Fig. 19.1
Classification scheme for elevated IOP in uveitis based on underlying mechanism and timing of IOP rise with respect to the onset of inflammation (Modified from Zelefsky and Cunningham [27]. With permission from American Academy of Ophthalmology)
1.
Inflammatory ocular hypertension syndromes (IOHS)
2.
Acute uveitic angle closure
3.
Corticosteroid-induced ocular hypertension/glaucoma
4.
Chronic mixed-mechanism ocular hypertension/glaucoma
Inflammatory Ocular Hypertension Syndromes (IOHS)
Elevated IOP in patients with IOHS is believed to result from direct inflammation of the trabecular meshwork, or trabeculitis. The IOP elevation in these syndromes parallels the course of the inflammation. The most common condition causing IOHS is herpetic anterior uveitis (HSV, VZV, and CMV); other well-recognized causes of IOHS include sarcoidosis, toxoplasmosis, syphilis, and Posner-Schlossman syndrome [2, 3].
Herpetic Anterior Uveitis
Herpetic anterior uveitis can be caused by herpes simplex virus (HSV), varicella zoster virus (VZV), or cytomegalovirus (CMV). Corneal epithelial or stromal involvement is common in HSV- and VZV-associated uveitis and should be looked for to support the diagnosis. Iris stromal atrophy, either patchy or sectoral (see Fig. 19.2), is caused by virus-induced ischemia and is a unique finding that differentiates herpetic anterior uveitis from other causes of IOHS, but may not be present in all cases.
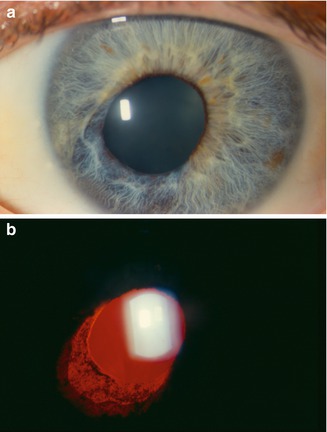

Fig. 19.2
A patient with herpes zoster keratouveitis. Corectopia (a) is present with sectoral iris atrophy and corresponding transillumination defect (b). Iris atrophy can be observed in the setting of both herpes simplex and herpes zoster infection (Reprinted from Zelefsky and Cunningham [27]. With permission from American Academy of Ophthalmology)
Ocular herpes simplex infection is typically due to the type 1 virus and most frequently presents as epithelial or stromal keratitis and keratouveitis. Up to one-third of patients with herpes simplex uveitis develop IOHS. Varicella zoster virus infection usually occurs during childhood; the virus remains dormant and reactivates within specific dermatomes with advancing age or due to systemic immunosuppression. VZV infection most commonly manifests as dermatitis, and when the trigeminal nerve is involved (herpes zoster ophthalmicus, HZO), up to two-thirds of patients can develop ocular involvement. Hutchinson’s sign is the presence of herpes zoster skin lesions at the tip, side, or root of the nose (along the distribution of the nasociliary nerve) and is a strong predictor of ocular involvement. Like HSV infection, the most common ocular manifestations with VZV are keratitis and keratouveitis. IOHS is more common with VZV uveitis, affecting nearly 50 % of affected patients. Decreased corneal sensation and sectoral atrophy can be present in both HSV and VZV infections. Cytomegalovirus-associated anterior uveitis is a less common cause of IOHS, and unlike typical CMV infection that occurs in immunocompromised individuals, CMV-associated IOHS [4] occurs in otherwise healthy patients with a chronic, recurrent pattern of inflammation that responds to specific anti-CMV therapy.
