Purpose
To investigate if ultrastructural alterations in the Descemet membrane (DM) are correlated with the clinical outcome after Descemet membrane endothelial keratoplasty (DMEK).
Design
Retrospective cohort study.
Methods
setting : Institutional, single-center. study population : One hundred and twelve residual DM specimens obtained after DM stripping. main outcome measures : Incidence of ultrastructural abnormalities in transmission electron microscopy, graft detachment rate, graft failure rate, best-corrected visual acuity (BCVA), endothelial cell density (ECD), and central corneal thickness (CCT). Examination dates were on the day before DMEK and 1, 3, 6, and 12 months after surgery.
Results
Abnormalities in the ultrastructure of DM were found in 16 of 112 specimens (14%) (abnormal DM group), comprising deposits of long-spacing collagen, fine filaments (proteoglycans), a posterior collagenous layer, pseudoexfoliative material, and guttae. The secondary graft failure rate was significantly higher in the abnormal DM group compared with the normal DM group ( P = .001). There was a trend for an increased graft detachment rate in the abnormal DM group (11/16) compared with the normal DM group (42/96) ( P = .103). There was no significant difference in mean CCT and ECD after surgery. Mean CCT in the eyes with graft failure in the abnormal DM group at the last follow-up before regrafting was 850 μm, indicating endothelial failure with stromal edema.
Conclusion
This study reveals a correlation between ultrastructural alterations of DM in donor corneas and the graft failure rate after DMEK. Thus, graft failure after DMEK not only is determined by surgical trauma and postoperative events but may also be influenced by intrinsic, graft-specific features.
Descemet membrane endothelial keratoplasty (DMEK), a technique for endothelial grafting in which the graft consists of Descemet membrane (DM) and endothelium only, has gained increasing importance during the last 10 years. In contrast to earlier techniques for corneal transplantation, the demands on DMEK are not only to replace diseased tissue but also to gain near-normal visual function. This is enabled by the fact that corneal structure after DMEK closely resembles its physiological constitution. Outcomes after DMEK are determined by different factors:
- (1)
Intraoperative complications: Intraoperative factors influencing DMEK outcomes are the experience of the surgeon, the surgical technique (for example, no-touch technique), and ocular comorbidities (for example, an unstable iris-lens-diaphragm).
- (2)
Postoperative complications: The most common complication of DMEK is graft detachment requiring repeated intracameral air injections. Because of the use of an undersized (compared to the descemetorrhexis size) and well-centered graft, sulfur hexafluoride gas, and a standardized surgical technique, the graft detachment rate has been decreased from up to 82% in the beginnings of DMEK to values below 20%, nowadays requiring additional air injections in only 5%–10%. Endothelial immune reactions occurring in less than 1% play only a minor role in the postoperative course after DMEK.
- (3)
Preoperative characteristics of the graft: Graft quality is determined by the origin and storage conditions of the donor tissue. Beneficial factors for DMEK include donor age above 50–60 years, absence of diabetes mellitus in the donor, and organ culture conditions. The individual structural features of Descemet membrane, for example its thickness, may also be a contributing factor (unpublished data).
DM is the specialized, 10- to 15-μm-thick basement membrane of the corneal endothelium consisting of collagens type IV, VIIII, and XVIII; glycoproteins; and proteoglycans. It is composed of a fetal anterior banded layer (ABL), about 3 μm in thickness, and a postnatal posterior nonbanded layer (PNBL), which gradually thickens with age, about 1 μm per decade, by continuous secretion by the endothelial cells. Owing to this lifelong appositional growth, the PNBL provides a historical record of endothelial function, and ultrastructural abnormalities in this layer reflect intrinsic disturbances in endothelial function.
