1. The patient has glaucoma
2. The patient may have glaucoma
3. The patient will develop visual field loss
4. The physician is fearful of being sued
5. The patient is more likely to return for a checkup if treated
6. The patient wants to be treated
7. The physician wants to receive a fee
8. The patient will develop additional field loss
9. Treatment will prevent the patient from getting worse
Table 5.2
Speculations predicting the rate of change in glaucomaa
Amount of existing nerve damage | |||
|---|---|---|---|
Level of IOPb (mmHg) | None | Mild/moderate | Advanced |
100 | 1 minute | 1 minute | 1 minute |
50 | 4 weeks | 2 weeks | 1 week |
35 | 12 months | 6 months | 1 month |
15 | 20 years | 10 years | 5 years |
1.
“I treated the patient because she has glaucoma.” Many patients with glaucoma will never develop any disability whatsoever from their glaucoma but will immediately develop a decrease in quality of life and some disability from the treatment.
2.
“I treated the patient because he may have glaucoma and I would have felt terrible if he got worse.” This is an even less justifiable reason than #1.
3.
“The patient had risk factors indicating that she would develop visual field loss.” How long would it take that field loss to develop? Would the field loss be sufficient to cause symptoms? If not, how can one justify treatment?
4.
“The patient was one of those people who is likely to sue, and therefore I started treatment.” Physicians have been found guilty of malpractice for treating unnecessarily as well as for not treating. The best defenses against suit are to hold out realistic expectations to oneself and to the patient and to provide good care that is appropriate for the patient.
5.
“It has been shown that patients who are treated are more likely to return for their visit.” Indeed, Bigger [8] did show that patients who are treated are more likely to return. However, that in itself is not a justification for treatment. It takes time to explain to patients why they need to return, but a thoughtful discussion is probably more likely to result in developing a good doctor–patient relationship than simply starting the patient on treatment. Thoughtful patients will wonder whether treatment was really justified and may elect another physician who seems more thorough.
6.
“But the patient wanted to be treated.” Few patients, of course, want to get worse. Therefore, many patients, as is true for many doctors, take a default position of believing that treatment is preferable to no treatment. They forget that treatments often cause problems that are greater than those the treatment is designed to prevent or alleviate. It is understandable that patients may prefer to be treated. However, when it is explained to patients that a treatment has a greater likelihood of causing problems than deferring a treatment, most patients choose not to be treated. There may be, however, some patients who are so strongly fearful of getting worse that it is appropriate to use a treatment, indicating to the patient that the need for the treatment will be carefully reconsidered in the future and perhaps stopped.
7.
Though rarely consciously articulated, all caregivers know that treatments generate fees. It is reasonable for all caregivers, including physicians, to expect some reimbursement for their services. However, the reimbursement should be related to the value of the service. Moreover, it is not proper to treat because one gets reimbursed, but rather because the treatment is appropriate, and in doing the appropriate thing, one deserves to get reimbursed.
8.
“But the patient already has visual field loss and may develop more field loss.” This is starting to sound reasonable. When patients already have definite glaucomatous visual field loss, they are at risk for developing more visual field loss, and in such a situation treatment is likely to be justified, but not always.
9.
“Treatment will prevent the patient from getting worse.” Aha! Here is the only justifiable reason for treating.
The reason that trumps all other reasons regarding treatment, specifically, the only justifiable reason for treatment, is a reasonable belief that treatment will prevent the patient from getting worse. Preventing visual field loss is not in itself a justifiable reason for treatment. Improving visual acuity is in itself not a justifiable reason for treatment. Lowering intraocular pressure by itself is certainly not a justifiable reason to treat (unless it is at a level certain to make the person worse) nor is prevention of a development of glaucomatous cupping, that is, worsening of the optic neuropathy that has, unfortunately, become the sine qua non of the diagnosis of glaucoma. It is well established that patients can have marked narrowing of the neuroretinal rim of the optic nerve head without developing any detectible visual loss. Lowering intraocular pressure is only of value if it relieves pain, prevents a vascular occlusion, or has some other direct beneficial effect on the person’s quality of life. Even if the risk attendant to improving visual acuity is minimal, it is still unjustifiably great if the person is already entirely satisfied with his or her vision. As far as visual field loss is concerned, mild field loss rarely is associated with any symptomatology, and even moderate bilateral visual field loss may not cause any disability. All of these characteristics of glaucoma are important and need to be monitored, because they are helpful in establishing a rate of change, but they all too frequently become inadvertent surrogates for the person himself or herself.
