Purpose
To investigate the incidence rate of massive submacular hemorrhage (SMH) in patients with polypoidal choroidal vasculopathy (PCV) and analyze the associated risk factors.
Design
Retrospective cohort study.
Methods
Patients diagnosed with PCV from May 2003 to May 2014 were included. Two hundred forty-five eyes of 245 patients were enrolled. The time between the initial visit to the clinic with subjective visual symptoms and the date of massive SMH was recorded. SMH larger than 4 disc diameters was defined as massive SMH. Age; hypertension; visual acuity (VA); indocyanine green angiography findings, including the greatest linear dimension, largest polyp size, and PCV type (cluster vs non-cluster); and treatment methods were reviewed for risk factor analysis using Kaplan-Meier survival and Cox regression analyses.
Results
The incidence rate of massive SMH within 1 year after the initial visit was 2.45%. Massive SMH occurred within 3, 5, and 10 years after the first visit in 6.17%, 11.09%, and 29.85% of patients, respectively. Cox regression analysis revealed that the cluster type of PCV was significantly associated with massive SMH (hazard ratio [HR], 3.418; P = .003). Photodynamic therapy followed by anti–vascular endothelial growth factor injection lowered the risk of massive SMH (HR = .242; P = .047]. Final VA in eyes with massive SMH was significantly lower than that in patients without massive SMH (1.34 ± 0.66 vs 0.63 ± 0.53 logMAR; P < .001).
Conclusions
Patients with PCV who develop massive SMH experience severe vision loss. The incidence rate of massive SMH in PCV increases with time. The cluster type of polyp in PCV is a significant risk factor for massive SMH.
Polypoidal choroidal vasculopathy (PCV), which is considered a subtype of neovascular age-related macular degeneration (AMD), is characterized by the formation of a branching vascular network (BVN) and polypoidal dilation of its termini. Since the first description of PCV by Yanuzzi and associates, several attempts have been made to reveal the natural course of PCV and optimal treatment modalities ; however, a definite course and prognosis remain unclear to date. In addition, the incidence rate of PCV differs among ethnic groups. PCV is most common in Asians (reported in 22%–55% of Asian patients with neovascular AMD) and has a relatively low prevalence in white patients (4%–9.8% of patients with neovascular AMD).
PCV persists for a long time and therefore it is difficult to clarify the natural history of this disorder. Several studies have reported that PCV is more chronic and results in a higher incidence of visual loss compared with neovascular AMD. According to previous studies, PCV is a chronic vascular abnormality resulting in multiple, recurrent, serosanguinous retinal pigment epithelial detachments (PEDs) secondary to leakage and bleeding from choroidal vascular lesions. Subretinal hemorrhages (SRH) or submacular hemorrhages (SMH) may occur because of the rupture of these abnormal areas of vascular dilation or aneurysmal venules. SMH in the subfoveal region, particularly when massive (ie, >4 disc diameter area), induces an abrupt decrease in the visual acuity (VA) and can lead to retinal pigment epithelium (RPE) and outer retinal degeneration, which result in a poor visual prognosis. Previous studies mostly showed the incidence rate of SRH by using cross-sectional data. Outcome variables were not uniform and any SRH, small or massive, was regarded as equal. To our knowledge, the incidence rate of massive SMH in patients with PCV during long-term follow-up has not been reported.
In the present study, we investigated the incidence rate of massive SMH in PCV patients undergoing treatment in actual clinical settings. In addition, we analyzed age, VA, hypertension, treatment history, indocyanine green angiography (ICGA) findings, and optical coherence tomography (OCT) findings as risk factors for massive SMH in PCV patients.
Methods
We retrospectively reviewed the medical records of patients diagnosed with PCV at Seoul National University Bundang Hospital from May 2003 to May 2014. This study was reviewed and approved by the institutional review board (IRB) of Seoul National University Bundang Hospital (IRB No. B-1509/314-103, Seongnam, South Korea). The study complied with the guidelines of the Declaration of Helsinki.
