe and raise IOP to fairly high levels.
Changes in plasma osmolality profoundly affect the IOP.4 Water passes freely across the blood-aqueous and blood-vitreous interfaces in either direction, but solute molecules such as ions exchange at much slower rates. If the total concentration of solute molecules in the blood exceeds the concentration in the aqueous and vitreous, water from the vitreous and probably aqueous is drawn into the plasma. This loss of water from the eye decreases the IOP. Interestingly, the empirically measured water loss is less than osmolality calculations predict, suggesting that there are limiting factors in the eye in addition to compensatory mechanisms.4 After several hours, the concentration of solute increases in the intraocular fluids, and the IOP returns toward baseline. It may even exceed baseline, resulting in a “rebound” phenomenon.84
The ocular response to plasma hyperosmolality is used clinically in situations such as acute angle closure glaucoma, in which the IOP has risen to dangerous levels. Hyperosmolar agents are given to raise plasma osmolality and lower IOP. An effective hyperosmolar agent must penetrate the eye relatively slowly. If osmotic equilibrium is obtained too quickly, the IOP effect is short-lived.5,61 Useful and relatively nontoxic agents include intravenous mannitol and oral glycerol and isosorbide. Oral ethanol can also raise plasma osmolality and reduce IOP indirectly by reducing the production of antidiuretic hormone.85
Conversely, if the concentration of solute in the plasma is lower than that of the intraocular fluids, water enters the eye from the plasma and raises IOP.4,86 This pressure increase may be considerable in glaucoma patients and forms the basis for the water-provocative test.5 In this test, a patient rapidly drinks a liter of water, lowering the plasma osmolality. Patients exhibiting an IOP increase of more than 8 mmHg were in the past thought to have glaucoma. The water provocative test, however, has a high false-positive rate and limited diagnostic value.5 Recent studies suggest that a positive water drinking test may identify patients whose glaucoma is at risk of progressing.87 Nevertheless, the sensitivity of the IOP to hypo-osmolality in glaucoma patients should be considered, particularly when there are alterations in systemic fluids and electrolytes. IOP, for example, may increase significantly during hemodialysis. Whether or this effect is mediated by changes in plasma osmolality remains controversial.88,89
A drop in body temperature causes a decrease in the IOP, probably by inhibition of aqueous secretion. A relatively large change in temperature (more than 1°) is necessary to produce significant pressure lowering, however; it is therefore unlikely that temperature plays a major role in physiologic variations of IOP.90 A relatively rapid ascent to high altitude (>4,000 m) causes a reduction in IOP, which returns to pre-ascent levels after acclimatization has taken effect.91
Finally, blood pH affects IOP, with systemic acidosis lowering IOP. In addition to inhibiting carbonic anhydrase in the ciliary body, oral and parenteral carbonic anhydrase inhibitors lower IOP to some extent by producing a systemic metabolic acidosis.92
Pharmacology
The most commonly used drugs that affect IOP include parasympathomimetics, sympathomimetics, carbonic anhydrase inhibitors, prostaglandins, osmotic agents, and corticosteroids.
Parasympathetic Drugs
Parasympathetic drugs, commonly known as miotics, cause constriction of the pupillary sphincter (miosis) and ciliary muscles. Contraction of the ciliary muscles, which insert on the scleral spur, causes a mechanical alteration in the trabecular meshwork, resulting in an increase in aqueous outflow facility and decrease in IOP.93 The decrease in IOP is independent of miosis and seems to be greater when the baseline pressure is higher. Ciliary muscle contraction also results in a significant reduction of uveoscleral outflow.94 In addition, miotics cause forward movement of the lens, anterior chamber shallowing, iris and ciliary body vasodilation, breakdown of the blood-aqueous barrier, and cataract formation.
The drugs in this group share the same qualitative effects; they differ mainly in their potency. Pilocarpine, the most commonly used miotic, acts directly on the ciliary muscle. Carbachol, which is more potent than pilocarpine, acts directly on the muscle and also inhibits acetylcholinesterase, the enzyme responsible for the catabolism of the neurotransmitter acetylcholine. Drugs acting as pure acetylcholinesterase inhibitors include physostigmine (eserine), echothiophate, demecarium bromide, and isoflurophate. The cholinesterase inhibitors are generally more potent than pilocarpine and carbachol.95 In eyes with closed anterior chamber angles, miotics are ineffective in increasing outflow facility. They may even cause a paradoxical rise in IOP by decreasing uveoscleral outflow.