In our previous studies investigating structural characteristics of DM, DM-stroma interface, and posterior corneal stroma, we have provided evidence of interindividual morphologic and biochemical variations of DM influencing graft preparation. We further showed that in a series of primary DMEK failures, the majority (8/14) of failed grafts revealed ultrastructural abnormalities indicative of a subclinical preoperative corneal endothelial dysfunction, which may have contributed to DMEK failure. Based on these observations, we hypothesize that DMEK grafts may display interindividual ultrastructural variations, and that abnormalities in ultrastructure indicating a preexisting endothelial dysfunction may affect the postoperative clinical outcome. Therefore, in a retrospective, single-center, nonrandomized consecutive series, 112 residual DM specimens obtained after DM stripping were examined by transmission electron microscopy, and the ultrastructural findings were correlated with the postoperative clinical parameters, for example, postoperative visual acuity, central corneal thickness, endothelial cell density, and graft detachment (rebubbling) rates.
Methods
The study complied with the tenets of the Declaration of Helsinki and adhered to all state laws of the country. The Institutional Review Board of the University of Erlangen-Nürnberg, Germany, waived the need for approval. Informed consent was obtained from the patients.
Tissue Specimens and Surgical Procedure
Corneal donor tissue (n = 112) was obtained from various eye banks in Europe and the United States. European donor corneas (86%) were organ-cultured for 385 ± 18 hours at 34 C in Dulbecco’s modified Eagle’s medium containing penicillin and streptomycin (Biochrom, Berlin, Germany) and fetal calf serum (Linaris, Bettingen am Main, Germany). The donor corneas obtained from US eye banks (14%) were short-term cultured at 4 C in Optisol-GS (Bausch & Lomb, Irvine, California, USA) for 216 ± 87 hours. Mean age of all donors was 72 ± 10 years, mean storage time was 363 ± 128 hours, and mean preoperative endothelial cell density was 2527 ± 216 cells/mm 2 .
DMEK procedures were performed in 112 eyes of 105 consecutive patients suffering from Fuchs endothelial corneal dystrophy (n = 92), primary DMEK failure (n = 7), primary failure after Descemet stripping automated endothelial keratoplasty (n = 2), pseudophakic bullous keratopathy (n = 2), failed penetrating keratoplasty (n = 1), and keratopathy in pseudoexfoliation syndrome (n = 1) between April 7, 2010 and September 29, 2011. Mean age of patients was 69 ± 9 years, 48% of patients were male and 52% female, and there were 57% right eyes and 43% left eyes. Immediately prior to transplantation, the endothelial cell–DM complex (EDM) was stripped from the donor corneal stroma as described previously in detail. Briefly, the corneoscleral buttons were mounted on a suction block (Hanna trephination system; Moria Instruments, Antony, France) and the endothelium was marked by gentle touch with an 8.0 mm trephine and stained with 0.06% trypan blue (Vision Blue; D.O.R.C. Deutschland GmbH, Berlin Germany) for 60 seconds. The submerged EDM peripheral to the mark was incised with a razor blade. The central edge of the EDM was lifted with a round knife (UltraSharp MVR knife; Alcon Grieshaber, Schaffhausen, Switzerland). The central EDM was stripped using 2 forceps and transferred into the patient’s eye. The remaining peripheral EDMs adjacent to the 8-mm graft edge were processed for transmission electron microscopy according to standard protocols.
Graft transplantation was performed as previously described in detail. Briefly, the recipient’s DM was removed from the stroma using an inverted hook (Price endothelial keratoplasty hook; Moria SA) in a central 9-mm area. The graft roll was injected into the anterior chamber and positioned centrally, and a small air bubble was injected into the lumen of the roll. The graft was spread out on the surface of the iris by increasing the air bubble and centered by variation of the air/fluid content. Then, the air bubble was removed and an air bubble injected between the graft and the iris to attach the graft at the stromal backside. Finally, the anterior chamber was filled with an air bubble. After 60 minutes, the air bubble was reduced to 50% of the anterior chamber volume. DMEK was combined with phacoemulsification and implantation of an intraocular lens (triple-DMEK) in 55 of 112 eyes (49%).