The inclusion criteria were as follows: diagnosis of PCV and a follow-up period that was longer than 1 year. PCV was diagnosed on the basis of criteria from the EVEREST study report 2. In brief, the diagnosis of PCV met all of the following diagnostic criteria: nodular hyperfluorescence of the polyp(s) on ICGA, hypofluorescent halo around the nodule(s), abnormal vascular channel(s) supplying the polyp(s) (BVN), and orange subretinal nodules on fundus photography corresponding to the polyp(s) on ICGA.
The exclusion criteria were as follows: presence of other concomitant diseases such as glaucoma, retinal vein occlusion, retinal artery occlusion, diabetic retinopathy, and keratitis; and PCV treatment at another hospital prior to the current visit. For patients with bilateral PCV, we selected 1 eye that clearly showed the initial symptom at onset.
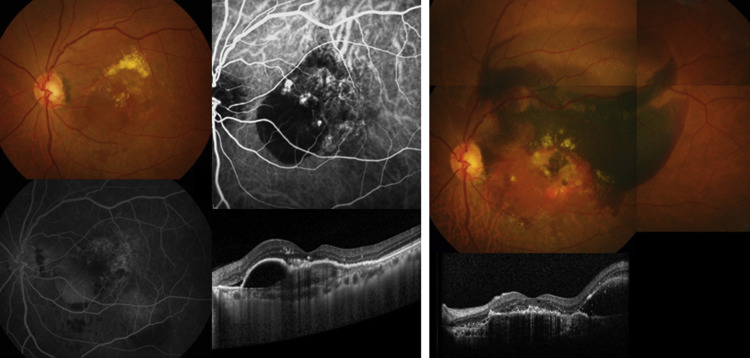
In total, 245 eyes of 245 patients satisfied all the inclusion and exclusion criteria. The day of the visit to our retina clinic with the first subjective signs of visual impairment was considered the initial visit. The data collected on the day of the initial visit included the patients’ medical history, age, sex, baseline VA, and slit-lamp microscopy, dilated fundus examination, OCT (Spectralis OCT; Heidelberg Engineering Inc, Heidelberg, Germany), fluorescein angiography (FA), and ICGA findings. OCT findings included the central foveal thickness (CFT) and the presence of subretinal fluid (SRF) or PED. ICGA findings included the size of the largest polyp and the greatest linear dimension (GLD) including the BVN. We also assessed whether the polyps were present in clusters (cluster type) or not (non–cluster type) ( Figure 1 ) and the location of the lesion (with or without foveal involvement). Then, the complete clinical course of the patients’ condition was reviewed, including the occurrence of any SMH larger than 1 optic disc diameter (DD) and treatments with anti–vascular endothelial growth factor (VEGF) injection or photodynamic therapy (PDT).