Posner-Schlossman Syndrome
Also known as glaucomatocyclitic crisis, this syndrome is characterized by unilateral, episodic and acute, severe elevation of IOP. The inflammation seen in this syndrome is characteristically mild and appears out of proportion to the rise in IOP. A number of authors have reported herpes virus DNA in patients diagnosed clinically as having Posner-Schlossman syndrome, suggesting that at least some patients with this condition have what might be considered form fruste herpetic anterior uveitis. Patients are generally young and present with blurred vision and minimal or no pain. On examination, circumcorneal flush, corneal edema, and mild iridocyclitis are evident with small to midsized keratic precipitates and trace cell and flare in the anterior chamber. The IOP elevation reaches levels in the range of 40–60 mmHg and is disproportionate to the level of inflammation. Gonioscopy usually reveals no abnormalities and most patients have normal optic nerves. Between attacks, the IOP is normal and characteristically there are no signs suggestive of prior inflammation such as posterior synechiae or peripheral anterior synechiae. Posner-Schlossman syndrome is usually a self-limiting condition, although some cases with advanced glaucomatous optic nerve damage have been reported [5]. The prognosis is relatively good and treatment is usually required only during attacks.
IOHS may be confused with clinical entities in which noninflammatory material blocks the trabecular pores, causing a rise in IOP. Thus, lens material in phacolytic glaucoma, red blood cells in microhyphema, and photoreceptor outer segments in Schwartz-Matsuo syndrome may all mimic inflammatory cells and result in misdiagnosis as IOHS.
The importance of identifying IOHS lies in the fact that most causes of this rather unique presentation are infectious in nature. Once IOHS is accurately diagnosed, an appropriate work-up to identify and treat the causative microbial agent can be initiated. In general, comanagement of these cases with a specialist in ocular inflammation is desirable.
Acute Uveitic Angle Closure
In this category, elevation in IOP occurs with or soon after the onset of inflammation but may take longer to resolve than the inflammation. Acute uveitic angle closure can be caused by two mechanisms:
1.
Pupillary block: Seclusion of the pupil by extensive posterior synechiae or an inflammatory pupillary membrane can result in iris bombe and angle closure (see Fig. 19.3). This mechanism can occur even in patients with open angles prior to the onset of uveitis.
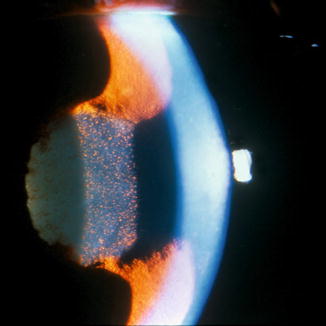

Fig. 19.3
Iris bombe in a patient with seclusion following a severe episode of HLA-B27-associated anterior uveitis (Reprinted from Zelefsky and Cunningham [27]. With permission from American Academy of Ophthalmology)
2.
Non-pupillary block: A forward movement of the lens-iris diaphragm due to ciliochoroidal effusion can also result in angle closure by a “pushing” mechanism. Iris bombe is not seen with this mechanism.
Detailed slit-lamp examination can usually help differentiate the two mechanisms; however, in some cases imaging of the posterior chamber with ultrasound biomicroscopy may be required. Identifying the mechanism of angle closure in this category is important because treatment differs significantly between the two. In pupillary-block angle closure, the recommended treatment is a laser peripheral iridotomy, whereas in non-pupillary-block angle closure due to a ciliochoroidal effusion and cycloplegia, systemic corticosteroids are the mainstay of treatment. Understanding the mechanism of angle closure also helps in establishing the diagnosis as ciliochoroidal effusions are more common in certain uveitic conditions such as Vogt-Koyanagi-Harada disease, scleritis, and sympathetic ophthalmia.
Corticosteroid-Induced Ocular Hypertension/Glaucoma
Corticosteroid-induced IOP elevation tends to occur days to weeks after the onset of uveitis and initiation of treatment with corticosteroids. The mechanism is believed to be related to a reduction in aqueous outflow mediated by wide-ranging effects on the trabecular meshwork at the cellular and biochemical levels. All routes of corticosteroid administration can be associated with elevated IOP, including topical, periocular intraocular, systemic, and intranasal (see Sidebar 19.2).
Chronic Mixed-Mechanism Ocular Hypertension/Glaucoma
Many patients with uveitis develop ocular hypertension months to years after the onset of inflammation for reasons unrelated to IOHS, acute angle closure, or corticosteroids. Mechanisms of IOP elevation in this group of patients can be classified as follows:
1.
Chronic closed angle: Angle closure by the “pulling” mechanism occurs via formation of peripheral anterior synechiae or of vascular membranes such as in Fuchs heterochromic iridocyclitis and other forms of chronic uveitis.
