Purpose
To investigate the clinical features, risk factors, and treatment outcomes following immunologic graft rejection in eyes that have undergone Descemet stripping automated endothelial keratoplasty (DSAEK).
Design
Retrospective case review.
Methods
The charts for 353 DSAEK procedures performed at a single clinical practice at the New York Eye and Ear Infirmary from August 2006 to November 2010 were reviewed. Cases with at least 3 months follow-up were included. Outcome measures included rates of graft rejection, clinical findings, treatment outcomes, and risk factor analysis.
Results
Thirty of 353 DSAEKs developed graft rejection (8.5%). Kaplan-Meier rate of rejection was 6.0% at 1 year (n = 175), 14.0% at 2 years (n = 79), and 22.0% at 3 years (n = 39). Rejection episodes occurred between 0.8 and 34 months. Clinical findings included anterior chamber cells, keratic precipitates, endothelial rejection line, and host-donor interface vascularization. Risk factors for development of graft rejection were cessation of postoperative steroid (hazard ratio 5.49, P < .0001) and black race (hazard ratio 2.71, P = .02). Recipient age, sex, surgical indication, glaucoma, postoperative steroid response, corneal neovascularization or peripheral anterior synechiae, graft size, prior keratoplasty in fellow eye, and concurrent or subsequent procedures were not associated with graft rejection. Twenty-two out of 30 rejection episodes (73.3%) resolved with steroid treatment.
Conclusions
Graft rejection is an important complication following DSAEK. In contrast to penetrating keratoplasty, rejection following DSAEK is almost exclusively endothelial. Among risk factors traditionally associated with graft rejection, cessation of topical steroids was most significant. Prompt recognition and treatment of DSAEK rejection can lead to favorable outcomes.
Descemet stripping automated endothelial keratoplasty (DSAEK) has gained popularity as an alternative to penetrating keratoplasty (PK) for patients with corneal endothelial failure. Compared to PK, DSAEK has demonstrated a reduced risk of wound dehiscence, intraoperative choroidal hemorrhage, and suture-related complications; lower postoperative astigmatism; and faster visual recovery. In addition, early literature suggests endothelial keratoplasty (EK) may confer a lower graft rejection rate compared to comparable cases of PK; however, the EK group studied included both deep lamellar endothelial keratoplasty and DSAEK cases. To date, there is only 1 series, from Price and associates, that has reported specifically on DSAEK rejection rates for up to 2 years of follow-up and identified certain risk factors for rejection, such as black race and history of glaucoma or postoperative steroid response. Herein, we report our 3-year results for DSAEK rejection rates, clinical features, and outcomes, and present an expanded risk factor analysis.
Methods
A retrospective chart review was performed of patients who underwent DSAEK from August 3, 2006 to November 30, 2010 with a minimum of 3 months follow-up at a clinical practice at the New York Eye and Ear Infirmary. The medical charts were reviewed for the following data: demographics, comorbid eye conditions, presence of corneal neovascularization or peripheral anterior synechiae, prior ocular surgery, indication for the DSAEK, concurrent procedures, use of postoperative steroids, subsequent procedures, and development of DSAEK rejection. Graft rejection was defined as presence of inflammation as evidenced by anterior chamber cells, keratic precipitates, or endothelial rejection line with or without concomitant corneal edema and/or the presence of corneal edema with conjunctival injection and symptoms of pain or light sensitivity. The medical charts of patients who experienced DSAEK rejection were further reviewed for pre-rejection visual acuity, rejection treatment regimen, and outcomes.
The probability of rejection with time was plotted using Kaplan-Meier survival analysis. For eyes that experienced rejection, time to event was determined as date from surgery to date when rejection was first noted. For eyes that remained rejection-free, the last visit date was used to determine follow-up. Cox proportional hazard regression was used to analyze the influence of known rejection risk factors from PK literature on the incidence of DSAEK rejection. These variables included recipient age, race, indication for the DSAEK, presence of glaucoma, postoperative steroid response, corneal neovascularization, peripheral anterior synechiae, graft size, prior keratoplasty in the fellow eye, concurrent procedures, subsequent procedures, and cessation of postoperative steroids. P values of equal to or less than .05 were accepted as statistically significant. Variables that achieved statistical significance in the univariate analysis were further subjected to multivariate analysis to adjust for confounding effects. All statistical analysis was performed using the SAS 9.1 software (SAS Institute, Inc, Cary, North Carolina, USA).