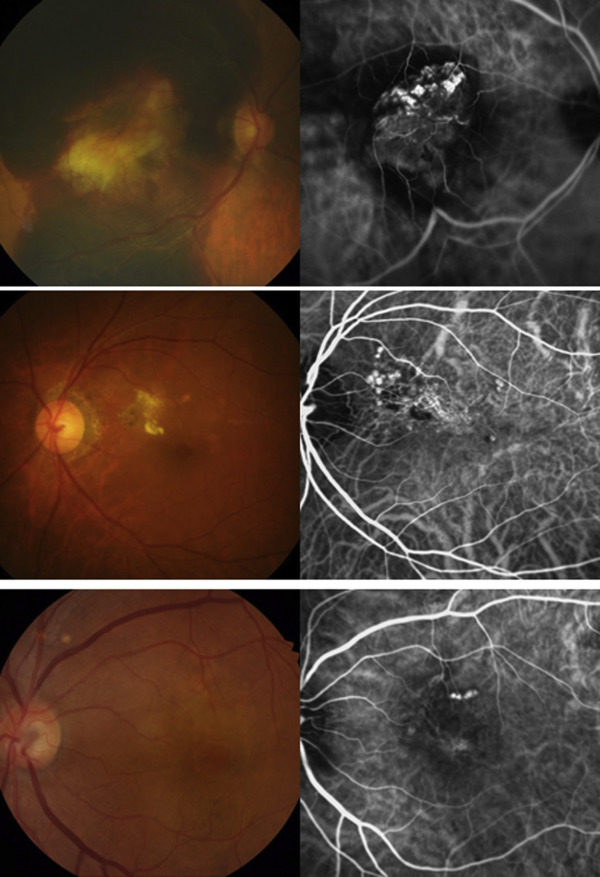
SMH larger than a circle with a radius of the distance between the optic disc and fovea was defined as massive SMH of area larger than approximately 4 DD. The size used to define a massive SMH was in accordance with that in a previous report. When the size of the SMH was less than that of a massive SMH, but more than 1 DD, it was considered a significant SMH. The occurrence of significant SMH was also checked during the clinical course. If a patient presented with a massive SMH, we discontinued reviewing the further course of that patient. If a patient did not present with a massive SMH, the latest visit was considered the last visit and it was regarded as censored data in the survival analysis. The follow-up period was defined as the number of years from the date of the initial visit to the date of the last visit or the occurrence of a massive SMH ( Figure 2 ). The VA at the last visit was recorded as the final VA for each patient. For eyes with a massive SMH, the VA at 1 year after the occurrence of massive SMH was reviewed.
Enrolled patients underwent treatment for PCV by 2 retinal specialists (K.H.P., S.J.W.). The protocol for treating PCV prior to 2006 at our center was PDT for primary treatment. After 2006, primary treatment for PCV was with intravitreal anti-VEGF injections. An initial 3 monthly injections of anti-VEGF agents (bevacizumab [1.25 mg/0.05 ml Avastin, Genentech, San Francisco, California, USA], ranibizumab [0.5 mg/0.05 ml Lucentis, Genentech, San Francisco, California, USA], or aflibercept [2.0 mg/0.05 ml Eylea, Regeneron Pharmaceuticals, Tarrytown, New York, USA]) were performed and the need for additional injections was determined if any of the following occurred: appearance of any qualitative change on the OCT images suggesting recurrent macular fluid, or persistent fluid on OCT 1 month after the previous injection. PDT was added if there was a limited response after administration of the anti-VEGF drug. The total number of PDTs or anti-VEGF injections for each patient was reviewed. We grouped patients according to their primary treatment (PDT or anti-VEGF injection). Additional grouping was performed if they were treated with an additional anti-VEGF injection or PDT following their initial treatment; PDT or anti-VEGF injection, respectively. All patients were grouped into 5 subgroups: (1) no treatment, (2) PDT only, (3) PDT followed by anti-VEGF injection, (4) anti-VEGF injection only, (5) anti-VEGF injection followed by PDT. Numbers and percentage of patients of each group and the mean follow-up period in each group were calculated. Also, the number of PDTs per year or number of anti-VEGF injections per year (ie, the average number of treatments performed in a year) was presented in each group.
We previously reported genetic and environmental risk factors in Korean patients with AMD and found that the single nucleotide polymorphisms (SNPs) ARMS2 rs10490924 and CFH rs800292 were risk SNPs for Korean AMD. Of 245 enrolled patients, gene data of 102 patients were available. Two SNPs, ARMS2 rs10490924 and CFH rs800292, were selected and a Cox regression analysis was performed using 3 different alleles in each of the 2 SNPs to check the association between the occurrence of massive SMH and SNPs.
