Purpose
To compare immunohistochemical and genetic overlaps and differences between intraocular medulloepitheliomas and embryonal tumors with multilayered rosettes of the brain.
Design
Retrospective histopathologic, immunohistochemical, and genetic analysis of 20 intraocular medulloepitheliomas.
Methods
(1) Review of clinical data and hematoxylin-eosin-stained sections with (2) immunohistochemical staining of paraffin sections using a polyclonal antibody against the protein LIN28A, and (3) fluorescence in situ hybridization (FISH) testing for the amplification of the genetic locus 19q13.42 involving the C19MC cluster of miRNA. Ten retinoblastomas served as controls and to determine the specificity of these biomarkers for intraocular medulloepitheliomas.
Results
Nineteen of the 20 intraocular medulloepitheliomas were either diffusely or focally LIN28A positive (weak, moderate, or strong). The most intense positivity correlated with aggressive behavior such as intraocular tissue invasion or extraocular extension. None of the cases studied by FISH harbored an amplicon for C19MC . The 10 retinoblastomas were LIN28A and C19MC negative.
Conclusion
LIN28A has a putative role in oncogenesis and is found only in embryonic cells and malignancies. Intraocular medulloepitheliomas and embryonal tumors with multilayered rosettes of the brain both display LIN28A positivity. Only the latter, however, display amplification of the 19q13.42 locus involving C19MC , implying that other causative factors are at play in intraocular medulloepitheliomas. More aggressive tumor behavior within the eye can be partially predicted by LIN28A staining intensity.
Intraocular medulloepithelioma is much more common than brain medulloepithelioma, with somewhere between 100 and 200 cases of the former reported mostly in the ophthalmic literature. Classical histopathologic descriptions of the brain tumors found in standard neuropathologic textbooks are brief when compared with those of ocular examples. Case reports of brain medulloepitheliomas predominate in delineating most of the information on its histopathologic and immunohistochemical features. No publication in the neuropathology literature is comparable to the 2 largest ocular series of 56 and 41 cases in the ophthalmic literature. Nonetheless, the shared histomorphologic features of the eye and brain tumors are unmistakable and point to the possibility of an ill-defined closeness. These shared features include papillary, cord-like, and trabecular arrangements and multilaminar “ependymoblastic” rosettes of neoplastic pseudostratified neuroepithelium, with frequently admixed small, round undifferentiated “neuroblastic” cells, and even occasional pigment epithelial cells and heteroplastic or divergent (“teratoid”) stromal differentiations (muscle, cartilage).
The grouping of several primitive neuroectodermal brain tumors with multilayered rosettes and shared immunohistochemical biomarkers and genetic alterations has recently transformed traditional neuropathologic diagnostic schemas and provided new insights into the pathogenesis of brain medulloepithelioma. Brain tumors now categorized together are the recently characterized embryonal tumor with abundant neuropil and true rosettes, ependymoblastoma, and medulloepithelioma, all under the rubric of embryonal tumor with multilayered rosettes. In this article we report 20 cases of intraocular medulloepithelioma using the same biomarkers employed for the evaluation of tumors of the brain. Immunohistochemical testing with a polyclonal antibody for the identification of LIN28A, and fluorescence in situ hybridization (FISH) testing to establish amplification of the genetic locus 19q13.42 involving the C19MC cluster of miRNA, form the core of this study. Through different mechanisms, each of these factors constitutes an enhancer and protector of the neural stem cell compartment, and serves as an oncogenic drive for the development of the brain tumors and possibly for intraocular medulloepitheliomas.
Methods
This retrospective study was conducted under the auspices of the Massachusetts Eye & Ear Infirmary’s Institutional Review Board (IRB protocol number 602340-2, reference number 14-064H), in coordination with those of the Wills Eye Hospital, Duke University, and the University of Heidelberg, in compliance with the US rules and regulations of the Health Insurance Portability and Accountability Act and all applicable US federal and state laws, and in adherence to the tenets of the Declaration of Helsinki.