Surgical Technique and Postoperative Management
All DSAEK procedures were performed as described previously. The donor lenticle was prepared by mounting the donor corneoscleral rim on a Moria ALTK artificial anterior chamber (Moria, Doylestown, Pennsylvania, USA) and sectioned using a microkeratome (ALT-CBm; Moria) with either a 350-μm or 300-μm head based on pachymetric measurements. A Hanna trephine (Moria) varying from 7.0 to 9.0mm was used to create the appropriate-sized donor lenticle based on the patient’s white-to-white measurements. A reverse Sinskey hook (Bausch & Lomb, San Dimas, California, USA) was used to score the Descemet membrane through a paracentesis tract. Midperipheral corneal venting incisions were made at the discretion of the surgeon. The donor lenticle was inserted through a 4.5- to 5.0-mm clear corneal wound with use of either the Busin glide (Moria) or Ogawa forceps (Moria), depending on surgeon preference. The lenticle was apposed to the host cornea by filling the anterior segment 90% with air. Interface fluid was aspirated from the venting incisions, if created. Concurrent surgeries (eg, cataract extraction, intraocular lens exchange, vitrectomy) were performed prior to introduction of the donor lenticle. All anterior chamber glaucoma drainage devices were repositioned to pars plana and all anterior chamber intraocular lenses were exchanged for posterior chamber lenses prior to the DSAEK procedure.
Postoperative management consisted of 1 week of topical fluoroquinolone and topical steroids, either prednisolone acetate 1% (Pred Forte; Allergan, Irvine, California, USA) or difluprednate 0.05% (Durezol; Alcon, Fort Worth, Texas, USA) every 2 hours while awake. Topical steroids were then tapered to 4 times daily for the remainder of month 1, 3 times daily for month 2, and twice daily for month 3, and maintained at once daily starting month 4. In some earlier cases, topical steroid was discontinued without precipitating factors at 4 months. In instances of elevated intraocular pressure (IOP >22 mm Hg or elevation of IOP ≥10 mm Hg compared to preoperative value) attributable to steroid response, the inciting steroid would be substituted with loteprednol etabonate 0.2% or 0.5% (Alrex or Lotemax; Bausch & Lomb, Rochester, New York, USA) or fluorometholone 0.25% (FML; Allergan) or discontinued entirely.
Results
From August 2006 to November 2010, 353 DSAEKs in 342 eyes with at least 3 months follow-up were identified. Mean age at time of DSAEK was 72 years (range 30–96). Mean follow-up was 16 ± 12 months (range 3–54 months). The main indications for DSAEK were Fuchs endothelial dystrophy (137/353; 38.8%), pseudophakic bullous keratopathy (109/353; 30.9%), and endothelial failure attributable to intraocular surgery (29/353; 8.2%). In 28 eyes (7.9%), the DSAEK was performed for a prior failed PK.
Thirty out of 353 DSAEK procedures (8.5%) developed graft rejection. Mean age of these patients at time of DSAEK procedure was 68 years (range 46–86). Mean follow-up was 23 ± 12 months (range 3–47 months). The indications for DSAEK in these patients were Fuchs endothelial dystrophy (12/30; 40.0%), pseudophakic bullous keratopathy (11/30; 36.7%), failed PK (4/30; 13.3%), endothelial failure attributable to intraocular surgery (2/30; 6.7%), and aphakic bullous keratoplasty (1/30; 3.3%).
Of the 30 patients, 25 (83.3%) presented with clinical symptoms, commonly blurry vision (43.3%), discomfort (36.7%), or redness (13.3%) ( Table 1 ). Clinical findings of graft rejection included diffuse keratic precipitates with corneal edema (63.3%), focal keratic precipitates without edema (23.3%), anterior chamber cells (23.3%), corneal edema with conjunctival injection (13.3%), and endothelial rejection line (10.0%). One patient developed keratic precipitates followed by extensive superficial and host-donor interface vascularization after stopping postoperative steroids ( Figure 1 ). For the 4 eyes presenting with corneal edema without anterior chamber inflammation, these episodes were considered graft rejection as all patients had discomfort and conjunctival injection; in 2, the edema resolved following steroid treatment.
