Purpose
To use iCare rebound tonometry in the home setting for documentation of diurnal intraocular pressure (IOP) fluctuations in children.
Design
Nonrandomized, prospective clinical study.
Methods
Pediatric ophthalmology clinic patients were recruited between October 2009 and February 2010 who were able to cooperate with IOP measurement by iCare rebound tonometry and whose caregiver was willing and able to obtain iCare measurements at home. The child’s IOP was measured first by iCare tonometry followed by a second method (Goldmann applanation [GAT]). The caregiver was instructed on the use of the iCare tonometer. The subject’s IOP was measured by the caregiver at home at designated time periods for at least 2 consecutive days.
Results
Seventeen children (17 eyes) with known or suspected glaucoma and 11 normal children were included. Excellent reliability was obtained by caregivers in 70% of iCare measurements. Mean difference between iCare and GAT in clinic was 2.0 ± 4.0 mm Hg, P = .08. Daily IOP fluctuation occurred in both subjects with glaucoma and normal subjects. In children with known or suspected glaucoma, relative peak and trough IOPs occurred in the early morning (45%) and late evening (43.5%), respectively. Comparison of the peak IOP measured at home vs in the clinic was >6 mm Hg in 5 of 16 subjects (31%) and affected glaucoma management in several subjects.
Conclusions
In selected children with glaucoma, home tonometry by iCare rebound tonometry was reliable, easily performed by caregivers, and well tolerated, and offered IOP information valuable in clinical management.
Fluctuations in intraocular pressures (IOP), thought to contribute to optic nerve damage, may not be captured during routine office visits. Diurnal IOP variations have been reported in adult glaucoma patients, where IOP fluctuation has been suggested as an independent risk factor for glaucoma or disease progression. However, there is little corresponding information regarding pediatric glaucoma patients. Further, there has been no ideal tonometer for self-monitoring or even home monitoring of IOP.
The iCare rebound tonometer (ICare Finland Oy, Helsinki, Finland) has been shown to be a useful screening tool in healthy children. Studies in adult populations, both normal and glaucoma subjects, demonstrate reasonable correlation with Goldmann applanation tonometry, with the iCare reading slightly higher than Goldmann applanation. We found similar results when we compared the iCare rebound tonometer and Goldmann applanation tonometry in children with known or suspected glaucoma. The iCare tonometer does not require topical anesthetic. It thus facilitates the often arduous task of obtaining an IOP measurement in young children, for whom eye drop instillation often provokes anxiety. The iCare tonometer may also permit adult family members to measure IOP in a child in the more familiar home environment, with minimal risk to the child, and at multiple times over several days. This home tonometry information may in turn confirm the presence or absence of IOP fluctuations in a child with known or suspected glaucoma, potentially impacting the child’s glaucoma management. The purpose of the present study was to use iCare tonometry at home for documentation of diurnal IOP fluctuations in children with glaucoma or suspected of having glaucoma.
Methods
Pediatric glaucoma patients were recruited from the practice of the senior author between October 1, 2009 and February 28, 2010. Inclusion criteria were children with known or suspected glaucoma, based on clinical criteria including optic nerve cupping, elevated IOP, and other features such as corneal enlargement, Haab striae (for congenital glaucoma), and visual field defects (when able to measure), and who were able to cooperate with IOP measurement by iCare tonometry. Additionally, only children with a parent willing and able to obtain reliable iCare measurements at home were included. All subjects continued their current glaucoma medication(s) during the period of home IOP monitoring. A control group of healthy children (normal subjects without glaucoma) were also recruited. Exclusion criteria for this control group were a refractive error (spherical equivalent) more than 3 diopters, family history of open-angle glaucoma, history of ocular surgery, and use of any ocular medication. Two unaffected siblings of a child with congenital cataracts and aphakic glaucoma were included.
The iCare tonometer is a handheld instrument for IOP measurement. It is held at a distance of 3 to 10 mm from the unanesthetized eye. A magnetized probe with a disposable round plastic tip is propelled against the eye, impacts, and then rebounds from the cornea. Six readings are taken and an average value (measurement) is generated automatically by the unit. The tonometer has a built-in system to indicate if there is any discrepancy among the readings. As designated by the instruction manual, we recorded these reliability indicators as Psolid, Pbottom, Pmiddle, and Ptop (in descending order of reliability). These reliability indicators reflect standard deviation of the individual IOP measurements of <1.8 mm Hg (Psolid), 1.8 to 2.5 mm Hg (Pbottom), 2.5 to 3.5 mm Hg (Pmiddle), and >3.5 mm Hg (Ptop) (personal communication, Markku Eräluoto of i-care Finland Oy, printed in Josephson A. Goldmann applanation tonometry versus i-care rebound tonometry. Opticianonline.com , 16.01.09).