Parasympatholytic drugs have the opposite effect of their parasympathetic counterparts. Parasympatholytic agents inhibit ciliary muscle contraction, reduce trabecular outflow facility, and increase uveoscleral outflow. They are used to dilate the pupil for fundus examination, prevent accommodation, reduce inflammation, and prevent ciliary spasm. Drugs in this class include atropine, homatropine, scopolamine, cyclopentolate, and tropicamide. Some patients, including those with open-angle glaucoma, are more sensitive than others to the effects of parasympatholytic agents and may respond to such an agent with pressure rises that can exceed 8 mmHg.96 These drugs are competitive inhibitors of the parasympathomimetics. If used concomitantly, these two types of drugs will usually cancel each other’s effects.97 In eyes with narrow angles, parasympatholytic drugs may precipitate angle closure as part of their pupil dilation effect. This is an effect distinct from their brief effect on the ciliary muscle. Systemic agents with anticholinergic action, such as phenothiazines and antihistamines, can elevate IOP either by directly inhibiting aqueous outflow or by inducing angle closure.
Sympathomimetic Drugs
The effects of adrenergic drugs on the IOP are complex and seemingly paradoxical. Topical adrenergic agonists such as epinephrine cause a decrease in IOP. Surprisingly, β-adrenergic blocking agents such as propranolol and timolol also reduce the IOP. Sympathomimetic pharmacology has not been fully elucidated, but the paradox may result from differing sites of action for each drug. The effect of epinephrine, an α- and β-agonist, is largely due to an increase in outflow facility, and is probably controlled by adrenergic receptors serving outflow pathways.98 Some evidence suggests that the uveoscleral outflow pathway is primarily affected.99 A study showed that dipivefrin, another α- and β-agonist, reduces blood flow in the ciliary body.100 β-Blockers such as timolol appear to exert their hypotensive effect by reducing aqueous production, perhaps by inhibiting tonic β-adrenergic-controlled formation of aqueous; however, some evidence exists to implicate a direct action of β-blockers on ciliary epithelium.101,102
Two kinds of α-adrenergic receptors exist: α1 and α2. Stimulation of ocular α1-receptors leads to elevation of IOP, mydriasis, eyelid retraction, and vasoconstriction. Ocular α2-receptors mediate a reduction in IOP, probably by inhibiting adenylate cyclase. This causes a decrease in cyclic adenosine monophosphate and subsequently a decrease in norepinephrine release and decrease in β2 stimulation, resulting in a reduction in aqueous production.103 Clonidine, apraclonidine, and brimonidine are α-adrenergic agonists that lower IOP in humans.104 Clonidine acts centrally on α-adrenergic receptors in the brainstem, reducing sympathetic outflow. This leads to decreased vascular resistance, hypotension, bradycardia, and sedation.105 In the eye, clonidine causes decreased aqueous production and conjunctival vasoconstriction. Apraclonidine, a more polar analogue of clonidine, crosses the blood-brain barrier less readily and has fewer systemic effects than clonidine.96 This drug decreases aqueous production, decreases EVP, and increases trabecular outflow facility.106 Brimonidine is more selective for α2-receptors than apraclonidine.107 Brimonidine has been shown to decrease aqueous production and increase uveoscleral outflow without affecting EVP.108 Interestingly, brimonidine has been shown to have neuroprotective activity in an animal model of optic nerve injury.109 Despite several studies confirming brimonidine’s neuroprotective activity in multiple in vitro and animal models, the clinical implications of this finding have remained elusive.