| Number (%) of DSAEK Procedures (n = 30) | |
|---|---|
| No symptoms | 5 (16.7) |
| With symptoms | 25 (83.3) |
| Blurry vision | 13 (43.3) |
| Discomfort | 11 (36.7) |
| Redness | 4 (13.3) |
| Photosensitivity | 2 (6.7) |
| Tearing | 2 (6.7) |
| Clinical signs | |
| Keratic precipitates with edema | 19 (63.3) |
| Keratic precipitates without edema | 7 (23.3) |
| Anterior chamber cells | 7 (23.3) |
| Corneal edema with conjunctival injection | 4 (13.3) |
| Endothelial rejection line | 3 (10.0) |
| Vascularization | 1 (3.3) |
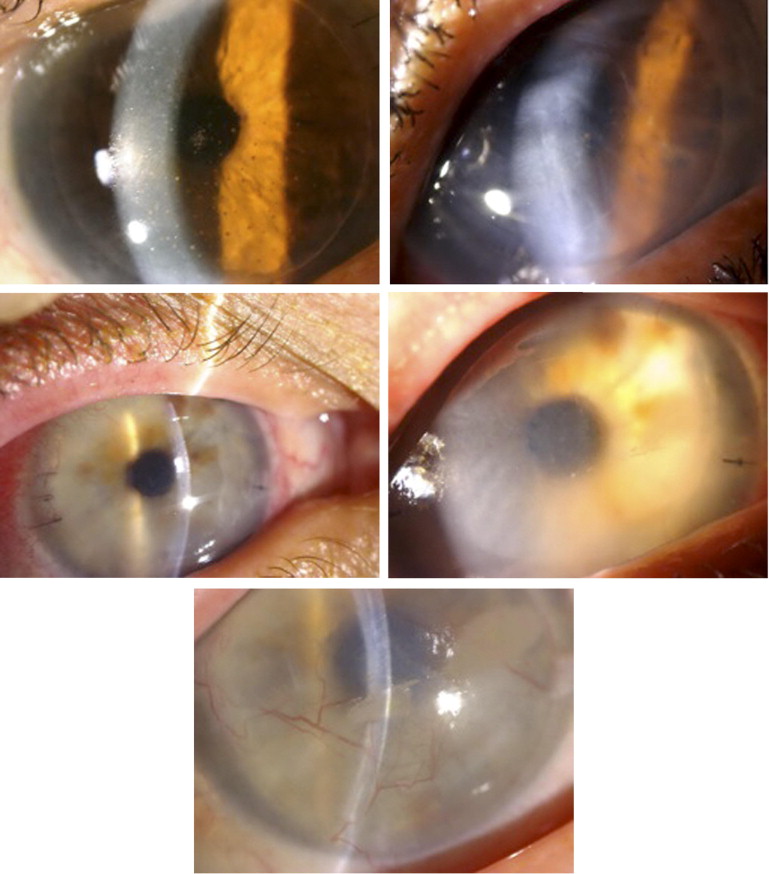
Graft rejection in the 30 eyes was observed within 1 month of the DSAEK procedure in 1 eye (3.3%), between 1 and 3 months in 4 eyes (13.3%), between 3 and 6 months in 4 eyes (13.3%), between 6 and 12 months in 6 eyes (20.0%), between 12 and 18 months in 9 eyes (30.0%), and beyond 18 months in 6 eyes (20.0%). Mean time to rejection following DSAEK was 13 ± 10 months (range 0.8–34 months). Kaplan-Meier survival curve is displayed in Figure 2 . The probability of developing rejection was 6.0% at 1 year (n = 175 eyes with at least 1 year follow-up), 14.0% at 2 years (n = 79 eyes with at least 2 years follow-up), and 22.0% at 3 years (n = 39 eyes with at least 3 years follow-up).
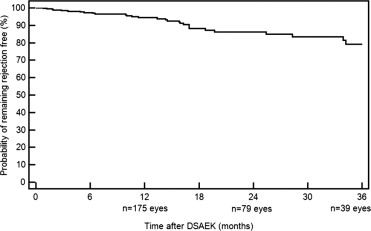
Table 2 shows rejection prevalence and unadjusted hazard ratio by demographics, surgical indication, absence or presence of glaucoma, postoperative steroid response, corneal vascularization, synechiae, graft size, prior keratoplasty (DSAEK or PK) in fellow eye, concurrent procedures, subsequent procedures, and whether postoperative topical steroids was discontinued.