In the office, prior to instillation of any eye drops, the child’s IOP was measured using the iCare tonometer by a clinician until a Psolid reading or 3 measurements of IOP were taken (with lower reliability indicators than Psolid, see paragraph above). The designated adult parent/guardian (“caregiver”) was then trained to use the iCare rebound tonometer by study personnel and was subsequently observed taking the child’s IOP with the iCare tonometer to ensure proper use. Next, Goldmann tonometry (Haag-Streit USA, Mason, Ohio, USA) was performed by a different clinician, who was masked to the iCare readings and performed all study Goldmann IOPs. If Goldmann applanation could not be performed, IOP was measured by Tonopen XL (Mentor Ophthalmics Inc., Norwell, Massachusetts, USA). The caregiver was instructed to measure the IOP at given time periods, for a total of 6 measurements per day (approximately every 2 to 3 hours from 6:00 AM to 11:00 PM [or bedtime]); recorded were IOPs as well as reliability designations (Psolid, Pbottom, Pmiddle, Ptop) as outlined by the instruction manual, using standardized study recording sheets. Both eyes were measured if possible, unless the child was monocular. iCare measurements were taken from each available eye until a Psolid reading or a total of 3 sets of measurements were taken. Telephone consultation was available to the caregiver for any problems while using the instrument at home.
The study duration was 2 full recording days for most subjects, as well as a partial third day (most readings began on a Friday afternoon and continued throughout Saturday and Sunday, with the instrument returned to the clinic on Monday). Both the child subject and the adult performing the iCare tonometry were specifically asked to record any difficulties in using the machine, as well as any discomfort encountered by the child during the measurements. Upon return of the instrument, these queries were verbally repeated by the investigator receiving back the iCare unit.
Demographic data and clinical features including subject age, gender, ethnicity, diagnosis, associated ocular disease, presence or absence of strabismus, presence or absence of nystagmus, anterior segment abnormality, best-corrected visual acuity, previous surgical procedures, and glaucoma treatment regimens were recorded. The subject’s clinic chart (for non-controls) was reviewed for IOP measurements taken in clinic (Goldmann applanation IOP was always recorded in preference to Tonopen XL IOP readings, but in three children, only Tonopen XL IOP readings were obtainable in clinic), surgical interventions, and glaucoma medication changes for 2 years prior to enrollment in the study. Subjects were selected for the home IOP monitoring for a variety of reasons ( Table ).
| Subject | Glaucoma Type | Indication for iCare Home Tonometry / Outcome of iCare Home Tonometry |
|---|---|---|
| 1 | Aphakic | Monocular patient after Baerveldt glaucoma drainage device implantation with dissolvable suture ligature. Monitored to determine when suture ligature dissolved. / Able to manage medications and avoid severe hypotony. |
| 2 | JOAG | IOPs borderline in clinic on maximal medication. Monitored to determine whether even higher IOPs occurred off office hours. / Confirmed stable IOPs. |
| 3 | Uveitic | High IOP recorded in early AM clinic visit, outside usual IOP range. Monitored to determine maximal early AM IOP in that eye. / Confirmed early AM IOP spikes and need for additional surgery. |
| 4 | Aphakic | Elevated IOPs in clinic in better-seeing eye. Monitored to determine need for further surgical intervention. / Confirmed elevated IOPs and need for surgery. |
| 5 | Aphakic | Elevated IOPs after endocyclophotocoagulation. Monitored to determine need for further surgical intervention. / Confirmed need for further surgery. |
| 6 | Aphakic | Sporadically elevated IOPs in clinic with possible noncompliance and difficult clinical examination secondary to small pupil, nystagmus, and unreliable visual fields. Monitored to determine if IOP remained elevated despite proper use of glaucoma medications. / Confirmed persistently elevated IOPs leading to an examination under anesthesia and subsequent surgical intervention. |
| 7 | Congenital | Bilateral congenital glaucoma with aqueous shunt in the left eye. Right eye stable off medications, left eye on monotherapy with 1 IOP reading over target in clinic. Monitored to determine control of IOP. / Confirmed need for treatment escalation left eye. |
| 8 | Aphakic | Monocular patient who lived >3000 miles from provider. Used tonopen at home by parents; however, desired nonanesthetized method. Monitored to determine control of IOP as well as comparison with tonopen method. / Confirmed control of IOP and became preferred method by child and parents. |
| 9 | Aphakic | Severe optic nerve damage and difficulty controlling IOP within low target despite multiple surgeries and maximum medical treatment. Monitored to determine IOP control and whether further surgery was indication. / Confirmed IOP control and surgical intervention delayed with close observation. |
| 10 | Aphakic | Bilateral severe optic nerve damage with aphakia and scleral buckles OU. IOP above target in clinic but suspected poor compliance. Monitored to determine need for risky glaucoma surgery vs continued maximum medical therapy. / Confirmed poor IOP control and likely prompted better compliance. |