Carbonic Anhydrase Inhibitors
Carbonic anhydrase inhibitors are sulfonamide derivatives that compete for the active site of carbonic anhydrase. Inhibition of this enzyme leads to decreased bicarbonate production and sodium co-transport, thereby reducing aqueous production. Carbonic anhydrase inhibitors also induce ciliary body vasoconstriction and disrupt normal intraocular buffering systems.110 Orally administered agents (acetazolamide, methazolamide, ethoxzolamide, dichlorphenamide) cause a systemic acidosis, which contributes to decreased IOP. Dorzolamide, a topical carbonic anhydrase inhibitor, does not alter systemic acid-base balance and has somewhat less effect on lowering pressure compared to the systemic agents.111 A similar effect can be seen with brinzolamide–another topical carbonic anhydrase inhibitor.112 Both topical agents lower IOP by decreasing aqueous production.113
Prostaglandins
Prostaglandins are fatty acid derivatives produced throughout the body, including ocular tissue. Initial studies using rabbits showed that prostaglandins given intracamerally or topically caused conjunctival hyperemia, miosis, iris vasodilation, blood-aqueous barrier breakdown, and increased IOP.114 In contrast, subsequent experiments in other animals with lower dosages showed marked ocular hypotension.115 Prostaglandin compounds initially tested in humans showed significant external side effects. The phenyl-substituted prostaglandin-F2α analog PhXA41 (latanoprost) was found to have relatively few side effects while significantly lowering IOP.116
Latanoprost increases uveoscleral outflow with minimal effect on outflow facility and aqueous production.117 The mechanism by which latanoprost increases uveoscleral outflow may involve activation of enzymes that degrade ciliary muscle extracellular matrix.118,119,120 Latanoprost has few systemic side effects but causes increased iris pigmentation by promoting increased melanin production in iris melanocytes. There appears to be little long-term significance to this unique ocular side effect. The prostaglandins also may increase periocular skin pigmentation and cause eye lashes to grow longer, darker and curlier.121 Travoprost is another prostaglandin agonist with effects similar to latanoprost.122 Bimatoprost, which may or may not be a prostaglandin analog, also lowers pressure by improving uveoscleral outflow; however, this agent may also increase trabecular outflow.123
Osmotic Agents
Urea and mannitol are intravenous osmotic agents used to raise plasma osmolality and reduce IOP relatively rapidly.5,124 Glycerol and isosorbide can be given orally. Glycerol should be used with great caution in diabetic patients because it may precipitate ketoacidosis. Ethyl alcohol may be used when no other agents are available. All osmotic agents can cause significant hemodynamic alterations in patients with compromised cardiac and renal function.
Corticosteroids
Prolonged administration of topical and systemic corticosteroids can produce a significant IOP increase in susceptible individuals.125,126 About 26% of the normal population have a modest increase of IOP of up to 8 mmHg after 4 to 6 weeks of topical use, whereas 4% have a >8-mmHg rise. More than 90% of primary open-angle glaucoma patients in one series had their IOP elevated by topical corticosteroids.127 In some otherwise normal patients, a syndrome indistinguishable from primary open-angle glaucoma can be produced complete with optic nerve damage after prolonged use of topical corticosteroids.128 Systemic corticosteroids take longer to raise IOP, probably because of less ocular penetration than topical preparations.129
Studies by both Becker130 and Armaly131 strongly suggest that the susceptibility to the IOP-raising effects of steroids is inherited and may be closely linked to the gene(s) for primary open-angle glaucoma. IOP response to steroids may be a risk factor for future development of primary open-angle glaucoma.132 Relatives of primary open-angle glaucoma patients have a higher percentage of steroid response than the general population.133,134 Furthermore, the pituitary-adrenal axis, conjunctival fibroblasts, and peripheral blood lymphocytes of steroid responders were more sensitive to the effects of corticosteroids than those of nonresponders.135 These studies suggest a genetically determined hypersensitivity to corticosteroids on the part of primary open-angle glaucoma patients, their relatives, and possibly some people with a genetic propensity for glaucoma, but no expressed manifestations of the disease. This hypothesis has been challenged by twin studies performed by Schwartz et al.136
Studies have shown that the increased IOP induced by corticosteroids is due to a decreased outflow facility.137 In rabbits, the decreased outflow may be due to a buildup of mucopolysaccharides in the trabecular meshwork. The exact mechanism for this pressure rise in humans is not known, however. The identification of mutations of the myocilin (trabecular meshwork inducible corticosteroid response [TIGR]) gene in a small but often severely affected percent of glaucoma patients has rekindled interest in the steroid response.138,139
Cannabinoids
Cannabinoids, derivatives of marijuana or other similar plants, have been shown to lower IOP. The mechanism may partly be related to systemic lowering of blood pressure.140,141 Delta-9-tetrahydrocannabinol’s effect on IOP is strongly attenuated (at least in rabbits) by pretreatment with indomethacin suggesting that some of the IOP effect is mediated by prostaglandins.142 Some cannibinoids may also reduce aqueous formation.143,144 Cannabinoid receptors have been found in the ciliary body.145 However, at least one synthetic cannabinoid appears to increase aqueous outflow.146 Tolerance and tachyphylaxis to the IOP lowering effects have been reported.147 Systemic side effects of smoking marijuana include systemic hypotension, postural hypotension, tachycardia, palpitations, and alteration of mental status. Furthermore, more chronic use adds irritability, insomnia, altered sleep cycles, nausea, sweating, salivation, tremor, and weight loss to the side effects seen with acute use.148 The active ingredient in marijuana seems to be delta-9-tetrahydrocannabinol; attempts at delivering synthetic aliquots topically have met with little success in providing significant IOP lowering effects.149,150 Despite over 30 years of looking, no synthetic or natural analogs have been found to be clinically useful either topically or systemically to chronically lower IOP.151 In one study in Costa Rica, chronic usage seemed to results in higher IOPs than nonuse.152
Control of IOP
The IOP in normal individuals varies within a somewhat limited range. Changes in IOP produce compensatory changes in aqueous secretion and outflow, tending to bring the IOP back toward baseline. As IOP rises, aqueous inflow decreases. This is called pseudofacility.153 Although these homeostatic mechanisms could be purely local, many studies have implicated a central control mechanism.
