Purpose
To investigate the prevalence and clinical relevance of incomplete posterior vitreous detachment (PVD).
Design
Prospective, observational cohort study.
Methods
setting: Institutional. patients: Consecutive patients without previous ocular history who were diagnosed with acute uncomplicated PVD. observations: Baseline kinetic ultrasound evaluation differentiated posterior vitreous separation as complete or incomplete. Prospective follow-up searched for complications related to PVD. Multivariate analysis evaluated associations of baseline demographic and clinical characteristics to incomplete PVD. A Kaplan-Meier analysis evaluated the probability and its standard error of experiencing an adverse outcome. The log-rank test determined whether incomplete PVD modifies the natural history of PVD. main outcome measures: Prevalence of incomplete PVD and the estimated incidence of late adverse outcomes such as new retinal tears, epimacular membranes, or both.
Results
A total of 54 of 207 patients had incomplete PVD (prevalence, 26.1%). Younger age and lattice degeneration were associated independently with incomplete PVD. After a mean follow-up of 5 years (range, 4 to 8 years), 16 patients (9.7%) experienced some adverse outcome. In 5 patients (2.7%), new retinal tears and 1 retinal detachment developed. In 12 patients (7.6%), epimacular membranes developed. Patients with incomplete PVD at baseline experienced significantly more adverse outcomes than patients with complete PVD (Kaplan-Meier estimated probability and standard error, 19.2% and 0.061 vs 5.4% and 0.02; P = .01, log-rank test).
Conclusions
Up to one fourth of symptomatic, acute, and uncomplicated PVDs show incomplete posterior vitreous separation. Delayed complications related to PVD, like retinal tears and epimacular membranes, develop more frequently in patients showing incomplete PVD.
Posterior vitreous detachment (PVD) generally is perceived as an acute and completed event. However, new diagnostic techniques recently challenged this notion. Optical coherence tomography (OCT) reveals that PVD slowly begins at the posterior pole at the fourth decade of life. Dynamic vitreous biomicroscopy with preset lenses and B-scan ultrasound provide evidence that incomplete or partial PVD is more prevalent than previously estimated by standard biomicroscopy. Ultrastructural and histologic studies support this finding, suggesting that biomicroscopy overestimates the rate of complete PVD. Despite its acute presentation, posterior vitreous cortex may not be detached completely in some eyes with symptomatic PVD. Abnormal separation of the posterior vitreous cortex from the retina plays a key role in the pathogenesis of many vitreoretinal disorders. The posterior vitreous cortex exerts dynamic traction over areas of persistent vitreoretinal adherence, which would contribute to the development of macular holes and retinal tears. Epiretinal membranes likely develop over residual posterior vitreous, which could have remained over the retina after PVD. The primary outcomes of the present study were to investigate whether incomplete PVD occurs at the time of acute symptomatic PVD presentation and to determine its clinical relevance.
Methods
This was an observational, prospective cohort study. Patients were enrolled between April 2001 and December 2004 and were followed-up until December 2009 in a nonreferral hospital that attends a population of all people living in a defined geographic area. The inception cohort consisted of a consecutive series of patients with symptomatic PVD who sought treatment on an emergency basis.
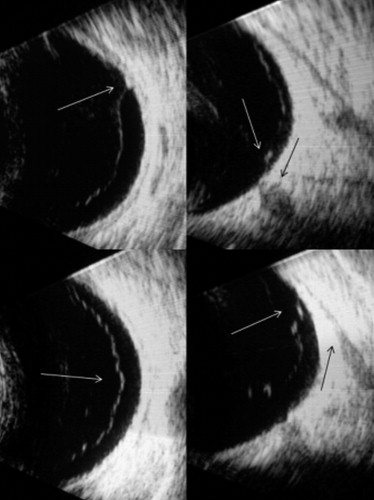
General ophthalmologists evaluated patients with recent onset of visual floaters, photopsia, or both. Diagnostic ophthalmic examination included slit-lamp vitreous biomicroscopy and peripheral retina evaluation with contact lens or indirect binocular ophthalmoscopy. The attending ophthalmologists established the diagnosis of PVD when the detached posterior vitreous cortex was detectable by slit-lamp biomicroscopy. Patients were excluded if their symptoms had been present for more than 1 month or if they had any ocular disease thought to modify the vitreous condition (eg, history of recent eye trauma, eye surgery, vascular retinal diseases, uveitis, or tumors). Patients with media opacity that precluded proper ophthalmoscopic examination (eg, cataract, vitreous hemorrhage, corneal opacities, or narrow pupil) also were excluded. After initial examination, all participants were referred for both B-scan ultrasound and expert retinal evaluation to confirm vitreoretinal findings. Patients who had retinal tears were treated with transpupillary photocoagulation and were excluded from the cohort. The author performed all ultrasound examinations under topical anesthesia with the patient in the supine position. A high-sensitivity gain setting of 105 dB, specifically designed for vitreous examination, was selected (B-scan V Plus/Biovision; Quantel Medical, Clermont-Ferrand, France). The patient’s eyelids were kept open, and the probe was placed directly on the ocular surface behind the limbus through 2.5% methyl cellulose as a coupling gel. The probe was positioned in cross-sections all around the conjunctiva and then progressively shifted to examine more peripheral aspects of the vitreoretinal interface. For adequate characterization of vitreoretinal relationships, vitreous movement was evaluated during voluntary saccadic motion of the patient’s globe while the probe was held stationary (kinetic examination). Characteristically, when the posterior vitreous was detached, the cortex showed distinct, continuous, homogeneous, inertial, and freely mobile linear echoes ( Figure 1 , Bottom left). The author classified the posterior vitreous status according to ultrasound findings as follows: (1) complete PVD when the detached posterior vitreous cortex could be delineated up to the vitreous base without showing any adherence to the retina located posterior to the vitreous base; and (2) incomplete PVD when any vitreoretinal adherence could be detected at or posterior to the globe equator. Additionally, ocular axial length was measured with the A-mode in each patient. After enrollment in the study, the author instructed all patients to return for further evaluation in case they had new or increased symptoms; otherwise, patients returned for follow-up evaluations on a yearly basis.