| 11 | Aphakic | Bilateral aphakic glaucoma with elevated IOPs in clinic but parents wanted to confirm prior to additional surgery. Monitored to determine IOP control and confirm need for surgery. / Confirmed need for further surgery. |
| 12 | Congenital | Isolated high IOP recorded in clinic, outside target IOP, with possible missed AM drop. Monitored to determine IOP control. / Confirmed IOP control below target with current medical therapy. |
| 13 | Aphakic | Variable IOPs measured in clinic; however, patient was nervous in clinic. Monitored to determine if variability was true. / Confirmed variability of IOPs OD and resulted in escalation medical therapy; confirmed stability OS. |
| 14 | Uveitic | High IOP measured at postoperative visit following Ahmed valve implantation; dropped with vigorous digital massage. Patient lived 3 hours from provider. Monitored to determine IOP stability and need for further digital massage or other intervention. / Confirmed IOP stability without further intervention. |
| 15 | Suspect | Currently off medications. Monitored to determine IOP control. / Confirmed adequate IOP control without medications. |
| 16 | Anterior segment dysgenesis | Monocular Peters anomaly status post penetrating keratoplasty, cataract extraction, aqueous shunt, and trabeculotomy with difficult IOP measurement in clinic. Monitored to determine IOP control. / Confirmed stable IOPs. |
| 17 | Uveitic | Variable IOPs recorded in clinic. Monitored to determine IOP control. / Confirmed IOP fluctuations resulting in goniotomy OD and close observation OS. |
For analysis of the home tonometry data in the non-control subjects, 1 eye was used per subject, either the eye with glaucoma (if unilateral) or the eye with the most time period measurements. For comparison of the iCare and Goldmann applanation tonometry, 1 eye was used per subject, either the eye with more tenuous glaucoma (if unilateral or asymmetrical) or the right eye. Thirteen of the 17 subjects (76%) were also enrolled in a clinic-based comparison of iCare vs Goldmann applanation tonometry. Both eyes were included in the normal controls. Only measurements with reliability of Psolid or Pbottom were used for analysis. The average IOP was applied when more than 1 measurement with reliability of Psolid or Pbottom was recorded at the same time period.
For purposes of diurnal IOP data evaluation, the IOPs recorded for each time period for 2 consecutive days of the study were averaged together for a given study eye. Eyes with more than 2 missing time periods during the day (24-hour time period) or without an early morning measurement were excluded. For evaluation of peak and trough IOP patterns, the peak and trough IOPs were defined as the maximum or minimum IOP reading, respectively, at a given time period or adjacent time periods, within the 24-hour day. In addition to the exclusion of eyes with more than 2 missing time period measurements during the day (24-hour time period) and eyes without an early morning measurement (as for diurnal IOP evaluation above), we also excluded eyes with less than 3 mm Hg variation throughout the day. According to our criteria, we assumed that each eye included had 1 IOP peak and 1 trough, respectively, per day and quantified this with incidence data.
Comparison was made between the highest and lowest recorded IOP during the iCare home tonometry study vs corresponding Goldmann applanation or Tonopen IOP measurements taken in the clinic. The latter “clinic ranges” included at least 3 visits during the preceding 2 years without changes in glaucoma medication or surgical intervention in the study eye.
Specific clinical case scenarios are presented for 4 selected subjects, to indicate the various situations where home tonometry was useful for different clinical indications (also see Table ).
Subject characteristics were tabulated with the use of number and percent for categorical data, and with the use of median, range, and standard deviation (SD) for continuous variables. Comparisons between groups for continuous variables were completed with the paired or unpaired t test, respectively, as appropriate. All statistical tests were 2-sided and the threshold for significance was set at α = 0.05. All values are reported as mean ± standard deviation unless otherwise noted. Range comparisons were performed using Graphpad prism version 5.01 for Windows (GraphPad Software, San Diego California, USA; www.graphpad.com ).
Results
Patient Population
Seventeen pediatric glaucoma patients seen at the Duke Eye Center from October 2009 to February 2010 who met inclusion criteria were enrolled. The median age was 10 years (range 2-16); 10 (56%) were female. Glaucoma diagnoses were aphakic (9), uveitic (3), congenital (2), suspect (1), and juvenile-onset open-angle glaucoma (1) and anterior segment dysgenesis (1). While 6 (35%) of the subjects had no previous glaucoma surgery, the remaining subjects had undergone 1 (4 subjects, 24%), 2 (6 subjects, 35%), and 3 or more (1 subject, 6%) prior glaucoma surgeries, respectively. IOP was controlled without any glaucoma medication in 2 subjects (12%), on 1 medication in 2 subjects (12%), on 2 medications in 4 subjects (24%), and on 3 or more medications in 9 subjects (53%). The mean Goldmann applanation tonometry IOP at time of home tonometry initiation (when the iCare tonometer left the clinic) among those with known or suspected glaucoma and able to cooperate with Goldmann applanation (n = 14) was 21.8 mm Hg (range 9-34).
