Purpose
To report the therapeutic efficacy of intracameral voriconazole injection in the treatment of fungal endophthalmitis resulting from keratitis.
Design
Retrospective, single-institution, consecutive case series.
Methods
Microbiologic and medical records were reviewed for patients with positive intraocular culture results or proven pathologic features for fungal organisms and clinically diagnosed fungal endophthalmitis resulting from keratitis. Ten eyes were treated with an intracameral injection of 100 μg voriconazole. Clinical characteristics, treatment, and causative organisms were analyzed.
Results
Fusarium and Aspergillus were the most common causative organisms. Voriconazole was injected intracamerally from 1 to 8 times. Of the 7 patients who received 5 or more repeat injections, 6 were caused by Fusarium and 1 by Acremonium . In the remaining 3 patients who were administered 4 or fewer voriconazole injections, the causative organisms were Aspergillus and Alternaria .
Conclusions
Intracameral voriconazole injection may be an effective treatment for fungal endophthalmitis contiguously spreading from keratitis.
Fungal endophthalmitis resulting from contiguous spread by keratitis is a potentially devastating infection. The most common organisms encountered in this disease are Fusarium species, followed by Acremonium and Aspergillus species. Although the prognosis of exogenous fungal endophthalmitis has been shown to be poor, an optimal therapy has not yet been well established, particularly in regard to the route of administration and dosage of antifungal agents. Clinically, exogenous fungal endophthalmitis tends to be more localized, with the fungal mass and inflammation often confined to the anterior chamber, pupillary space, or anterior vitreous. Recently, intracameral amphotericin B injection has been used successfully in the treatment of severe keratomycosis and endophthalmitis, but several shortcomings of intraocular amphotericin B injection have been reported. It can cause retinal necrosis at low concentrations, immediate anterior chamber reactions can develop after injection, and a variety of fungal species showing resistance to amphotericin B have been reported.
Voriconazole, a second-generation triazole, differs from fluconazole by the addition of a methyl group to the propyl backbone and by the substitution of a triazole moiety with a fluoropyrimidine group, resulting in a marked change in activity. Previous studies have shown voriconazole to have a broad spectrum of activity against A spergillus species, Candida species, Paecilomyces lilacinus , Cryptococcus neoformans , Scedosporium species, and others. Clinically, voriconazole has been shown to be effective as a primary therapy in the treatment of invasive aspergillosis and is an effective salvage therapy for refractory infections caused by Fusarium species. In experimental studies, voriconazole has been shown to be less toxic to the retina than amphotericin B and to exhibit exponential decay with a half-life of 2.5 hours in rabbit vitreous and a very low aqueous concentration, below the therapeutic levels of fungal species. Therefore, intracameral voriconazole injection is indicated to achieve a higher aqueous concentration and is considered to be an alternative in the treatment of fungal endophthalmitis spreading as a result of keratitis.
Methods
The medical records of patients diagnosed with filamentous fungal keratitis progressing to endophthalmitis who had undergone treatment with intracameral voriconazole injection and were followed up for at least 4 months at Taichung Veterans General Hospital between January 2005 and December 2008 were reviewed. Risk factors, visual acuity, fungal species involved, presenting clinical findings, treatment, and final outcome data were collected.
Ten cases with positive corneal and intraocular culture results or fungal organisms proven by pathologic analysis were enrolled in this study. All patients had been subjected to the following routine. On presentation, corneal lesions were scraped with a surgical blade for routine diagnostic cultures and smears. Intraocular specimens were obtained under the microscope. Before surgery, the eye was cleaned by a standard method using povidone iodine. A clear corneal incision was made, and the fungal infiltrate and the endothelial plaque region were aspirated gently. The contents of the tape subsequently were inoculated on chocolate agar, blood agar, thioglycollate broth, and Sabouraud agar plate.