Clinical Evaluation
Clinical outcome parameters of patients analyzed in this study were best-corrected visual acuity (BCVA), central corneal thickness (CCT; Pentacam; Oculus, Wetzlar, Germany), and endothelial cell density (ECD; SeaEagle; Rhine-Tec GmbH, Krefeld, Germany). A previous analysis revealed no differences between measurements of the CCT with Pentacam Scheimpflug technique and ultrasound pachymetry (data not shown). The Pentacam data only are presented because these measurements are more reproducible.
The occurrence of graft detachments was assessed at the slit lamp and by the use of slit-lamp optical coherence tomography (Heidelberg Engineering, Heidelberg, Germany). Significant graft detachment requiring a repeated air injection was defined as detachment of more than 1 quadrant of the graft with a gap of more than 1 corneal thickness. Primary graft failure was defined as persisting corneal edema after surgery without initial clearance of the cornea within the first 3 months after surgery. Secondary graft failure was defined as corneal edema after initial clearance of the stroma.
Examination dates were on the day before DMEK and 1, 3, 6, and 12 months after surgery.
Transmission Electron Microscopy
The residual peripheral EDM complexes (n = 112) were fixed in 2.5% glutaraldehyde in 0.1 M phosphate buffer for 48 hours, postfixed in 2% buffered osmium tetroxide for 2 hours, dehydrated in graded alcohol concentrations, and embedded in epoxy resin according to standard protocols. Then, 1-μm semithin sections for orientation were stained with toluidine blue. Ultrathin sections were stained with uranyl acetate and lead citrate and examined with a transmission electron microscope (EM 906E; Carl Zeiss AG, Oberkochen, Germany).
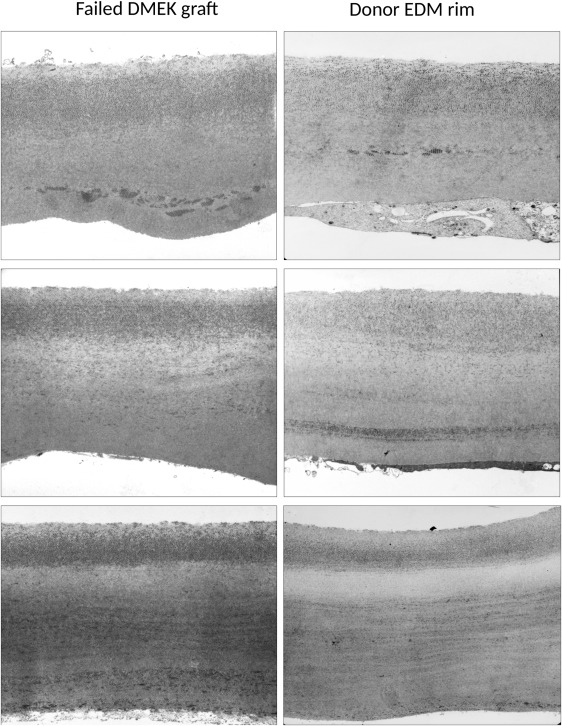
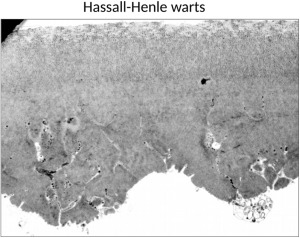
The specimens were examined and assessed independently by 2 examiners experienced in transmission electron microscopy (U.S.S., J.W.) regarding thickness and ultrastructural features (abnormal fibrillary inclusions, abnormal collagen deposits, etc) of DM. Three different levels of sectioning were examined per specimen. In order to demonstrate the reliability of the approach investigating the corneoscleral rims, we provided some examples showing comparable ultrastructural alterations in both peripheral rims and central DM specimens obtained from patients after re-DMEK ( Figure 1 ). Care was taken to avoid outer regions containing Hassall-Henle bodies at the periphery of the cornea ( Figure 2 ); these wart-like excrescences of the posterior surface of DM contain collagenous and fibrillary materials together with numerous fissures filled by endothelial cell processes, and are believed to reflect physiological aging processes of the cornea. According to the assessment of both examiners, the EDMs were classified as ultrastructurally normal or abnormal.