The incidence rate of massive SMH in PCV patients was calculated using a Kaplan-Meier survival analysis. The date of the initial visit was designated as an index date. The date of massive SMH occurrence was used as the event date in survival curves. To investigate the risk factors for massive SMH, univariable Cox regression analysis was performed for each clinical variable, including age, VA at the initial visit, hypertension, GLD, largest polyp size, foveal involvement, cluster type, and the occurrence of significant SMH. Treatment modalities of the 5 subgroups were also analyzed using a Cox regression analysis to check the association of the 4 treated groups compared with the “no treatment” group. Multivariable Cox regression analysis was then performed and included factors that were significant in the univariable analysis. To assess the visual prognosis in the 2 groups, patients with and without massive SMH and the initial and final VAs were compared between the 2 groups using independent t tests and χ 2 tests. Statistical analyses were performed using SPSS version 22.0 for Windows (SPSS, Inc, Chicago, Illinois, USA), and a P value <.05 was considered to indicate a statistically significant difference.
Results
The baseline characteristics of the 245 enrolled patients (245 eyes) are shown in Table 1 . The mean age ± standard deviation (SD) was 66.5 ± 7.8 years, and the male-to-female ratio was 169:76 (69.0%:31.0%). At the initial visit, the mean BCVA was 0.59 ± 0.51 logMAR and the mean CFT was 341.19 ± 140.29 μm. The number of eyes with SRF and with PED was 171 (69.8%) and 137 (55.9%), respectively. ICGA revealed polyps arranged in grape-like clusters (cluster type) in 107 eyes (43.7%) and foveal involvement in 167 (68.2%). The mean ± SD GLD was 2039.9 ± 1049.9 μm and the mean size of the largest polyp was 227.9 ± 114.0 μm. The mean follow-up period for all patients was 4.57 ± 2.70 years, with 1 patient having a maximum follow-up period of 11.35 years ( Table 1 ). The mean follow-up period without the occurrence of massive SMH was 4.7 ± 2.7 (range 1.1–11.4) years. The mean period until the occurrence of massive SMH was 3.7 ± 2.7 (range 0.1–9.3) years. Ninety-seven of 245 eyes (39.6%) developed significant SMH during the follow-up period.
| Characteristics | |
|---|---|
| Eyes | 245 |
| Age of onset | 66.5 ± 7.8 |
| Men (n, %) | 169 (69.0) |
| Hypertension (n, %) | 126 (51.4) |
| Visual acuity, logMAR | 0.59 ± 0.51 |
| Baseline OCT characteristics | |
| Central foveal thickness (μm) | 341.2 ± 140.3 |
| Presence of subretinal fluid (n, %) | 171 (69.8) |
| Presence of pigment epithelial detachment (n, %) | 137 (55.9) |
| Indocyanine green angiography findings | |
| Cluster type (n, %) | 107 (43.7) |
| Foveal involvement (n, %) | 167 (68.2) |
| Greatest linear dimension (μm) | 2039.9 ± 1049.9 |
| Largest polyp size (μm) | 227.9 ± 114.0 |
| Mean follow-up period (y, range) | 4.6 ± 2.7 (1.0–11.4) |
The number of eyes with massive SMH was 32 (13.1%) during the follow-up period. Kaplan-Meier analysis revealed that the calculated incidence rate of massive SMH within a year after the initial visit was 2.45% ( Figure 3 ). Thereafter, massive SMH occurred in 6.17%, 11.09%, and 29.85% of eyes within 3, 5, and 10 years, respectively. Univariable Cox regression analysis with each of the 9 variables revealed a statistically significant association of massive SMH with the cluster type of PCV (hazard ratio [HR], 3.418; 95% confidence interval [CI], 1.527–7.648; P = .003). Other variables such as age, hypertension, VA, CFT, GLD, size of the largest polyp more than 200 μm, foveal involvement, and the occurrence of significant SMH showed no significant association with the occurrence of massive SMH ( Table 2 ).