The procedure for intracameral voriconazole injections was as follows. Voriconazole (VFEND; Pfizer, Inc, New York, New York, USA) was obtained in pure powder form and was reconstituted in sterile water to obtain a concentration of 100 μg/100 μL. Intracameral voriconazole injection was administered under aseptic conditions using an operating microscope. After instillation of topical proparacaine, a speculum was inserted. If necessary, an aqueous aspiration was performed for diagnosis before voriconazole injection was administered. A volume of 100 μg voriconazole in 0.1 mL was injected into the anterior chamber using a 30-gauge needle attached to a 1.0-mL regular insulin syringe. Intracameral voriconazole injection was given once daily, and the treatment was discontinued while the eyes showed resolution of the anterior chamber fungal infiltrate. If anterior chamber fungal infiltrate or an enlargement of the endothelial plaque subsequently developed in these patients, intracameral voriconazole injection was added to the treatment regimen again.
Keratoplasty was considered in corneas with extensively deep ulcers and melting, impending perforation (descemetocele formation) or presence of perforation, and poor response to antifungal treatments. Graft techniques varied according to size, severity, and stromal depth of infective process. The goal of surgery was removal of infected stroma, detection of causative organisms, and maintenance of the anatomic integrity of the eye.
Results
Of the 10 eyes from 10 patients who met the inclusion criteria, 4 were right eyes, and the mean patient age was 58.0 years (range, 38 to 78 years). Risk factors for the development of keratitis were trauma (7 cases), facial palsy (1 case), and unknown (2 case). Topical corticosteroids were used before the diagnosis of fungal keratitis in 4 of 10 cases. Of the 4 patients who had a history of using topical corticosteroids, 2 were given at a local clinic under the diagnosis of conjunctivitis. The other 2 were combined infections of bacteria and fungus. They underwent topical corticosteroid treatment after resolution of the bacterial keratitis and the fungal keratitis developed subsequently after that. Visual acuity at presentation with endophthalmitis was 6/30 or better in 4 of 10 cases and counting fingers or worse in 5 cases. Intraocular culture results were positive in all patients, including fungal infection proven by pathologic analysis in 3 patients. Fusarium species and Aspergillus species accounted for 6 and 2 of 10 of causative organisms, respectively ( Figure 1 ).
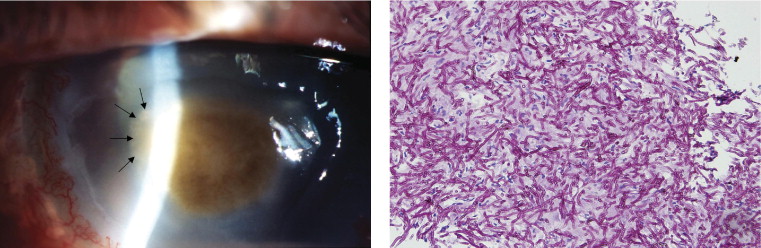
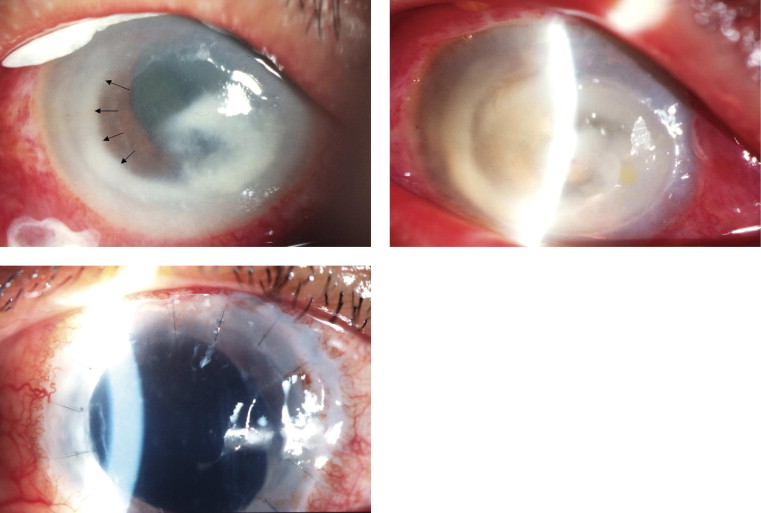
In all eyes, a corneal ulcer exhibiting deep stromal infiltrate was present before eyes were treated with an intracameral injection of voriconazole. A hypopyon and endothelial plaque were present in 6 and 8 of 10 cases, respectively, and an intraocular fungal invasion was visualized on examination in 7 of 10 cases, presenting as a fungal web in 5 cases and as fungal balls in 2 cases. In 2 patients, a rapid melting cornea with extensive corneal inflammation developed ( Figure 2 ).
