Purpose
To investigate intraocular concentrations and pharmacokinetics of ranibizumab after a single intravitreal injection in humans.
Design
Prospective, noncomparative, interventional case series.
Methods
We included 18 nonvitrectomized eyes of 18 patients (age range, 61–85 years) that were diagnosed with both clinically significant cataract and macular edema secondary to either exudative age-related macular degeneration, diabetic maculopathy, or retinal vein occlusion. Each eye received a single intravitreal injection of 0.5 mg ranibizumab. An aqueous humor sample was obtained during cataract surgery between 1 and 37 days after injection. Concentrations of unbound ranibizumab in these samples were quantified by enzyme-linked immunosorbent assay.
Results
Ranibizumab concentration in aqueous humor peaked the first day after injection (range, 36.9–66.1 μg/mL) and subsequently declined in a mono-exponential fashion. Nonlinear regression analysis determined an initial peak concentration (c max ) of 56.1 μg/mL and an elimination half-life (t 1/2 ) of 7.19 days with a coefficient of determination (R 2 ) of 0.90. Correction of ranibizumab concentrations for ocular volume as calculated from axial length measurements did not alter regression analysis results significantly (t 1/2 , 7.15 days; R 2 , 0.89).
Conclusions
In human nonvitrectomized eyes, the aqueous half-life of 0.5 mg intravitreally injected ranibizumab is 7.19 days, slightly shorter than the half-life of 9.82 days previously determined for bevacizumab by comparable methods.
Ranibizumab (Lucentis; Genentech, South San Francisco, California, USA) is a recombinant humanized Fab antibody fragment that blocks human vascular endothelial growth factor (VEGF). It is injected intravitreally as an approved treatment for exudative age-related macular degeneration (AMD), diabetic macular edema (DME), and macular edema secondary to central or branch retinal vein occlusion (RVO). In various other retinal pathologies, such as choroidal neovascularization secondary to pathologic myopia or other etiologies and retinopathy of prematurity (ROP), its efficacy is under investigation. Despite this widespread clinical application, the pharmacokinetics of ranibizumab in the human eye are still unknown. Information about its pharmacokinetic properties such as the intraocular elimination half-life is of clinical relevance for optimizing application of the drug and, thus, patient outcome, as well as for comparison with other anti-VEGF agents such as bevacizumab and aflibercept. We have previously reported the pharmacokinetic parameters of bevacizumab in the human eye. Here we determined the pharmacokinetics of ranibizumab in humans using comparable methods.
Methods
Patient Selection and Aqueous Humor Sampling
We included patients treated at the Department of Ophthalmology, University of Bonn. Inclusion criteria were planned elective cataract surgery for clinically significant lens opacification and a concurrent macular edema secondary to AMD, diabetic maculopathy, or central or branch RVO in the same eye that had been treated with a single intravitreal injection of 0.5 mg ranibizumab within 40 days before surgery. Patients with any additional intravitreal injections within 6 months before surgery or with any previous intraocular surgeries were excluded from the study. During cataract surgery, an aqueous humor sample of approximately 0.15 mL was obtained via a corneal paracentesis at the beginning of the procedure and immediately stored at −80 C until analysis.
Ranibizumab ELISA
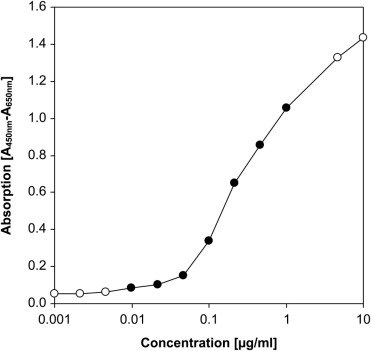
Ranibizumab concentrations in aqueous humor samples were quantified by ELISA, as described by us previously. To reduce experimental variability, all samples were measured together in the same experiment and each sample was analyzed in triplicate. We diluted the samples 1:100 in phosphate-buffered saline (PBS) and incubated 100 μL of each diluted sample in 96-well plates coated with human IgG-specific goat IgG (BD Biocoat; BD Biosciences, San Jose, California, USA) for 1 hour at room temperature. Subsequently, wells were incubated for 1 hour with 40 ng biotinylated recombinant human VEGF 165 (Fluorokine Biotinylated Human VEGF; R&D Systems, Minneapolis, Minnesota, USA) in 100 μL PBS. This was followed by 100 μL peroxidase-conjugated streptavidin (Extravidin-Peroxidase; Sigma, St. Louis, Missouri, USA) diluted 1:2000 in PBS for 1 hour. Peroxidase activity was determined by incubation with 200 μL peroxidase substrate solution (SigmaFast OPD; Sigma). After 30 minutes, absorbance at 450 nm was quantified in a microplate reader with subtraction of reference absorbance at 650 nm. A standard curve was obtained by serial dilutions of ranibizumab in PBS ( Figure 1 ). The ELISA detection range employed in this study comprised 10–1000 ng/mL. Mean results of triplicate measurements were used for statistical analysis.
Correction for Ocular Volume
Axial length of each study eye was measured by optic biometry (IOL Master; Carl Zeiss Meditec, Jena, Germany). Ocular volume was estimated by assuming that the eye approximates a sphere, and thus the following calculation was applied:
Ocular volume [ mL ] = 4/3 × π × ( 1/20 × axial length [ m m ] ) 3
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