| Risk Factors | Hazard Ratio (95% CI) | P Value a |
|---|---|---|
| Age | 1.011 (0.966–1.058) | .634 |
| Hypertension | 1.021 (0.509–2.045) | .954 |
| Visual acuity on the day of onset (logMAR) | 0.899 (0.439–1.840) | .771 |
| Central foveal thickness | 1.002 (1.000–1.004) | .103 |
| Polypoidal lesion | ||
| Greatest linear dimension | 1.000 (1.000–1.000) | .401 |
| Largest polyp >200 μm | 0.702 (0.349–1.413) | .322 |
| Foveal involvement | 0.744 (0.357–1.551) | .430 |
| Cluster type | 3.418 (1.527–7.648) | .003* |
| Occurrence of significant SMH b | 1.467 (0.733–2.937) | .279 |
a Univariable Cox regression analysis.
b Size more than 1 disc diameter, but not fulfilling the criteria for massive SMH.
Another Kaplan-Meier curve was created according to the type (cluster vs non-cluster) of polyps using a Cox proportional hazard regression analysis ( Figure 4 ). The incidence rate of massive SMH for the cluster type was 4.67% and 9.79% within 1 and 3 years, respectively, whereas that for the non–cluster type was 0.72% and 3.30%, respectively (HR, 3.418; P = .003).
A total of 143 patients (58.3%) received PDT and 210 (85.7%) received anti-VEGF injections (bevacizumab, ranibizumab, or aflibercept). Sixteen patients (6.53%) did not need treatment; their mean follow up period was 3.0 ± 1.8 years. Characteristics of the 5 subgroups are shown in Table 3 . Twenty patients (8.2%) received PDT alone, whereas 58 patients (20.7%) required anti-VEGF injections in addition to PDT. Seventy-eight patients (31.8%) received anti-VEGF injections for primary treatment and did not need additional PDT; however, 73 (29.8%) needed PDT in addition to anti-VEGF injections. The mean follow-up periods of the 5 groups were statistically different ( P < .001). This might be because the initial symptom onset of patients who got PDT for the first treatment was mostly before 2006 and the follow-up period might be longer than for those with anti-VEGF injection for the first treatment. Although the mean number of PDT per year was not statistically different among groups, the mean number of anti-VEGF injections per year was significantly higher in 2 groups, which received anti-VEGF injection for the first treatment ( P = .001). Risk factor analysis for treatment methods revealed that the “PDT followed by anti-VEGF injection” group had a significantly lower occurrence of massive SMH than the “no treatment” group (HR = .242; P = .047) ( Table 3 ).
| Treatment Group | N (%) | Mean Follow-up Period (y) | Mean Number of PDTs per Year | Mean Number of Anti-VEGF Injections per Year | Hazard Ratio Compared With “No Treatment” Group (95% CI) | P Value a |
|---|---|---|---|---|---|---|
| No treatment | 16 (6.53) | 3.00 ± 1.77 | ||||
| PDT only | 20 (8.16) | 5.65 ± 2.94 | 0.46 ± 0.93 | 0.654 (0.157–2.722) | .560 | |
| PDT followed by anti-VEGF injection | 58 (23.67) | 6.07 ± 3.13 | 0.40 ± 0.29 | 1.84 ± 1.32 | 0.242 (0.059–0.984)* | .047* |
| Bevacizumab | 30 | |||||
| Ranibizumab | 27 | |||||
| Aflibercept | 1 | |||||
| Anti-VEGF injection only | 78 (31.84) | 3.75 ± 2.21 | 2.49 ± 2.02 | 0.406 (0.107–1.541) | .185 | |
| Bevacizumab | 14 | |||||
| Ranibizumab | 63 | |||||
| Aflibercept | 1 | |||||
| Anti-VEGF injection followed by PDT | 73 (29.80) | 4.31 ± 2.32 | 0.44 ± 0.34 | 2.91 ± 1.85 | 0.362 (0.095–1.383) | .137 |
| Bevacizumab | 18 | |||||
| Ranibizumab | 55 | |||||
| P value b | <.001* | .057 | .001* |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