Statistical Analysis
IBM SPSS software version 20.0 (IBM, Armonk, New York, USA) was used for statistical analysis. Differences of samples between groups were assessed by Mann-Whitney U test. Categorical data were analyzed with χ 2 test and Fisher exact test, if the expected value of each group was less than 5. The significance level was set at P = .05.
Results
Ultrastructural Findings
Transmission electron microscopy was performed on 112 residual EDM specimens at 3 different levels of sectioning. Most of the EDM specimens analyzed (96/112; 86%) revealed a regular thickness of DM and a normal structure comprising a narrow interfacial matrix zone (∼1 μm), an anterior banded layer (∼2–3 μm), and an amorphous posterior nonbanded layer (9–11 μm) without any fibrillary inclusions ( Figure 3 ). The endothelial cell layer was frequently discontinuous owing to forceps manipulation of the peripheral EDM margins. Sixteen of 112 specimens (14%) revealed abnormalities in the ultrastructure of the DM. These comprised abnormal inclusions of collagen fibers representing long-spacing collagen (n = 11, 69%) and/or fine filaments, probably representing proteoglycans (n = 2, 13%), within the posterior nonbanded layer of DM, either in a lamellar ribbon-like or a rather diffuse pattern ( Figure 3 ). Another 2 of the 16 specimens (13%) revealed a fibrillar collagenous layer deposited posteriorly onto the nonbanded layer of the DM proper ( Figure 3 ). One specimen showed retrocorneal deposits of typical fibrillar pseudoexfoliative material indicative of pseudoexfoliation syndrome of the donor, and 1 specimen showed typical guttae formations indicative of Fuchs endothelial corneal dystrophy of the donor ( Figure 3 ). Mean thickness of all DM specimens was 15.8 ± 5.5 μm; there were no significant differences in DM thickness between the structurally normal (15.7 ± 5.5 μm) and the structurally abnormal (16.6 ± 5.5 μm) specimens ( P = .455).
The donor corneas with normal or abnormal DM did not show any significant differences regarding type of tissue culture (short-term/long-term culture: normal DM group, 14%/86%; abnormal DM group, 33%/66%, P = .164), mean age of donor (normal DM group, 73 ± 10 years; abnormal DM group, 73 ± 12 years, P = .916), preoperative endothelial cell density (normal DM group, 2529 ± 220/mm 2 ; abnormal DM group, 2514 ± 201/mm 2 , P = .781), or the mean storage time (normal DM group, 368 ± 125 hours; abnormal DM group, 339 ± 141 hours, P = .859).
Correlation of Clinical Outcomes After Descemet Membrane Endothelial Keratoplasty With Ultrastructural Findings
Clinical parameters were compared between eyes having received a structurally normal EDM graft (n = 96, normal DM group) or a structurally abnormal EDM graft (n = 16, abnormal DM group). There was no significant difference in the patients’ mean age at surgery (normal DM group, 69 ± 9 years; abnormal DM group, 72 ± 6 years, P = .407). Fifty of 96 eyes of the normal DM group and 5 of 16 eyes in the abnormal DM group underwent triple-DMEK ( P = .177).
Graft detachment requiring an additional air injection into the anterior chamber occurred in 42 of 96 eyes (44%) in the normal DM group and in 11 of 16 eyes (69%) in the abnormal DM group; this difference did not reach statistical significance ( P = .103).
Graft failure was noticed in 13 of 112 eyes and revealed a statistically significant difference between both groups: in the abnormal DM group graft failures occurred in 6 of 16 eyes (38%, secondary failure in 4 of 6 eyes), and in the normal DM group in 7 of 96 eyes (7%, secondary failure in 1 of 7 eyes) ( P = .003). Respecting only secondary graft failures, the difference was highly significant ( P = .001). In the group of normal DM eyes, the (only) secondary graft failure occurred after 9 months. In the group of abnormal DM eyes, the secondary graft failures occurred after 6, 9, 36, and 53 months.