Afferent fibers in the long posterior and short ciliary nerves that respond to changes in IOP have been identified in animals; however, no clear relation between discharge frequency and IOP has been found.154,155 Specialized nerve endings in the scleral spur have been discovered that closely resemble mechano- or baroreceptors elsewhere in the body. These nerve endings may respond to changes in ciliary muscle tone or IOP.156 Stimulation of some parts of the diencephalon causes IOP to increase, whereas stimulation of other areas causes the pressure to decrease.157,158 Changes in IOP can also be produced by administration of certain drugs into the cerebrospinal fluid. These drugs, however, may act by altering blood pressure and body temperature, which are also known to influence IOP. If the optic nerve is cut, the IOP fails to respond in the usual way to intravenous hyperosmotic agents, suggesting that the optic nerve contains fibers controlling IOP.159 Sectioning the optic nerve, however, may reduce the vascular supply to the eye, which may also explain these findings.160
When the cervical sympathetic ganglion is stimulated, the IOP decreases because of diminished aqueous secretion. It gradually returns toward normal, although it never regains baseline levels. Surprisingly, when the cervical sympathetic ganglion is removed, the IOP drops initially and returns to normal in 3 to 4 days. This effect appears to be due to an increase in outflow facility. If catecholamines are given topically or intracamerally, there is a dramatic decrease in IOP known as “denervation hypersensitivity.”161,162 Holland et al. used the drug 6-hydroxy-dopamine to produce a chemical sympathectomy. After the administration of hydroxydopamine, even small doses of topical catecholamines dramatically reduced the IOP.163
Stimulation of the parasympathetic system, which supplies the eye by the third cranial nerve, also causes a decrease in IOP through an increase in aqueous outflow facility.164 Conversely, a decrease in parasympathetic tone decreases the facility of outflow.
Regulation of IOP, however, probably involves more sophisticated mechanisms than the opposing actions of the sympathetic and parasympathetic systems. Work using immunohistochemical techniques has demonstrated the presence of several neurotransmitters and neuropeptides in the anterior segment.165 Vasointestinal peptide, neuropeptide Y, substance P, calcitonin gene-related peptide, and cholecystokinin have been found in ciliary body and trabecular meshwork.
In summary, many aspects of the central nervous system influence the IOP. Pressure-sensitive afferent fibers have been identified, and the IOP is affected by sympathetic, parasympathetic, and diencephalic influences. Innervation of the eye also involves a diversity of biologically active neuropeptides, whose exact function is the subject of continuing investigation. Studies suggest the existence of a control center that maintains pressure homeostasis within the eye, but the nature and location of such a center have yet to be determined.
Population Data
Several descriptive studies of IOP in a presumed normal population have been published. The number of eyes tested ranges from about 100 to almost 20,000. The statistics are remarkably similar, despite differing ethnic groups and methods of measurement. The largest study, by Leydhecker, used the Schiøtz tonometer and found a mean IOP of 15.5 mmHg, with a standard deviation of 2.57 mmHg.8 Studies using the more accurate Goldmann applanation tonometer have found almost the same mean and scatter as the Leydhecker study.8,166,167
Although there is excellent agreement regarding the mean IOP, determining the lower and particularly the upper limits of normal is much more difficult. The classic method for determining the upper limit of normal is to assume that the values for IOP are distributed in a “normal” (gaussian) fashion. Under this assumption, the normal values cluster symmetrically on either side of the mean. If one arbitrarily chooses a point 2 standard deviations above the mean as the upper limit of normal (about 20.5 mmHg), 95% of the population will have values between 10.5 and 20.5 mmHg and 2.5% of the population will be included as abnormal. Alternatively, if one chooses 2.5 standard deviations above the mean as the upper limit of normal (about 24 mmHg), then <1% of the normal population would be included as abnormal.