In the initial management, all 10 eyes were administered 5% topical natamycin every half hour, 1% atropine twice daily, and 200 mg systemic ketoconazole twice daily. In addition, 0.15% topical amphotericin B was given every hour and 1% topical voriconazole was given every hour, which was added to treatment in 3 patients and 2 patients, respectively. Two patients underwent intracameral amphotericin B injection once. Because of the inflammation flaring in the anterior chamber, intracameral injections shifted to voriconazole. Voriconazole was injected intracamerally in all patients, whereas the number of injections varied from 1 to 8. Of the 7 patients who received 5 or more repeat injections, infection was caused by Fusarium species in 6 patients and by Acremonium species in 1 patient. In the remaining 3 patients who were administered 4 or fewer voriconazole injections, the causative organisms were Aspergillus species and Alternaria species.
Of the 4 eyes that underwent keratoplasty, 2 eyes infected with Fusarium species had extensive corneal melting needing a large keratoplasty to maintain the anatomic integrity of the eye. A therapeutic penetrating keratoplasty was performed for intraocular culture and corneal pathologic features in 1 eye with an enlarged central brown infiltrate, and Aspergillus flavus was confirmed. The other case was a patch graft sutured to a perforated cornea during treatment. The 4 eyes showed a partial resolution before keratoplasty, and they continued to receive the treatment of intracameral voriconazole injection after keratoplasty.
In all cases, regardless of ultimate outcome, clinical appearance of the fungal anterior chamber invasion, including hypopyon, fungal web, fungal ball, and endothelial plaque, resolved after the treatment. In cases in which the corneal ulcer was resolved, complete resolution occurred between days 16 and 30. Final visual acuity depended on the location of the remaining scar, the procedure of keratoplasty, and additional intraocular complications. In 4 eyes, vision improved at least 1 Snellen line after treatment. In the remaining 5 eyes, vision did not improve. Patient data are shown in the Table .
| Case No. | Age (yrs) | Sex | Visual Acuity (Initial) | Visual Acuity (Final) | History | Clinical Features | Fungal Organism | Injection Number | Treatment | Surgery |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 78 | M | LP | LP | Trauma, Steroid | Hypopyon, EP, FW | Aspergillus flavus | 4 | TN, OK | TPK |
| 2 | 70 | M | CF/(10 cm) | HM (20 cm) | — | Hypopyon, EP, melting | Fusarium species | 7 | TN, TA, OK, TV | — |
| 3 | 68 | M | 6/30 | 6/20 | Facial palsy | EP; FW, Perforation | Acremonium species | 5 | TN, OK, TV | Patch graft |
| 4 | 38 | F | CF/20 cm | 6/30 | Trauma, Steroid | Hypopyon, FB, melting | Fusarium solani | 7 | TN, OK, ICAI | TPK |
| 5 | 66 | M | CF/20 cm | NLP | Trauma, Steroid | Hypopyon, FB, melting | Fusarium solani | 8 | TN, OK, TA | TPK |
| 6 | 40 | M | CF/20 cm | 2/60 | Trauma | EP, FB | Fusarium species | 5 | TN, OK, TA, ICAI | — |
| 7 | 49 | M | 6/20 | 6/15 | Trauma, Steroid | EP, FW | Aspergillus species | 3 | TN, OK, TA | — |
| 8 | 62 | F | 6/30 | 6/20 | Trauma | EP, FW, Hypopyon | Alternaria species | 1 | TN, OK | — |
| 9 | 46 | F | 6/20 | 3/60 | Trauma | EP, Hypopyon | Fusarium oxysporum | 5 | TN, OK | — |
| 10 | 63 | M | 3/60 | CF/50 cm | — | EP, Hypopyon, FW | Fusarium species | 6 | TN, OK | — |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