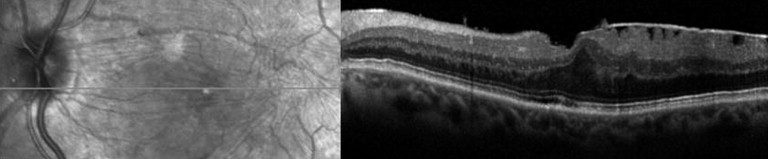
The author performed all follow-up examinations, which included a full dilated funduscopic examination to search for new vitreoretinal pathologic features. Only epimacular membranes showing foveal involvement, wrinkling of the inner retina, and vascular tortuosity were considered to be clinically significant adverse outcomes. OCT confirmed the diagnosis of epimacular membrane. Patients dropped out of the study if they had any intraocular surgery or ocular disease likely to change the vitreous condition, moved from the study area, died, or wanted to discontinue the study. Patients who failed to maintain a follow-up appointment were contacted by phone and were offered a new appointment. In case they finally missed the annual follow-up visit or could not be contacted, they also were censored. Patient flow and individuals lost to follow-up through the study were analyzed.
A stepwise binary logistic regression analysis estimated the association of age, sex, ocular axial length, presence of lattice-like degenerations, and reports of flashes to incomplete PVD at presentation. A Kaplan-Meier analysis with all available data from each patient in the cohort estimated the probability of developing any adverse outcome of interest and its standard error. The time-to-event curves of patients with complete and incomplete PVD were compared with the log-rank test to determine whether incomplete PVD at presentation modifies the natural history of complete PVD. Statistical calculations were carried out with the Statistical Package for the Social Sciences software version 19 (IBM-SPSS, Inc, Chicago, Illinois, USA).
Results
The initial sample comprised 215 patients, 8 of whom were excluded because of incorrect data registration during follow-up. Therefore, the inception cohort included 207 patients. The accrual period was 4 years. The mean and median follow-up were both 5 years (range, 4 to 8 years). Analysis of dropouts throughout the study revealed that 85% (176/207) of participants completed the 4-year follow-up visit and 73.3% (44/60) of the theoretic sample (patients recruited during the first year only) also completed the 8-year follow-up visit. Among a total dropout of 45 patients during the study period, there were 30 patients (14% of the sample) who dropped out of the study because of unintentional causes (eg, deceased, moved from the study area, underwent ocular surgery). However, there were 15 patients who missed their follow-up appointments or wanted to discontinue the study. These voluntary dropouts represented 7% of the sample. A sensitivity analysis showed no significant differences in any baseline studied variable between patients lost to follow-up and those remaining in the study.
Table 1 describes demographics and vitreous status at baseline. A total of 153 patients (73.9%) showed complete PVD ( Figure 1 , Bottom left), whereas 54 patients (26.1%) showed incomplete PVD. Among patients with incomplete PVD, 26 patients (12.5%) had incomplete peripheral PVD, where the anterior limit of PVD was at the globe equator ( Figure 1 , Bottom right). Twelve patients (5.7%) showed an isolated peripheral vitreoretinal attachment ( Figure 1 , Top left), and 16 patients (7.7%) had incomplete PVD showing strands of posterior vitreous cortex still attached to the posterior pole ( Figure 1 , Top right). Baseline ophthalmoscopic evaluation disclosed lattice with atrophic holes in 4 eyes (1.9%), retinal holes in 4 eyes (1.9%), lattice without holes in 20 eyes (9.6%), retinal whitening in 2 eyes (1%), retinoschisis in 2 eyes (1%), benign retinal findings in 12 eyes (5.7%), and normal fundus in 163 eyes (78.7%). Multivariate logistic regression analysis revealed that younger age at PVD onset and presence of lattice-like degeneration were associated significantly and independently with incomplete PVD. However, axial length, reports of flashes, and gender were not related to incomplete PVD ( Table 2 ).