The control group comprised 11 healthy children (22 eyes) with median age 10 years (range 6-15). The mean Goldmann applanation tonometry IOP at time of home tonometry initiation among the normal controls was 14.5 mm Hg (range 11-19).
Reliability of iCare Tonometry by Caregivers
The iCare tonometry was well tolerated by all enrolled subjects, without any reported complaints of pain or discomfort. Both caregivers and subjects found the iCare tonometer easy to use and all subjects completed the study. In the glaucoma group, a Psolid reading was obtained in 70% of the recordings included for each time point. The same reliability (70% Psolid) was obtained in the normal control group. Psolid IOP readings were obtained by the experienced clinician at the time of dispensing of the iCare unit in 79% of subjects.
Comparisons Between iCare and Goldmann
Fourteen of the 17 subjects were included in the analysis comparing iCare tonometry IOP to Goldmann applanation IOP. Goldmann applanation could not be obtained in 3 subjects while the child was awake (iCare and Tonopen XL only obtainable in clinic). One eye—the right eye or the eye with the worst glaucoma (eye of interest)—was included in the analysis. The mean difference between IOP by iCare tonometry vs Goldmann applanation tonometry was 2.0 ± 4.0 mm Hg (range -4 to +12 mm Hg, P = .08). iCare and Goldmann IOP readings were within 1 mm Hg of one another in 8 of 14 eyes (57%) and 3 mm Hg of one another (a clinically similar IOP) in 10 of 14 eyes (71%). In the remaining 4 eyes, IOP recorded by iCare was higher than Goldmann applanation by greater than 3 mm Hg in 3 eyes (3/14; 21%) and lower than Goldmann applanation by more than 3 mm Hg in just 1 eye (1/14; 7%).
Diurnal IOP Fluctuation
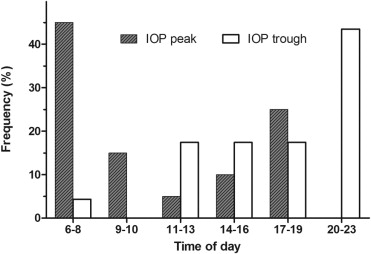
Of the 17 subjects with known or suspected glaucoma, 13 subjects (13 eyes) had data analyzable for evaluation of diurnal IOP fluctuation ( Figure 1 ). Of the 4 subjects (4 eyes) excluded from this analysis, 1 had IOP missing from more than 2 time periods during the 24-hour period, and 3 were missing early morning IOP measurements on both days of data collection. The diurnal curve had a relative morning peak and a secondary peak in the afternoon. The normal control eyes (11 subjects, 11 eyes) followed a similar overall pattern of diurnal IOP ( Figure 1 ). Of the 17 subjects with known or suspected glaucoma, 12 subjects (12 eyes) had data at every time interval for evaluation of peak and trough IOP ( Figure 2 ); using incidence data, relative peak and trough IOPs appeared to occur in the early morning (45%) and late evening (43.5%), respectively ( Figure 2 ). In addition to the 4 subjects (4 eyes) excluded from the diurnal IOP fluctuation analysis above, 1 additional subject (1 eye) was excluded due to IOP fluctuation less than 3 mm Hg during the 24-hour period.
Comparison of IOP Range Recorded During iCare Home Tonometry vs in the Clinic
Of the 17 subjects with known or suspected glaucoma, 16 were included in the comparison of IOP range between home and clinic tonometry. One child was excluded because the iCare tonometer was used to monitor early postoperative IOP fluctuation; therefore, there was no corresponding “clinic range” for comparison (see details in Case 4 presentation). The difference between peak IOP recorded during the iCare home tonometry and in the clinic was less than 4 mm Hg in 8 subjects (50%), between 4 and 6 mm Hg in 3 subjects (19%), and greater than 6 mm Hg in 5 subjects (31%) ( Figure 3 ). Comparison of the mean IOP measured by iCare home tonometry vs IOP measured in the clinic, and comparison of the peak IOP measured by iCare home tonometry vs IOP measured in the clinic, respectively, failed to achieve statistical significance (n = 16 eyes, P = .160 vs P = .201, respectively). The fluctuation of IOP (maximum IOP minus minimum IOP for a given subject) varied from case to case. For all subjects (n = 16 eyes), the smallest and largest clinic IOP fluctuations were 2 mm Hg and 22 mm Hg (mean = 11.6 ± 6.37), respectively; the corresponding smallest and largest iCare home IOP fluctuations were 8 mm Hg and 20 mm Hg (mean = 13.1 ± 4.25), respectively.