Applying the Bonferroni correction for multiple statistical tests, the significance level for this study has to be set at .002 (29 single tests). At this level, the comparison of overall graft failure rates between the 2 groups is not statistically significant ( P = .003), but the comparison of secondary graft failures is still statistically significant ( P = .001).
Analyzing the last CCT value before regrafting, the mean CCT in the abnormal DM group was 850 μm (range 591–1267 μm), indicating endothelial failure with stromal edema.
Postoperative clinical results (BCVA, CCT, ECD) are shown in Table 1 for all eyes, eyes with abnormal DMs, and eyes with normal DMs. Regarding BCVA, a significant difference between both groups was found after 3 and 6 months postoperatively. However, there was a significant difference in BCVA already before DMEK surgery. Therefore, the change of BCVA was analyzed and a significant difference between both groups was found between the preoperative BCVA and BCVA 1 month after DMEK. In the abnormal DM group (starting with a worse BCVA), the increase of BCVA was higher (−0.72 ± 0.61) than in the normal DM group (−0.24 ± 0.53, P = .019).
| Group | All Patients (n = 112) | Normal DM Group (n = 96) | Abnormal DM Group (n = 16) | P Value a |
|---|---|---|---|---|
| CCT preoperative | 689 ± 129 μm | 681 ± 118 μm | 723 ± 173 μm | .530 |
| CCT 1 month | 559 ± 109 μm | 556 ± 111 μm | 572 ± 102 μm | .509 |
| CCT 3 months | 554 ± 134 μm | 548 ± 111 μm | 585 ± 219 μm | .836 |
| CCT 6 months | 548 ± 92 μm | 540 ± 64 μm | 602 ± 201 μm | .865 |
| CCT 12 months | 542 ± 85 μm | 546 ± 90 μm | 517 ± 33 μm | .266 |
| ECD preoperative | 2526 ± 216/mm 2 | 2529 ± 220/mm 2 | 2514 ± 201 | .781 |
| ECD 1 month | 1544 ± 309/mm 2 | 1550 ± 319/mm 2 | 1516 ± 259 | .652 |
| ECD 3 months | 1514 ± 315/mm 2 | 1531 ± 331/mm 2 | 1431 ± 208 | .246 |
| ECD 6 months | 1532 ± 321/mm 2 | 1532 ± 332/mm 2 | 1534 ± 239 | .792 |
| ECD 12 months | 1480 ± 292/mm 2 | 1479 ± 292/mm 2 | 1484 ± 308 | .491 |
| BCVA preoperative | 0.73 ± 0.48 | 0.65 ± 0.42 | 1.2 ± 0.65 | .002* |
| BCVA 1 month | 0.40 ± 0.46 | 0.37 ± 0.47 | 0.54 ± 0.44 | .085 |
| BCVA 3 months | 0.37 ± 0.43 | 0.32 ± 0.38 | 0.64 ± 0.60 | .012* |
| BCVA 6 months | 0.28 ± 0.35 | 1.21 ± 0.20 | 0.73 ± 0.69 | .007* |
| BCVA 12 months | 0.21 ± 0.30 | 0.21 ± 0.31 | 0.26 ± 0.17 | .136 |
| Change of BCVA preoperative – 1 month | −0.32 ± 0.56 | −0.24 ± 0.53 | −0.72 ± 0.61 | .019* |
| Change of BCVA 1 to 3 months | −0.02 ± 0.35 | −0.05 ± 0.36 | 0.14 ± 0.3 | .182 |
| Change of BCVA 3 to 6 months | −0.01 ± 0.15 | −0.02 ± 0.1 | 0.08 ± 0.31 | .782 |
| Change of BCVA 6 to 12 months | −0.02 ± 0.2 | −0.01 ± 0.14 | −0.15 ± 0.47 | .778 |
No significant differences were found between groups regarding CCT and ECD before DMEK and 1, 3, 6, and 12 months after DMEK, respectively.
Subgroup analyses for the organ-cultured grafts and for eyes with Fuchs endothelial corneal dystrophy are shown in Tables 2 and 3 , respectively.