| Men | Women | P Value | |
|---|---|---|---|
| No. (%) | 81 (39%) | 126 (61%) | |
| Age (mean) | 66.9 ± 5.3 | 64.1 ± 7.1 | .004 a |
| Axial length (mean) | 23.77 ± 0.93 | 23.12 ± 1.16 | .001 a |
| Incomplete PVD (%) | 22 (27.2%) | 32 (25.4%) | NS b |
| Complete PVD (%) | 59 (72.8%) | 94 (74.6%) | NS b |
| Covariates | All PVDs | Complete PVDs | Incomplete PVDs | Significance a | Odds Ratio a | 95% CI a | |
|---|---|---|---|---|---|---|---|
| Superior | Inferior | ||||||
| Male gender (%) | 39.1 | 38.6 | 40.7 | .87 | .65 | .26 | 1.64 |
| Axial length (mm) | 23.36 | 23.37 | 23.33 | .81 | 1.20 | .87 | 1.64 |
| Age (years) | 65.25 | 66.13 | 62.72 | .006 | 1.08 | 1.02 | 1.14 |
| Flashes (%) | 14.6 | 11.8 | 15.5 | .64 | 1.30 | .45 | 3.73 |
| Lattice (%) | 11.6 | 5.9 | 27.8 | .001 | 7.29 | 2.61 | 20.3 |
Over a mean and median follow-up of 5 years, 1 or more adverse outcomes developed in 16 participants (9.7%). In 5 patients (2.7%), late retinal tears developed in the study eye. Four of these patients had incomplete PVD at study entry, and only 1 initially had a complete PVD ( Table 3 ). Epimacular membranes developed in 12 patients (7.6%; Figure 2 ) . In 1 of these patients, both 1 retinal tear and epimacular membrane developed in the study eye ( Table 4 , Case 4). Nine patients reported symptomatic loss of vision, 2 of whom underwent macular surgery. Comparing both groups, the time-to-event curves of patients with incomplete PVD were different from that of patients with complete PVD (log-rank chi-square = 6.55; P = .01; Figure 3 ) . Patients with incomplete PVD were more likely to experience adverse outcomes related to PVD (retinal tears, epimacular membranes, or both) than patients with complete PVD ( Figure 3 ).
| Baseline Findings | No. | Retinal Tears | EMM | Any Adverse Outcome a |
|---|---|---|---|---|
| Incomplete PVD | 54 | 4 (7.5%), 0.036 | 5 (11,4%), 0.052 | 9 (19.2%), 0.061 |
| Complete PVD | 153 | 1 (0.8%), 0.008 | 7 (5.4%), 0.02 | 7 (5.4%), 0.02 |
| Total | 207 | 5 (2.7%) | 12 (7.6%) | 16 (9.7%) |
a Number and percentage of eyes. Several adverse outcomes may occur in the same eye.
| Case No. | Age (y) | Gender | Eye | Axial Length (mm) | Echographic Findings | Fundus Findings | Adverse Outcome | Elapsed Time | New-Onset Symptoms |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 59 | F | R | 23.27 | Incomplete PVD + VR traction | Lattice | EMM | 4 y | ↓Vision |
| 2 | 73 | F | R | 22.72 | No | Normal | EMM | 3 y | ↓Vision |
| 3 | 73 | F | L | 22.50 | No | Normal | EMM | 4 y | ↓Vision |
| 4 | 65 | M | L | 24.96 | PVD + synchysis | Normal | EMM + operculum RT + EMM (FE) | 3 y | None |
| 5 | 68 | F | R | 22.79 | Incomplete PVD + posterior adherence | Normal | EMM | 1 y | ↓Vision (PPV) |
| 6 | 72 | M | L | 24.38 | Incomplete PVD + traction | Retinal hole | RT | 1 wk | Floaters |
| 7 | 60 | F | R | 22.70 | Incomplete PVD | Lattice | EMM | 4 y | ↓Vision |
| 8 | 53 | F | R | 23.33 | Incomplete PVD + synchysis | Normal | RD | 1 mo | ↓Visual field (PPV) |
| 9 | 66 | F | R | 22.78 | Synchysis | Normal | EMM | 4 y | ↓Vision |
| 10 | 73 | F | L | 21.83 | PVD | Normal | EMM | 1 y | None |
| 11 | 65 | F | R | 22.38 | Incomplete PVD + traction | Normal | EMM | 3 y | None |
| 12 | 64 | M | R | 22.47 | Incomplete PVD + synchysis | Normal | EMM | 7 y | ↓Vision (PPV) |
| 13 | 59 | F | R | 21.95 | Incomplete PVD + traction + synchysis | Lattice + RH | RT + RT | 2 y, 7 y | Floaters + floaters |
| 14 | 64 | M | L | 22.70 | PVD + synchysis | Normal | EMM | 2 y | ↓Vision |
| 15 | 69 | F | L | 22.74 | PVD | Peripheral cystoid degeneration | EMM | 1 y | ↓Vision |
| 16 | 63 | M | R | 26.26 | Incomplete PVD + traction + synchysis | Lattice + RH | Operculum + RH | 1 mo | Floaters |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