After a search for the diagnosis of intraocular medulloepithelioma in the regular diagnostic and consultation files in the ophthalmic pathology laboratories of the Massachusetts Eye and Ear Infirmary and the Wills Eye Hospital and in the Duke University Pathology Department from January 1996 to April 2014, 10 cases were found. An additional 10 cases were contributed by the Clinical Cooperation Units of the German Cancer Research Center (DKFZ) and the Department of Neuropathology, Heidelberg University Hospital, Heidelberg, Germany, bringing the total up to 20 cases. Criteria necessary for inclusion in this study were based on the availability of glass slides used in the original diagnosis and of archived blocks for the preparation of additional unstained paraffin sections for immunohistochemical staining or FISH testing.
The histopathologic features displayed in formalin-fixed, paraffin-embedded, and hematoxylin-eosin-stained sections on glass slides were reexamined in all cases (by F.A.J. and A.K.), with complete concordance. The paraffin blocks were used to obtain additional immunohistochemical staining and to perform DNA analysis. Five cases have been separately reported ; the use of a different immunohistochemical stain in the present study and the introduction of DNA analysis with FISH testing and a new investigative focus were the bases for the inclusion of these cases in the present study. Patients’ medical records were evaluated for demographic information. Follow-up information was available for 19 of the 20 cases.
Immunohistochemical staining was performed on all cases employing a polyclonal antibody (A117, #3978, Cell Signaling Inc, Boston, Massachusetts, USA) for the detection of LIN28A. Immunostaining was done using an automated stainer (Benchmark; Ventana XT, Tucson, AZ, USA) with appropriate controls to validate antibody quality. Endogenous peroxidase activity was blocked by H 2 O 2 before antibody incubation. The chromogen diaminobenzidine was used and the tissues were counterstained with hematoxylin. Positive staining of LIN28A was cytoplasmic and not nuclear. The staining was graded as weak, moderate, or strong, and as unifocal, multifocal, or diffuse. Such staining was evaluated on the basis of a single slide. It is possible that multiple-step sections throughout a given lesion might have disclosed slightly different results, although most of the tumors were small and therefore the slides were likely to be representative. In parallel, FISH testing was performed on all samples studied for an abnormality in the 19q13.42/ C19MC locus, according to the methodology for FISH previously described. Fluorescein isothiocyanate–labeled locus probe 634C1 (19q13.42/ C19MC) and digoxigenin-labeled reference probe 2658N19 (19p13) were applied. Signals were scored in 200 nonoverlapping intact nuclei. Specimens were considered amplified for C19MC locus when more than 10% of tumor cells showed either more than 8 signals of this locus probe or innumerable tight clusters of signals. The forgoing tests were conducted in the Department of Neuropathology, University Hospital, Heidelberg, Germany.
Results
Patient demographics, salient histopathologic features, and immunohistochemical and genetic findings are summarized in the Table . Of the 20 patients included in the study, 11 were female and 9 were male, ranging in age from 3 months to 53 years (4 were older than 20 years). The median age was 4.5 and the average age was 11.8 years. The right and left eyes were equally affected. All but 1 tumor arose in the ciliary body region; the exception filled the posterior half of the vitreous cavity and was associated with a postequatorial retinochoroidal coloboma. Follow-up information was available on 19 of the 20 cases. Detailed ocular history was also available on approximately 60% of cases. One patient with a prolonged delay in diagnosis experienced spontaneous extraocular extension and eventually died with distant metastases ( Table , Case 19). Eighteen other patients are alive, and 1 patient ( Table , Case 8) was lost to follow-up. Another patient had a lensectomy performed at the time of a subtotal intraocular biopsy of a tumor, just prior to enucleation ( Table , Case 11). None of the other patients were known to have had previous ocular surgery (cataract or glaucoma) prior to enucleation. Of the 10 cases from Europe, however, 1 ( Table , Case 5) had undergone histopathologically detectable evidence of glaucoma drainage device implantation with extraocular extension around the plate.