2.
Open angle: In spite of an open angle, there is increased outflow resistance due to chronic damage to the trabecular meshwork.
Juvenile idiopathic arthritis, sarcoidosis, and Fuchs heterochromic iridocyclitis are commonly associated with chronic mixed-mechanism IOP elevation. In Fuchs iridocyclitis, there is chronic, usually unilateral, low-grade iridocyclitis characterized by diffusely distributed stellate keratic precipitates and iris heterochromia (see Fig. 19.4), often accompanied by whitish vitreous cellular infiltrates. Unlike many types of uveitis, the inflammation in this entity is often asymptomatic, and posterior synechiae are uncommon despite the presence of persistent inflammation. Nearly all patients develop posterior subcapsular cataract and up to 60 % develop secondary glaucoma.
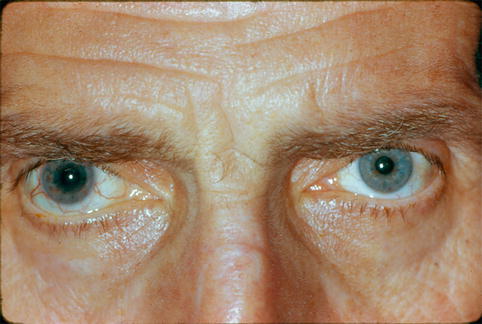

Fig. 19.4
Iris heterochromia in a patient with Fuchs heterochromic iridocyclitis. Note that the affected right eye is darker in appearance in this patient with lightly pigmented irides, whereas the affected iris in a patient with darkly pigmented irides is typically lighter in appearance (Reprinted from Zelefsky and Cunningham [27]. With permission from American Academy of Ophthalmology)
In sarcoidosis-related anterior uveitis, the incidence of glaucoma ranges from 11 to 26 %. The inflammation in sarcoidosis tends to be “fibrin rich” with the formation of iris nodules (see Fig. 19.5) and keratic precipitates on the trabecular meshwork, resulting in the development of posterior and anterior peripheral synechiae, respectively. The uveitis also tends to be chronic with morphological changes in the angle being common in the later stages.
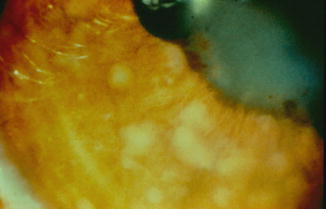

Fig. 19.5
Busacca nodules on the iris in a patient with sarcoidosis (Reprinted from Cunningham [28]. With permission from American Academy of Ophthalmology)
Managing Elevated IOP in Patients with Uveitis
General Principles
Comanagement with a uveitis specialist is ideal when feasible. In general, the underlying inflammation must be treated as necessary. Undertreating uveitis with corticosteroids in order to minimize IOP elevation is a shortsighted strategy because the long-term risks of chronic inflammation far outweigh the benefits of short-term IOP control. Treatment of IOP elevation must be undertaken with the optic nerve status in mind. In many patients with uveitis, the optic disc is not severely damaged, and the target IOP does not need to be very low. The timing of IOP elevation can also help in management – a patient with acute rises in IOP observed only during episodes of inflammation can probably be managed adequately with medical therapy alone, whereas a patient with chronic mixed-mechanism glaucoma is more likely to require surgical intervention. In patients who develop ocular hypertension in response to corticosteroids, alternative therapy with weaker corticosteroids or immunosuppressive therapy may be considered, although it should be remembered that corticosteroids that are less likely to raise IOP are also less likely to control moderate to severe inflammation. The use of long-acting depot corticosteroid preparations in these patients must be undertaken with particular caution and preferably only after a sustained trial of either topical or systemic corticosteroid therapy has shown that the risk of corticosteroid-induced ocular hypertension is low.