Predictors of the Future
Accurate predictors come primarily in two forms: (1) a single marker that is almost always a sign of things to come [9, 10] or (2) a trend that will almost always continue [11]. “Almost” is present in this sentence, because accurate prediction of a future assumes that there is a future. For example, an intraocular pressure (IOP) of 100 mmHg will almost always cause optic nerve damage; the “almost” is added because an IOP of 100 mmHg probably does not cause damage when it is momentary, as it is when using digital massage following a filtering procedure or as it is in some types of refractive surgery or when the person will die 30 s after the pressure becomes 100 mmHg. The “almost” is there because it is usually necessary to consider the duration of the causative force. More on that later. Single markers that are highly predictive of making the person worse as a result of glaucoma then must be combined with information about rate of change and duration of change. For some situations, such as an IOP greater than 100 mmHg, the rate of change is usually a minute or even seconds, so a duration of, say, 5 min is of concern. Table 5.2 gives rough guidelines for IOP-producing damage that takes into account the amount of damage already present as well as the duration of time involved to cause the damage. The estimates shown in Table 5.2 have not been established, but they are conceptual guidelines of some importance. Because accurate prediction of the future is important in decision-making, there has long been an interest in so-called risk factors, that is, findings that increase the risk of change occurring.
Standard risk factors for developing visual field loss in glaucoma are listed in Table 5.3. It should be noted that these are not factors for developing disability, but rather for developing field loss. Except for acquired pit of optic nerve or rim narrowing, all of these factors are relative. That is, their significance is relative to their severity. Though age is frequently listed as a risk factor, age in itself may not be a valid risk factor for the development of glaucomatous visual field loss. As persons age, they lose visual field due to lost neurons; that is, the process of aging is itself associated with progressive visual field loss. This loss tends to be global; it does not appear to follow the pattern of glaucomatous visual field loss (limited initially to one hemifield, a nasal step, an inferior paracentral scotoma, an arcuate pattern, etc.), though this needs better investigation. Because glaucoma involves loss of neurons and aging involves loss of neurons, aging probably contributes to the process of glaucomatous nerve loss. But it is far less clear that age itself plays a role. Mean values can be misleading in this regard. For example, Caprioli’s group showed that age was highly significantly related to further visual field loss, but, when considering the range of the ages, the oldest patients did not have any field loss [11].
Table 5.3
Risk factors for developing or worsening of glaucomatous visual field loss
Finding | Significance |
|---|---|
Acquired pit of the optic nerve | Always |
Narrowing of the neuroretinal rim in excess of that occurring related to aging | Almost always |
Aging | May not be a predictive factor, but is related to loss of nerve cells |
Intraocular pressure | The higher the IOP, the greater the predictive value |
Family history | The more positive, the greater the predictive value |
Myopia | The more myopic, the greater the predictive value |
Disc hemorrhage | Not definitely an independent factor for worsening |
Age | May be merely associated |
Pigment dispersion syndrome | Probably not an independent risk factor other than its relationship to intraocular pressure and myopia |
Exfoliation syndrome | Probably not an independent risk factor except for its relationship to intraocular pressure, but this is not certain |
The most reliable predictor of the future often is a trend that is likely to continue. It is for this reason that patients with glaucoma and without known glaucoma need to be followed, in order to permit the accumulation of enough data points to allow establishing a reliable trend. The number of such data points that are required relates to the accuracy of the data and the rapidity of change. Because changes in the optic disc and retinal nerve fiber layer usually precede changes in the visual field [12, 13] and because changes in the disc and nerve fiber layer can be obtained relatively objectively, it would be ideal to record this information for every person around the age of 15 to serve as a baseline measurement for future comparisons. Once visual field loss is present, the more frequently visual field examinations are repeated, and the less noisy those examinations are, the more quickly a valid trend can be established. A minimum of two visual fields per year is probably a rough guideline for most patients with glaucoma, but more frequent fields will establish a trend more rapidly [14]. Any reproducible change in the width of the neuroretinal rim within a period of 5 years is probably clinically significant, and a change of 2 dB per year of mean defect is also probably indicative of change that exceeds that occurring normally.
Colored Glaucoma Graph
Figure 5.1 shows a graphic way of considering the issues just discussed from a clinical point of view [15, 16]. This “colored glaucoma graph” is used by us every day in our clinical practice. The vertical axis concerns units of glaucoma damage and the horizontal axis units of time (duration). The surest way to predict the future, then, is to extrapolate from valid observations of what has already happened.