| Case# | Age | Sex | Laterality | Routine Histopathology | Follow-up a (Years) | Intensity of LIN28A Staining | Pattern of LIN28A Staining | 19q13.42 Amplification | Other Notable Features b |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 6 years | F | Left | Malignant | 4 | Strong | Diffuse | None | Ruptured globe with extrusion of retina; tumor invading cornea/sclera with destruction of iris/ciliary body |
| 2 | 1 year | F | Left | Malignant | 3 | Moderate | Diffuse | None | Epibulbar extension, intracorneal tumor with destruction of iris |
| 3 | 6 years | M | Right | Malignant | 3 | Strong | Diffuse | None | Large tumor filling eye with neuroblastic areas, extensive invasion of retina |
| 4 | 2 years | M | Right | Malignant | 2 | Moderate | Multifocal | None | Necrotic, retroiridal tumor invading ciliary body |
| 5 | 2 years | M | Left | Malignant | 3 | Moderate | Diffuse | None | Small ciliary body mass, iris neovascularization with angle closure, tumor in glaucoma valve cavity and in angle; neuroblastic areas |
| 6 | 3 years | M | Right | Malignant | 21 | Moderate | Multifocal | None | Small ciliary body mass extending into anterior chamber and along surface of retina, destruction of iris, high nuclear/cytoplasmic ratio, neuroblastic areas |
| 7 | 14 years | F | Right | Malignant | 4 | Moderate | Diffuse | None | Tumor filling vitreous cavity with invasion of cornea/sclera, destruction of retina/ciliary body (choroid spared) |
| 8 | 1 year | F | Left | Malignant | 1 c | Weak | Multifocal | None | Extensive necrosis, invading ciliary body, iris neovascularization |
| 9 | 6 years | M | Left | Malignant | 12 | Strong | Diffuse | None | Massive extraocular extension with extruded retina, circumferential ciliary body involvement, pseudo-neuropil |
| 10 | 3 years | F | Right | Malignant | 4 | Weak | Unifocal | None | Small necrotic ciliary body mass with broad iris-cornea synechiae |
| 11 | 45 years | M | Right | Benign | 1 | Strong | Diffuse | None | Focally pigmented, growing onto retina and iris with iris neovascularization |
| 12 | 30 years | F | Left | Malignant | 5 | Weak | Multifocal | None | Malignant cytology, invading choroid |
| 13 | 3 months | F | Right | Malignant | 5 | Strong | Multifocal | None | Large tumor arising in retinochoroidal coloboma, teratoid with striated muscle “retinoblastic” features, focally invading choroid with iris neovascularization; premedullary epithelium |
| 14 | 6 months | M | Left | Benign | 2 | Negative | Negative | None | Neuropil, benign cytology; premedullary epithelium |
| 15 | 46 years | F | Left | Benign | 0.5 | Strong | Multifocal | None | Focally pigmented tumor extending onto iris and retina without invasion; premedullary epithelium |
| 16 | 2 years | M | Right | Malignant | 2 | Moderate | Unifocal | None | Mostly necrotic with minimal viable tumor extending onto anterior surface of lens, neovascularization of iris |
| 17 | 9 years | M | Right | Malignant | 18 | Strong | Unifocal (large) | None | Large tumor, pseudoadenomatous hyperplasia of nonpigmented ciliary epithelium, neuroblastic areas, neovascularization of iris |
| 18 | 9 months | F | Right | Malignant | 14 | Weak | Focal | None | Neuroblastic areas LIN28A-negative, invades ciliary body/iris |
| 19 | 53 years | F | Left | Malignant | 11 (died of disease) | Strong | Diffuse | None | Extraocular extension, tumor on optic nerve head, marked cytologic atypia |
| 20 | 5 years | F | Left | Benign | 4 | Strong | Multifocal | None | Focally pigmented, neoplastic epiretinal membrane and neoplastic retrolental membrane |
a Alive unless otherwise specified.
b All tumors to varying degrees displayed rosettes, small epithelial clusters with or without lumens, festoons, and strands of cuboidal or pseudostratified epithelium with the additional features listed.