The Therapeutic Armamentarium
Treatment options available in the management of uveitic glaucoma are as follows:
Medications
Topical hypotensive therapy for raised IOP in uveitis is similar to treatment for POAG with a few caveats. Prostaglandin analogs, which have replaced beta-blockers as the first line of therapy for POAG in many practices, must be used with caution in patients with uveitis because they may have a pro-inflammatory effect. Although no clear cause-effect relationship has been demonstrated, a number of case reports have suggested that these drugs may exacerbate preexisting iridocyclitis, promote recurrences of herpetic keratitis or keratouveitis, and promote the formation of cystoid macular edema [6, 7]. It seems most prudent, therefore, that these agents be used as the second- or third-line therapy in patients with uveitis and that when they are used, the treating physician remains mindful of the possibility for inflammatory side effects. It must be noted, however, that prostaglandin analogs are not contraindicated in uveitic ocular hypertension/glaucoma and that they can be very useful in many patients, especially when controlling the IOP would otherwise require surgery. Brimonidine is another IOP-lowering medication that has been associated with granulomatous uveitis in several case reports [8, 9]; the mechanism has been postulated to be toxic or immunologically mediated. Medications that are definitely pro-inflammatory and must be avoided in patients with active uveitis include pilocarpine, epinephrine, and epinephrine prodrugs. For these reasons, beta-blockers are usually the first line of management for raised IOP in uveitis, followed by carbonic anhydrase inhibitors (topical and/or systemic) and alpha agonists.
Laser Procedures
Laser peripheral iridotomy is the treatment of choice for acute angle closure due to pupillary block. The inflammatory response following laser iridotomy in patients with uveitis tends to be more severe and prolonged than in patients with primary angle closure; hence, the need for aggressive treatment with corticosteroids must be anticipated. In addition, there is a higher rate of closure of the iridotomy [10], and hence, performing at least two iridotomies is recommended.
Laser trabeculoplasty: Laser trabeculoplasty does not play a significant role in the management of elevated IOP related to uveitis.
Cyclophotocoagulation must be used with extreme caution in uveitic glaucoma for two reasons. Firstly, the procedure can exacerbate inflammation, and, secondly, there is a greater risk of atrophy in a ciliary body that has already sustained damage from recurrent inflammation; this can eventually lead to hypotony and/or phthisis.
Surgery
The decision to perform surgery depends on the IOP, the degree of nerve damage, and response as well as tolerance to medical therapy. It is beneficial to have the inflammation controlled prior to surgery, but in many patients with uveitis-associated increase in IOP, surgery is performed on an emergent rather than elective basis. These patients are for the most part already on some form of corticosteroid therapy, and it is worthwhile to consider the use of systemic, periocular, and/or intraocular corticosteroids in the perioperative period. Many uveitis specialists recommend IV Solu-Medrol 250–1,000 mg given intraoperatively followed by a quickly tapered dose of oral prednisone, 0.5–1.5 mg/kg/day, in the postoperative period.
Both trabeculectomy with antimetabolites and aqueous drainage devices have been successfully used in the management of uveitic glaucoma. The choice depends on the likelihood of failure of trabeculectomy and surgeon preference. Active uveitis, inflammation that is difficult to control, younger age, and history of prior trabeculectomy are all associated with high risk for failure of filtering surgery, and an aqueous drainage device is preferable in these patients. Patients with uveitis have a greater risk of developing hypotony following either type of surgery, and modifications to the surgical technique are performed to safeguard against it. In recent years, intravitreal drug delivery devices, such as the Retisert implant [11], are being used with increasing frequency in patients with uveitis. It is important to communicate with the surgeon that the inferotemporal quadrant may be the best position for the placement of such sustained release devices so that a superior trabeculectomy or placement of a drainage device in the superotemporal or inferonasal quadrants may be performed in the future as indicated.
Trabeculectomy
Trabeculectomy without antifibrosis agents has a relatively low (30–50 %) chance of survival in patients with uveitis [12, 13]; therefore, the use of either 5-fluorouracil (5-FU) or mitomycin-C is recommended. Success rates of 75–90 % with mitomycin-C [14, 15] and 50–90 % with 5-FU [13, 16–18] have been reported depending on the definition of success, lens status, and level of risk for failure. There is no direct comparative study to support the use of one agent over the other, and the choice depends mainly on surgeon preference. Many surgeons also use 5-FU injections postoperatively.