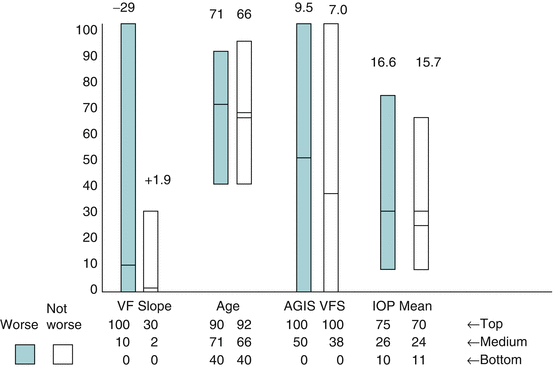

Fig. 5.1
The darker blue bars represent patients who showed progressive visual field loss in the Advanced Glaucoma Intervention Study. The Y axis has arbitrary units designed to include the majority of the range of values. The figures on top of the bars indicate the actual numbers. For the left-hand column, this represents the slope of the visual field, the left-hand bar indicating a rapidly deteriorating slope and the right-hand bar – one which was slightly positive. The mean values are indicated by the lines crossing the eight bars. The highest value of the range is indicated by the figures entitled, “Top.” Thus, when considering age, the mean age of patients getting worse is 71, and the mean age of patients not getting worse is 66. The oldest patient getting worse is 90, and the oldest patient not getting worse is 92. VF visual field; AGIS Advanced Glaucoma Intervention Study; VFS visual field score; IOP intraocular pressure (Based on data from Ref. 10)
Because what the physician and the patient want to know relates to what is going to happen to the patient’s ability to perform the activities of daily living (ADL), plotting a reliable course of change in ADL would in some ways be ideal. However, by the time such changes have occurred, the amount of glaucoma damage is already either moderate or marked, and neither the doctor nor the patient wants to await further change in ability to perform ADL before deciding whether treatment is necessary. Early visual field changes precede loss of functional ability. Also, early visual field changes are often variable and may be seriously misleading. Once definite loss of field is present, and especially in patients in whom the testing results are highly reproducible, then serial visual fields can be of great value. Were it possible to count retinal ganglion cells, that might be the ideal unit to place on the “Y” axis of the patient’s clinical course, that is, a measurement of the deterioration in the patient’s stage. Though this is still not possible today, it probably will be in time. That will be a great step ahead! Estimates of nerve fiber layer thickness are a surrogate for retinal ganglion cells, but may not yet be sufficiently reliable or reproducible measures of number of ganglion cells. Furthermore, it seems likely that in many cases a structural change in the topography of the optic nerve is the initiating event, causing the damage to the neurons and thus preceding change in nerve fiber layer thickness or, for that matter, ganglion cell loss. It is possible that in some patients ganglion cell damage precedes optic nerve topography change, but this has not been established and seems unlikely. Some measure, however, of perimetric damage is appropriate, and the more valid and quantitative that is, the better.
Except for the extraordinarily rare situation in which a child is born with glaucomatous nerve damage, nobody starts with glaucoma damage. That is, glaucoma damage is acquired. This gives the physician and the patient the opportunity to detect the damage in its early stages and plot changes prior to the time that sufficient functional damage has occurred that the glaucomatous process can be established with certainty as being present. Obtaining two, or preferably three, reliable evaluations in which the separation between the points exceeds the noise and biological variability of the points allows establishing points that can be used to estimate the future results of accuracy. Hitchings and others believe the course of glaucoma occurs in a linear pattern [17]. However, it is probably wise to recognize the limits of an extrapolated projection and use each new point to refine the previously defined curve, recognizing the variability of the point caused by noise and short-term fluctuations. In actual practice, one is looking for a trend that will indicate whether the condition is roughly stable or significantly deteriorating. As shown by Caprioli, the rate of change is an excellent prognostic indicator, far better than amount of field loss, level of intraocular pressure, or fluctuation of intraocular pressure [11] (Fig. 5.2).
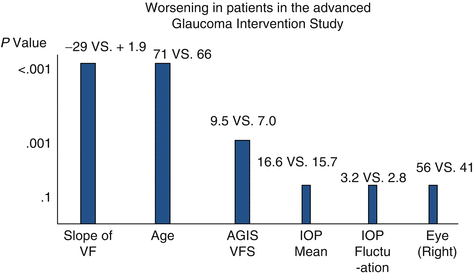

Fig. 5.2
Likelihood that characteristics of patients in the AGIS study were related to worsening of the visual field [Based on data from Ref. 10]
The course of glaucoma can be roughly divided into three: (1) early or questionable tissue damage, (2) definite but still asymptomatic damage, and (3) sufficient visual loss to cause disability. The initial stages of glaucoma, then, are in the period of time when changes cannot at a single examination be determined to be signs of pathology or variations of normal. While cup/disc ratios can be helpful in detecting change, they are of almost no help in establishing the presence or absence of glaucoma, unless the cup/disc ratio is greater than 0.8. This is because the cup/disc ratio system does not take into account the position of the cup [18] or the size of the disc [19]. A cup/disc ratio of 0.5 with a concentrically placed cup is not likely to be a sign of glaucoma, but a cup/disc ratio of 0.5 that is eccentrically placed, so that the rim is absent in one area, is a highly reliable sign of glaucomatous damage (Fig. 5.3). A cup/disc ratio of 0.5 in a large disc with a vertical diameter of 3 mm is not likely to be a sign of pathology, whereas a cup/disc ratio of 0.5 in a small disc of 1 mm vertical diameter is a disturbing finding (Fig. 5.4). The Heidelberg Retinal Tomography Moorfields Regression Analysis can provide help, but it takes neither pattern nor disc size into sufficient account to be a highly reliable diagnostic tool. In contrast, the Disc Damage Likelihood Scale (Fig. 5.5) takes into account the width of the neuroretinal rim and the size of the disc and correlates better the visual field loss than the Heidelberg Retinal Tomography parameters [15, 20–24]. It can, then, serve as a useful guide to the “Y” axis of any graph depicting the course of glaucoma.