Histopathologically, all tumors showed a varying prominence of multilaminar rosettes or neurotubules, cords or festoons of cells, and small cellular units that were either solid or displayed different-sized lumens and were distributed in an Alcian blue-positive (hyaluronic acid) matrix. Owing to the vagaries of the selection process for the inclusion of tumors in this study, only 1 lesion in this series ( Table , Case 13) was judged to be teratoid based on the presence of myogenin-positive spindle cells. In consideration of the number of mitotic figures per 5 high-power fields (generally >10 for malignancy), the degree of cytologic atypia, focal necrosis, and evidence of invasion or destruction of intraocular structures ( Figure 1 , Upper left), 16 lesions were diagnosed as malignant according to standard criteria in routine hematoxylin-eosin sections before LIN28A testing was performed. Noncytologic features included the following: iris neovascularization (6 cases); spread onto and into the retina (6 cases), 1 extending backward to the optic nerve head ( Figure 1 , Upper right); ciliary body invasion or destruction (6 cases); tumor extrusion or extension through a corneoscleral rupture ( Figure 1 , Middle left) (4 cases); extensive and subtotal tumor necrosis (4 cases); premedullary cuboidal strands of epithelium, usually located behind or on the iris (4 cases); partial pigmentation (3 cases); iris destruction (3 cases); choroidal invasion (2 cases); retrolenticular neoplastic cyclitic membrane (1 case); pseudoadenomatous hyperplasia of the nonpigmented ciliary epithelium contiguous to the tumor (1 case); and neuropil formation (1 case). Regarding the identification of a neuropil, care had to be taken not to confuse the background of invaded retina or reactive retinal gliosis adjacent to the tumor for this feature (observed in 3 cases).
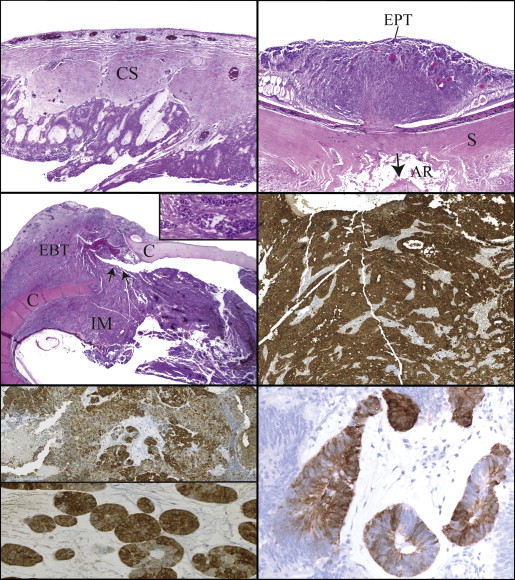
Nineteen of 20 tumors were immunohistochemically LIN28A positive. The immunostaining was localized to the cytoplasm and was strongly positive in 9 cases ( Figure 1 , Middle right), moderately positive in 6 cases ( Figure 1 , Bottom left), and weakly but unequivocally positive in 4 cases ( Figure 1 , Bottom right). Moderately staining tumors manifested either intermediate staining density or heterogeneous staining with some cells being denser than others ( Figure 1 , Bottom left). The staining was diffuse in 8 cases ( Figure 1 , Middle right) or multifocal ( Figure 2 , Upper left, top panel) or unifocal ( Figure 2 , Upper left, bottom panel) in 11 cases. Staining was totally negative in all components of the 1 case with a permeative neuropil and scattered multilaminar rosettes. In correlating these results with local ocular behavior, as revealed in hematoxylin-eosin-stained sections, large size, retinal extension, invasion of the choroid or ciliary body, iris destruction, and extraocular extension ( Figure 2 , Middle left) were encountered only in those tumors exhibiting diffuse cytoplasmic staining that was either moderately or strongly intense. For example, 3 of 4 tumors with extraocular extension stained strongly positive and 3 diffusely positive. The 3 partially pigmented areas of 3 tumors were LIN28A positive, as determined by a lighter brown staining by the chromogen in comparison with the black melanin granules ( Figure 2 , Middle right, left and right panels). The adjacent pseudoadenomatous hyperplasia of the nonpigmented ciliary epithelium discovered next to a medulloepithelioma did not stain for LIN28A.
By means of FISH testing all tumors were found to be negative for 19q13.42 ( C19MC ) amplification ( Figure 2 , Bottom left). Balanced profile for 19q/19p was detected in all ocular medulloepitheliomas, with a similar number of locus/reference probes signals (from 2 to 4 per nuclei) and with reference/control ratio in a range from 0.8 to 1.3 per tumor. For comparison, an example of a positive FISH test of an embryonal tumor with multilayered rosettes is also illustrated ( Figure 2 , Bottom right).
Ten retinoblastomas, 5 almost completely undifferentiated and 5 with both prominent rosettes and undifferentiated round cells, were evaluated for LIN28A and failed to disclose any immunoreactivity in both components in the cells’ nuclei or cytoplasm. DNA rearrangement at 19q13.42 leading to amplification of C19MC was also not discovered in any case.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