Uveitic eyes are particularly at risk for postoperative hypotony; therefore, the scleral flap must be sutured tightly. If hypotony does develop and is accompanied by shallowing of the anterior chamber, cycloplegia can help move the lens-iris diaphragm posteriorly. The frequency of postoperative corticosteroid therapy is determined based on the balance between the need to control active inflammation and the need to accelerate healing (thereby decreasing overfiltration). Topical corticosteroids are generally used for 6–8 weeks postoperatively and may need to be tapered slowly.
Aqueous Drainage Devices
In recent years, the indications for aqueous drainage implantation have expanded considerably. For many surgeons, aqueous drainage devices are the procedure of choice in uveitic glaucoma. Both the valved Ahmed and the non-valved Baerveldt implant have been used for IOP control in such patients. When using the Baerveldt implant, the choice between the 250 and the 350 mm2 device should theoretically be based on the balance between the opposing risks of hypotony and postoperative encapsulation. In practice, however, the 350 mm2 device tends to be used most commonly. There are a relatively small number of studies that have reported outcomes of aqueous drainage device implantation specifically in uveitic patients. Of these, the longest follow-up with Ahmed implants was reported by Papadaki and associates [19], and the largest series of uveitic patients with Baerveldt implants was reported by Ceballos and associates [20].
Success rates in the study by Papadaki and associates were 77 and 57 % at 1 and 4 years, respectively, with the definition of success being IOP of 5–21 mmHg plus 25 % lowering from preoperative levels. At 4 years follow-up, 74 % of patients required topical medication for IOP control. The overall complication rate was 12 % per person-years with corneal edema or graft failure being the most common (3.2 % per person-years). The occurrence of severe hypotony defined as flat anterior chamber, choroidal effusion, hypotony maculopathy, or any combination thereof was1.2 % per person-years. In the study by Ceballos and colleagues [20], the success rate was 92 % at 2 years with the definition of success being IOP of 5–21 mmHg without need for further glaucoma surgery, loss of light perception, or phthisis. The most common complications were choroidal effusions (4 of 24 eyes), hypotony, and cystoid macular edema (3 of 24 eyes for both).
Other Surgical Procedures
Goniotomy [21], viscocanalostomy [22], and trabeculodialysis [23, 24] have all been reported for the treatment of uveitic glaucoma with success rates varying from 50 to 71 %. Trabeculodialysis is a modified goniotomy procedure in which the trabecular meshwork is further released via wing incisions to maximize direct aqueous access into Schlemm’s canal. In our experience, this procedure is especially useful in children with uveitis and elevated IOP. A newer device, called the Trabectome [25], has recently been developed for ab interno trabeculectomy. Although there is no data yet on its efficacy in patients with uveitic glaucoma, it may be an attractive option in patients with well-controlled inflammation and mild optic nerve damage.
Specific Treatment Strategies
Management can be tailored to the individual patient based on the mechanism of IOP elevation:
Management of IOHS
A complete history and directed uveitis work-up should be performed in all patients with uveitis, regardless of whether the IOP is elevated, low, or normal (see Sidebar 19.1). In patients with IOHS and no clear evidence of herpetic infection, most would recommend testing for sarcoidosis with a chest X-ray and serum angiotensin-converting enzyme (ACE) or lysozyme level and for syphilis with an FTA-ABS and a VDRL or an RPR. The fundus should also be examined carefully for any evidence of toxoplasmic retinochoroiditis. Patients with Posner-Schlossman syndrome can be especially challenging to diagnose as the inflammatory response can be very subtle and the eye examination is typically normal between attacks. The IOP elevation in patients with IOHS typically reverses with treatment of the underlying inflammatory/infectious condition with topical corticosteroids and/or antimicrobial therapy as indicated. Herpetic anterior uveitis should be treated with oral antiviral agents. Acyclovir 400 mg 5 times daily is typically used for HSV. For VZV, acyclovir 800 mg 5 times daily, famciclovir 500 mg 3 times daily, or valacyclovir 1,000 mg 3 times daily may be used. In patients with HZO, antiviral treatment should be administered within 72 h of the onset of the rash. Patients with herpetic keratouveitis can develop a chronic relapsing-remitting course and may eventually require surgical intervention.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