| No. Rejected/No. of DSAEK | Rejection % | Unadjusted Hazard Ratio | 95% CI | P Value | |
|---|---|---|---|---|---|
| Sex | |||||
| Female | 18/204 | 8.8 | Reference | ||
| Male | 12/149 | 8.1 | 0.90 | 0.43–1.86 | .77 |
| Age | |||||
| ≥80 | 6/108 | 5.6 | 2.01 | 0.50–8.05 | .32 |
| 70–79 | 9/99 | 9.1 | 2.66 | 0.72–9.82 | .14 |
| 60–69 | 3/86 | 3.5 | Reference | ||
| 30–59 | 12/60 | 20.0 | 5.34 | 1.51–18.93 | .01 |
| Race | |||||
| White | 17/234 | 7.3 | Reference | ||
| Black | 10/52 | 19.2 | 2.69 | 1.23–5.88 | .01 |
| Other a | 3/67 | 4.5 | 0.64 | 0.19–2.19 | .48 |
| Indication | |||||
| Endothelial failure | 2/35 | 5.7 | Reference | ||
| Fuchs | 12/149 | 8.1 | 1.32 | 0.30–5.90 | .72 |
| PBK | 11/128 | 8.6 | 1.65 | 0.37–7.43 | .52 |
| Failed PK | 4/28 | 14.3 | 3.59 | 0.65–19.68 | .14 |
| Other b | 1/13 | 12.5 | 1.28 | 0.12–14.13 | .84 |
| Glaucoma | |||||
| No | 20/215 | 9.3 | Reference | ||
| Yes | 10/138 | 7.3 | 0.86 | 0.40–1.83 | .69 |
| Steroid response | |||||
| No | 23/285 | 8.1 | Reference | ||
| Yes | 7/68 | 10.3 | 1.10 | 0.47–2.56 | .83 |
| Vascularization | |||||
| No | 28/343 | 8.2 | Reference | ||
| Yes | 2/10 | 20.0 | 2.31 | 0.55–9.75 | .25 |
| Synechiae | |||||
| No | 26/320 | 8.1 | Reference | ||
| Yes | 4/33 | 12.1 | 1.63 | 0.57–4.68 | .36 |
| Graft ≥8.5 mm | |||||
| No | 10/133 | 8.9 | Reference | ||
| Yes | 20/240 | 8.3 | 1.38 | 0.63–3.01 | .42 |
| Prior DSAEK in fellow eye | |||||
| No | 24/303 | 7.9 | Reference | ||
| Yes | 6/50 | 12.0 | 1.66 | 0.67–4.09 | .27 |
| Prior PK in fellow eye | |||||
| No | 29/315 | 9.2 | Reference | ||
| Yes | 1/38 | 2.6 | 0.25 | 0.03–1.85 | .18 |
| Concurrent procedures | |||||
| No | 16/198 | 8.1 | Reference | ||
| Yes | 14/155 | 9.0 | 1.03 | 0.50–2.11 | .93 |
| Subsequent procedures | |||||
| No | 25/300 | 8.3 | Reference | ||
| Yes | 5/53 | 9.4 | 0.88 | 0.34–2.31 | .80 |
| Cessation of steroids | |||||
| No | 13/309 | 4.2 | Reference | ||
| Yes | 17/44 | 38.6 | 6.18 | 2.98–12.81 | <.0001 |
a Hispanic, Asian, and Indian did not occur in sufficient numbers to allow for subgroup analysis.
Young age (30–59 years of age), black race, and cessation of postoperative steroids achieved statistical significance in the univariate analysis and were mutually adjusted ( Table 3 ). Cessation of postoperative steroids emerged as the most significant risk factor associated with development of graft rejection (hazard ratio 5.49, 95% confidence interval [CI] 2.54–11.84, P < .0001). Kaplan-Meier survival analysis shows that when postoperative steroid was discontinued, probability of rejection was 17.0% at 1 year, 37.0% at 2 years, and 55.0% at 3 years, significantly higher than the 3.0%, 7.0%, and 10.0%, respectively if postoperative steroids were continued ( Figure 3 , Top). Overall, postoperative steroid was discontinued in 44 eyes (27/323 in the nonrejection group and 17/30 in the rejection group). Discontinuation was because of steroid response in 12 eyes (10 in the nonrejection group and 2 in the rejection group). For the remaining 32 eyes, steroids were stopped either by the physician without precipitating factor or by the patient because of noncompliance. Adjusting for development of steroid response did not alter the increased risk of rejection conferred by cessation of steroids (unadjusted hazard ratio 6.18, 95% CI 2.98–12.81, P < .0001; adjusted hazard ratio 6.17, 95% CI 2.98–12.76, P < .0001). Of the patients who were switched to a lower-potency steroid (loteprednol or fluorometholone) during their postoperative course, the percentage was comparable between the nonrejection and rejection group (15.2% vs 16.7%).