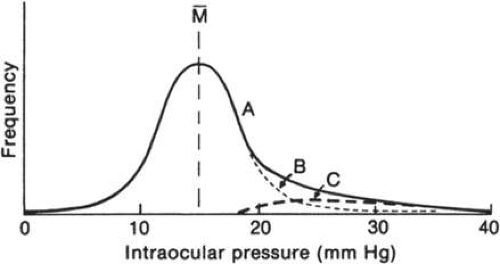
Unfortunately, the distribution of IOP in the population does not fit a bell-shaped gaussian curve.2,8 Instead, there is a skew to the right (Fig. 3), meaning that the number of people who have IOPs higher than the mean is greater than the number having pressures lower than the mean. This situation arises with many physiologic parameters, including blood pressure and fasting blood sugar. Gaussian statistics cannot be accurately applied to a parameter that does not have a gaussian distribution.
It has been assumed that two different populations explain the skewed distribution (Fig. 3). The normal population has a true bell-shaped distribution of IOP. Superimposed on this normal curve is the population with glaucoma, with high pressures explaining the long tail on the right side of the distribution curve. Because the upper end of the normal population and the lower end of the glaucoma population overlap, it is impossible to separate the glaucomatous individual from the high-normal individual using pressure criteria alone.
Armaly168,169 proposed that if the normal population were divided into several subpopulations based on gender and age, the distribution curve of each of these subpopulations would be gaussian in nature. In a study of more than 2,000 individuals, he found a slight increase of mean IOP in each decade above the age of 40. In addition, in people 40 years of age and older, women had slightly higher pressures than men.
The notion that age has an independent effect on IOP has been challenged. Large cross-sectional studies of almost 200,000 Japanese subjects by Shiose actually show a decrease in IOP with age. Shiose suggested that the Japanese data may be reconciled with Western data by postulating that age normally leads to a reduction in IOP, but this effect is overcome in Western populations by the increased prevalence of systolic hypertension and obesity, both of which are associated with increased IOP.170,116 This issue can only be resolved by long-term observations of individuals and not by cross sectional methods. One 10 year prospective study in over 2,000 Japanese confirmed the tendency for IOP to decline with age.171 However, yet another longitudinal study of almost 70,000 Japanese subjects did find an increase in IOP with age associated with an increase in systemic blood pressure.172 Toris et al. showed a decrease in aqueous humor production with a corresponding decrease in uveoscleral outflow at least in Midwestern Americans.173
In the population as a whole, the right and left eyes have equal pressures. The difference between the two eyes rarely exceeds 4 mmHg. An abnormality should be suspected whenever a greater difference is found consistently.8 Women tend to have a slightly lower IOP than men in the same age range.174 In the Barbados Eye Study, systolic blood pressure, diabetes mellitus and age were the most correlated findings with increased IOP in this population of largely black African ancestry.175 Other factors that correlated positively with IOP were female gender (opposite of what was found in Caucasian populations), higher body mass, darker complexion, family history of glaucoma, and smoking. That study also showed, similar to the Baltimore Eye Study, that black subjects had higher IOP than white.176
Armaly’s studies on various populations in Iowa suggest that the level of IOP is inherited as a polygenic multifactorial trait.177 The effects of a common environment are less clear; two studies comparing the IOP of spouses had contradictory results.178,179 In addition, the Framingham Eye Study identified elevated IOP as a marker for decreased life expectancy180; the significance of this finding is not clear.
Abnormal IOPS
Glaucoma
Glaucoma is a disorder usually associated with elevated IOP, in which the optic nerve undergoes a characteristic pattern of damage (cupping) leading to loss of visual function.
Not too long ago, all people with IOPs higher than 2 standard deviations above the mean (22 mmHg) were considered to have glaucoma. This definition has proved inadequate. Longitudinal studies by Armaly,181 Perkins,182 and, most recently, the Ocular Hypertension Treatment Study (OHTS)183 have shown that only a small percentage of individuals with pressures >22 mmHg develop the full clinical picture of glaucoma, at least over a 7- to 10-year period. The OHTS study did confirm that the higher the IOP the greater the risk of developing glaucoma but IOP alone (at least between 24 and 32 mmHg) was a poor predictor of subsequent development of glaucoma. Furthermore, a significant proportion of patients with typical glaucomatous optic nerve damage have IOPs well below 22 mmHg, so-called “normal” or “low-tension” glaucoma. These observations imply that the level of IOP that damages the optic nerve differs between individuals. A cutoff of 21 mmHg for “normal” is arbitrary because it is a combination of optic nerve susceptibility and IOP level that produces glaucoma. Glaucoma is primarily an optic neuropathy, not an IOP abnormality.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree