Glaucoma
9.1 Primary Open Angle Glaucoma
Symptoms
Usually asymptomatic until the later stages. Symptoms may include visual field defects. Tunnel vision and loss of central fixation typically do not occur until late in the disease.
Signs
Intraocular pressure (IOP): Although most patients will have an elevated IOP (normal range of 10 to 21 mm Hg), nearly half have an IOP of 21 mm Hg or lower at any one screening.
Gonioscopy: Normal-appearing, open anterior chamber angle on gonioscopic evaluation. No peripheral anterior synechiae (PAS).
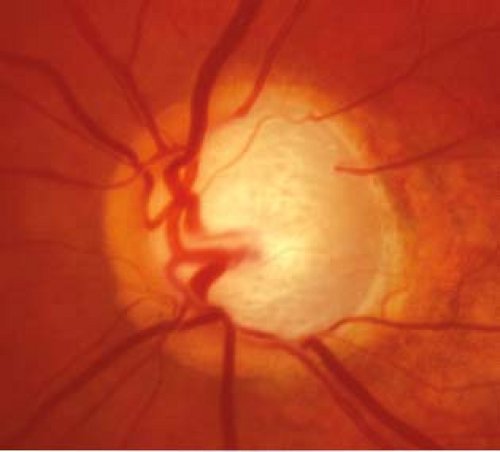
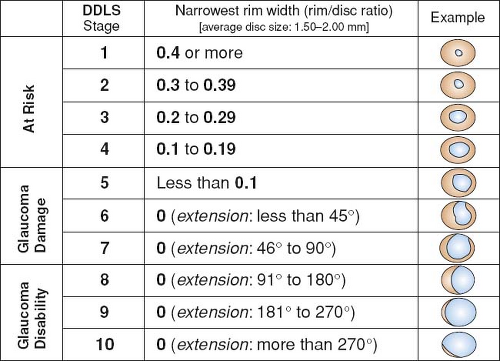
Optic nerve: See Figure 9.1.1. Characteristic appearance includes loss of rim tissue (includes notching; increased and/or progressive narrowing most commonly inferiorly followed by superiorly, more rarely nasally or temporally), splinter or nerve fiber layer hemorrhage that crosses the disc margin (Drance hemorrhage), acquired pit, nerve fiber layer defect, cup/disc (C/D) asymmetry >0.2 in the absence of a cause (e.g., anisometropia, different nerve sizes), bayoneting (sharp angulation of the blood vessels as they exit the nerve), enlarged C/D ratio (>0.6; less specific), progressive enlargement of the cup, greater Disc Damage Likelihood Scale (DDLS) score (see Figure 9.1.2).
Visual fields: Characteristic visual field loss patterns include nasal step, paracentral scotoma, arcuate scotoma extending from the blind spot nasally (defects usually respect the horizontal midline or are greater in one hemifield than the other), altitudinal defect, or generalized depression (see Figure 9.1.3).
Other. Large fluctuations in IOP, inter-eye IOP asymmetry >5 mm Hg, beta-zone peripapillary atrophy, absence of microcystic corneal edema, no ocular inflammation.
Differential Diagnosis
If Anterior Chamber Angle Open on Gonioscopy:
Ocular hypertension: Normal optic nerve and visual field. SEE 9.3, OCULAR HYPERTENSION.
Physiologic optic nerve cupping: Static enlarged C/D ratio without rim notching or visual field loss. Usually normal IOP and large optic nerve (> about 2 mm). Often familial.
Secondary open angle glaucoma: Identifiable cause for open angle glaucoma including inflammatory, exfoliative, pigmentary, steroid-induced, angle recession, traumatic (as a result of direct injury, blood, or debris), and glaucoma related to increased episcleral venous pressure (e.g., Sturge–Weber syndrome, carotid–cavernous fistula), intraocular tumors, degenerated red blood cells (ghost cell glaucoma), lens-induced, degenerated photoreceptor outer segments following chronic rhegmatogenous
retinal detachment (Schwartz–Matsuo syndrome), or developmental anterior segment abnormalities.
Low tension glaucoma: Same as primary open angle glaucoma (POAG) except normal IOP. SEE 9.2, LOW TENSION PRIMARY OPEN ANGLE GLAUCOMA (NORMAL PRESSURE GLAUCOMA).
Previous glaucomatous damage (e.g., from steroids, uveitis, glaucomatocyclitic crisis, trauma) in which the inciting agent has been removed. Nerve appearance now static.
Optic atrophy: Characterized by disproportionally more optic nerve pallor than cupping. IOP usually normal unless a secondary or unrelated glaucoma is present. Color vision and central vision are often decreased. Causes include tumors of the optic nerve, chiasm, or tract; syphilis, ischemic optic neuropathy, drugs, retinal vascular or degenerative disease, others. Visual field defects that respect the vertical midline are typical of intracranial lesions localized at the chiasm or posterior to it.
Congenital optic nerve defects (e.g., tilted discs, colobomas, optic nerve pits): Visual field defects may be present but are static.
Optic nerve drusen: Optic nerves not usually cupped and drusen often visible. Visual field defects may remain stable or progress unrelated to IOP. The most frequent defects include arcuate defects or an enlarged blind spot. Characteristic calcified lesions can be seen on B-scan ultrasonography (US) (as well as on computed tomography [CT]). Autofluorescence can also highlight nerve drusen.
If Closed or Partially Closed Angle on Gonioscopy:
Chronic angle closure glaucoma (CACG): Shallow anterior chamber, episodic blurred vision or headache. PAS present on gonioscopy. SEE 9.5, CHRONIC ANGLE CLOSURE GLAUCOMA.
Work-Up
History: Presence of risk factors (family history of blindness or visual loss from glaucoma, older age, African descent, diabetes, myopia, hypertension, or hypotension)? Previous history of increased IOP, chronic steroid use, or ocular trauma? Refractive surgery including laser in
situ keratomileusis (LASIK) in past (i.e., change in pachymetry)? Review of past medical history to determine appropriate therapy including asthma, chronic obstructive pulmonary disease (COPD), congestive heart failure, heart block or bradyarrhythmia, renal stones, allergies?
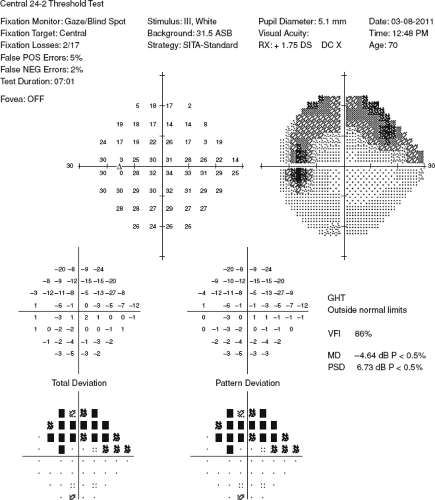
Figure 9.1.3 Humphrey visual field (HVF) showing a superior arcuate defect or scotoma of the left eye.
Baseline glaucoma evaluation: All patients with suspected glaucoma of any type should have the following:
Complete ocular examination including visual acuity, pupillary assessment for a relative afferent pupillary defect (RAPD), confrontational visual fields, slit lamp examination, applanation tonometry, gonioscopy, and dilated fundus examination (if the angle is open) with special attention to the optic nerve. Color vision testing is indicated if any suspicion of a neurologic disorder.
Baseline documentation of the optic nerves. May include meticulous drawings,
stereoscopic disc photos, red-free photographs, and/or computerized image analysis (e.g., optical coherence tomography [OCT] or Heidelberg retina tomography [HRT]) (see Figure 9.1.4).
Formal visual field testing (e.g., Humphrey automated visual field). Goldmann visual field tests may be helpful in patients unable to take the automated tests adequately.
Measure central corneal thickness (CCT). Corneal thickness variations affect apparent IOP as measured with applanation tonometry. Average corneal thickness is 535 to 545 microns. Thinner corneas tend to underestimate IOP, whereas thicker corneas tend to overestimate IOP. Of note, the relationship between corneal thickness and measured IOP is not exactly linear. A thin CCT is an
independent risk factor for the development of POAG.
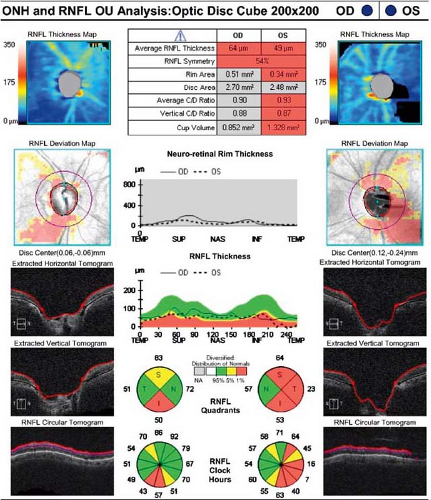
Figure 9.1.4 Optical coherence tomography of the optic nerve head (ONH) and retinal nerve fiber layer (RNFL) thickness.
Evaluation for other causes of optic nerve damage should be considered when any of the following atypical features are present:
Optic nerve pallor out of proportion to the degree of cupping.
Visual field defects greater than expected based on amount of cupping.
Visual field patterns not typical of glaucoma (e.g., defects respecting the vertical midline, hemianopic defects, enlarged blind spot, central scotoma).
Unilateral progression despite equal IOP in both eyes.
Decreased visual acuity out of proportion to the amount of cupping or field loss.
Color vision loss, especially in the red–green axis.
If any of these are present, further evaluation may include:
History: Acute episodes of eye pain or redness? Steroid use? Acute visual loss? Ocular trauma? Surgery, systemic trauma, heart attack, dialysis, or other event that may lead to hypotension?
Diurnal IOP curve consisting of multiple IOP checks during the course of the day.
Consider other laboratory work-up for nonglaucomatous optic neuropathy: Heavy metals, vitamin B12/folate, angiotensin-converting enzyme, antinuclear antibody, Lyme antibody, rapid plasma reagin (RPR) or Venereal Disease Research Laboratory (VDRL), and fluorescent treponemal antibody absorption (FTA-ABS) or treponemal-specific assay (e.g., MHA-TP). If giant cell arteritis (GCA) is a consideration, check erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and platelets (SEE 10.17, ARTERITIC ISCHEMIC OPTIC NEUROPATHY [GIANT CELL ARTERITIS]).
In cases where a neurologic disorder is suspected, obtain magnetic resonance imaging (MRI) of the brain and orbits with gadolinium and fat suppression if no contraindications present.
Check blood pressure, fasting blood sugar, hemoglobin A1c, lipid panel, and CBC (screening for anemia). Refer to an internist for a complete cardiovascular evaluation.
Treatment
General Considerations
Who to treat?
The decision to treat must be individualized. Some general guidelines are suggested.
Is the glaucomatous process present?
Damage is present if the DDLS score is >5 (see Figure 9.1.2) or there is visual field loss, the presence of an acquired pit or notched rims, IOP higher than 34 mm Hg, and/or IOP asymmetry more than 10 mm Hg.
Is the glaucomatous process active?
Determine the rate of damage progression by careful follow-up. Certain causes of cupping may be static (e.g., prior steroid response). Disc hemorrhages suggest active disease.
Is the glaucomatous process likely to cause disability?
Consider the patient’s age, overall physical and social health, as well as an estimation of his or her life expectancy.
What is the treatment goal?
The goal of treatment is to enhance or maintain the patient’s health by halting optic nerve damage while avoiding undue side effects of treatment. The only proven method of stopping or slowing optic nerve damage is reducing IOP. Reduction of IOP by at least 30% appears to have the best chance of preventing optic nerve damage. An optimal goal may be to reduce the IOP at least 30% below the threshold of progression. If damage is severe, greater reduction in IOP may be necessary.
How to treat?
The main treatment options for glaucoma include medications, laser trabeculoplasty (LT) (both argon [ALT] and selective [SLT]), and glaucoma surgery. Typically, medications are first-line therapy. Often ALT and SLT are also appropriate initial therapies, especially in patients at risk for poor compliance, with medication side effects, and who have significant trabecular meshwork (TM) pigmentation. Surgery may be appropriate initial treatment if damage is advanced in the setting of a rapid rate of progression or difficult follow-up.
Options include glaucoma filtering surgery (e.g., trabeculectomy), tube shunt procedures, laser cyclophotocoagulation of the ciliary body (e.g., with diode laser or endolaser), and cyclocryotherapy. Surgery should always be considered for any patient with advanced/progressive disease or IOP uncontrolled by other methods.
Medications
Unless there are extreme circumstances (e.g., IOP >40 mm Hg or impending loss of central fixation), treatment is often started by using one type of drop in one eye (monocular therapeutic trial) with reexamination in 3 to 6 weeks to check for effectiveness.
Prostaglandin agonists (e.g., latanoprost 0.005% q.h.s., bimatoprost 0.01% or 0.03% q.h.s., travoprost 0.004% q.h.s., tafluprost 0.0015% q.h.s. [preservative free]) are to be used with caution in patients with active uveitis or cystoid macular edema (CME) and are contraindicated in pregnant women or in women wishing to become pregnant. Inform patients of potential pigment changes in iris and periorbital skin, as well as hypertrichosis of eyelashes. Irreversible iris pigment changes rarely occur in blue or dark brown eyes; those at highest risk for iris hyperpigmentation have hazel, grey irides.
Beta-blockers (e.g., levobunolol or timolol 0.25% to 0.5% daily or b.i.d.) should be avoided in patients with asthma, COPD, heart block, bradyarrhythmia, unstable congestive heart failure, depression, or myasthenia gravis. In addition to bronchospasm and bradycardia, other side effects include hypotension, decreased libido, CNS depression, and reduced exercise tolerance.
Selective α2-receptor agonists (brimonidine 0.1%, 0.15%, or 0.2% b.i.d. to t.i.d.) are contraindicated in patients taking monoamine oxidase inhibitors (risk of hypertensive crisis) and relatively contraindicated in children under the age of 5 (risk for cardiorespiratory and CNS depression). SEE 8.11, CONGENITAL/INFANTILE GLAUCOMA. Apraclonidine 0.5% or 1% may be used for short-term therapy (3 months), but tends to lose effectiveness and has a high allergy rate.
Topical carbonic anhydrase inhibitors (CAIs) (e.g., dorzolamide 2% or brinzolamide 1% b.i.d. to t.i.d.) should be avoided, but are not contraindicated, in patients with sulfa allergy. These medications theoretically could cause the same side effects as systemic CAIs, such as metabolic acidosis, hypokalemia, gastrointestinal symptoms, weight loss, paresthesias, and aplastic anemia. However, systemic symptoms from topical CAIs are extremely rare. There have been no reported cases of aplastic anemia from topical use. Corneal endothelial dysfunction may be exacerbated with topical CAIs; these medications should be used cautiously in patients with Fuchs corneal dystrophy and post keratoplasty.
Miotics (e.g., pilocarpine q.i.d.) are usually used in low strengths initially (e.g., 1% to 2%) and then built up to higher strengths (e.g., 4%). Commonly not tolerated in patients <40 years because of accommodative spasm. Miotics are usually contraindicated in patients with retinal holes and should be used cautiously in patients at risk for retinal detachment (e.g., high myopes and aphakes).
 NOTE: Pilocarpine is not routinely used at Wills Eye due to its adverse side effect profile including associated increased risk for uveitis and retinal detachment, possibility for miosis-induced angle closure, and symptoms such as headache.
NOTE: Pilocarpine is not routinely used at Wills Eye due to its adverse side effect profile including associated increased risk for uveitis and retinal detachment, possibility for miosis-induced angle closure, and symptoms such as headache.
Sympathomimetics (dipivefrin 0.1% b.i.d. or epinephrine 0.5% to 2.0% b.i.d.) rarely reduce IOP to the degree of the other drugs, but have few systemic side effects (rarely, cardiac arrhythmias). They often cause red eyes and may cause CME in aphakic patients.
Systemic CAIs (e.g., methazolamide 25 to 50 mg p.o. b.i.d. to t.i.d., acetazolamide 125 to 250 mg p.o. b.i.d. to q.i.d., or acetazolamide 500 mg sequel p.o. b.i.d.) are relatively contraindicated in patients with renal failure. Potassium levels must be monitored if the patient is taking other diuretic agents or digitalis. Side effects such as fatigue, nausea, confusion, and paresthesias are common. Rare, but severe, hematologic
side effects (e.g., aplastic anemia) and Stevens–Johnson syndrome have occurred. Allergy to sulfa drugs is not an absolute contraindication to the use of systemic CAIs, but extra caution should be exercised in monitoring for an allergic reaction. Intravenous forms of systemic CAIs (e.g., acetazolamide 250 to 500 mg i.v.) may be utilized if IOP decrease is urgent or if IOP is refractory to topical therapy.
Argon Laser Trabeculoplasty
In some patients, as previously defined, ALT may be used as first-line therapy. It has an initial success rate of 70% to 80%, dropping to 50% in 2 to 5 years.
Selective Laser Trabeculoplasty
The IOP-lowering effect of SLT is equivalent to ALT. SLT utilizes lower energy and causes less tissue damage, which has led to the suggestion that it may be repeatable.
Guarded Filtration Surgery
Trabeculectomy may obviate the need for medications. Adjunctive use of antimetabolites (e.g., mitomycin-C, 5-fluorouracil) may aid in the effectiveness of the surgery but increases the risk of complications (e.g., bleb leaks and hypotony).
Follow-Up
Patients are reexamined 4 to 6 weeks after starting a new β-blocker or prostaglandin or after ALT/SLT to evaluate efficacy. Topical CAIs, α-agonists, and miotics quickly reach a steady state, and a repeat examination may be performed at any time after 3 days.
Closer monitoring (e.g., 1 to 3 days) may be necessary when damage is severe and the IOP is high.
Once the IOP has been reduced adequately, patients are reevaluated in 3- to 6-month intervals for IOP and optic nerve checks.
Typically gonioscopy is performed annually, or more often as needed to assess angle anatomy.
Formal visual fields and optic nerve imaging (e.g., photographs, OCT, or HRT) are rechecked as needed, often about every 6 to 12 months. If IOP control is not thought to be adequate, visual fields may need to be repeated more often. Once stabilized, formal visual field testing can be repeated annually.
Dilated retinal examinations should be performed yearly.
If glaucomatous damage progresses, check patient compliance with medications before initiating additional therapy.
Patients must be questioned about side effects associated with their specific agent(s). They often do not associate eye drops with impotence, weight loss, lightheadedness, or other significant symptoms.
REFERENCES
Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: A randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open angle glaucoma. Arch Ophthalmol. 2002;120(6):701–713.
9.2 Low Tension Primary Open Angle Glaucoma (Normal Pressure Glaucoma)
Definition
POAG occurring in patients without IOP elevation.
Signs
Critical. SEE SIGNS IN 9.1, PRIMARY OPEN ANGLE GLAUCOMA, except IOP is consistently below 22 mm Hg. There appears to be a greater likelihood of optic disc hemorrhages. Some believe visual field defects are denser, more localized, and
closer to fixation. A dense nasal paracentral defect is typical.
closer to fixation. A dense nasal paracentral defect is typical.
Differential Diagnosis
POAG: IOP may be underestimated secondary to large diurnal fluctuations or thin corneas. SEE 9.1, PRIMARY OPEN ANGLE GLAUCOMA.
Shock-related optic neuropathy from previous episode of systemic hypotension (e.g., acute blood loss, myocardial infarction, coronary artery bypass surgery, arrhythmia). Visual field loss should not progress.
Intermittent IOP elevation (e.g., angle closure glaucoma, glaucomatocyclitic crisis).
Previous glaucomatous insult with severe IOP elevation that has subsequently resolved. Nonprogressive (e.g., traumatic glaucoma, steroid-induced glaucoma).
Nonglaucomatous optic neuropathy and others. SEE DIFFERENTIAL DIAGNOSIS IN 9.1, PRIMARY OPEN ANGLE GLAUCOMA.
Etiology
Controversial. Most investigators believe that IOP plays an important role in low tension POAG. Other proposed etiologies include vascular dysregulation (e.g., systemic or nocturnal hypotension, vasospasm, or loss of autoregulation), microischemic disease, accelerated apoptosis, and autoimmune disease.
Work-Up
SEE WORK-UP IN 9.1, PRIMARY OPEN ANGLE GLAUCOMA. Also consider:
History: Evidence of vasospasm (history of migraine or Raynaud phenomenon)? History of hypotensive crisis, anemia, or heart disease? Previous use of corticosteroids by any route? Trauma or uveitis in past? Has the vision loss been acute or chronic? GCA symptoms? Additional cardiovascular risk factors such as elevated cholesterol, hypertension, and systemic hypotension (including nocturnal “dippers” in the early morning hours)?
Check color plates.
Check gonioscopy to rule out angle closure, angle recession, or PAS.
Consider obtaining a diurnal curve of IOP measurements to help confirm the diagnosis.
Consider carotid dopplers to evaluate ocular blood flow. Check blood pressure (consider 24-hour automated blood pressure home monitor).
Consider CT or MRI to rule out compressive lesions of the optic nerve or chiasm.
Treatment
Research suggests that further lowering of IOP plays an important role in preventing progression of low tension POAG. Target IOPs are at least 30% lower than the level at which progressive damage was occurring. Therapies are those for POAG. SEE 9.1, PRIMARY OPEN ANGLE GLAUCOMA, for a more in-depth discussion of these therapies. There is evidence that initial therapy with brimonidine 0.2% b.i.d. is superior to timolol 0.5% b.i.d. in preventing visual field progression in low tension POAG.
Ischemia to the optic nerve head may play a role in the pathogenesis of low tension POAG. Modification of cardiovascular risk factors is appropriate in managing general health, but has not proven beneficial in managing glaucoma. Refer to an internist for control of blood pressure, cholesterol, and optimal management of other comorbid conditions to maximize optic nerve perfusion. If possible avoid use of antihypertensive drugs at bedtime and use preferentially in the morning.
9.3 Ocular Hypertension
Signs
Critical. Generally defined as IOP >21 on two or more visits. Normal-appearing, open anterior chamber angle with normal anatomy on gonioscopy. Apparently normal optic nerve and visual field.
Differential Diagnosis
Work-Up
SEE 9.1, PRIMARY OPEN ANGLE GLAUCOMA.
If any abnormalities are present on formal visual field testing, consider repeat testing in 2 to 4 weeks to exclude the possibility of learning curve artifacts. If the defects are judged to be real, the diagnosis is glaucoma or ocular hypertension along with another pathology accounting for the field loss.
OCT and HRT may reveal glaucomatous optic nerve defects. These objective tests may show pathology earlier than visual field testing.
Treatment
If there are no suggestive optic nerve or visual field changes and IOP is <24 mm Hg, no treatment other than close observation is usually necessary.
Patients with an IOP >24 to 30 mm Hg but with normal examinations are candidates for IOP-lowering therapy (threshold varies per glaucoma specialist). A decision to treat a patient should be based on the patient’s choice to elect therapy and baseline risk factors such as age, CCT, initial IOP, optic nerve appearance, and family history. The results of the ocular hypertension treatment study (OHTS) showed that treatment reduced the development of visual field loss from 9.5% to 4.4% at 5 years, with a 20% average reduction of IOP. If treatment is elected, a therapeutic trial in one eye, as described for treatment of POAG, should be used. Some clinicians may elect to monitor these patients with close observation. Risk calculators have been developed to approximate the level of risk progression to glaucoma if left untreated. These may help guide clinicians and patients as to whether treatment should be initiated. SEE 9.1, PRIMARY OPEN ANGLE GLAUCOMA.
Follow-Up
Close follow-up is required for patients being treated and observed. All patients should initially be followed similarly to POAG; SEE 9.1, PRIMARY OPEN ANGLE GLAUCOMA. If there is no progression in the first few years, monitoring frequency can be decreased to every 6 to 12 months. Stopping medication may be considered in patients who have been stable for several years to reassess the need for continued treatment.
REFERENCES
Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: A randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of POAG. Arch Ophthalmol. 2002;120:701–713.
9.4 Acute Angle Closure Glaucoma
Symptoms
Pain, blurred vision, colored halos around lights, frontal headache, nausea, and vomiting.
Signs
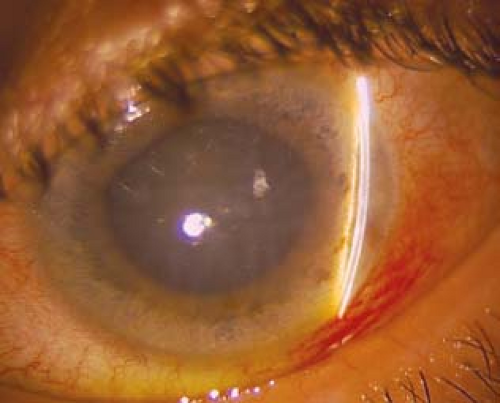
(See Figure 9.4.1.)
Critical. Closed angle in the involved eye, acutely increased IOP, microcystic corneal edema. Narrow or occludable angle in the fellow eye if of primary etiology.
Other. Conjunctival injection; fixed, mid-dilated pupil.
Etiology of Primary Angle Closure
Pupillary block: Apposition of the lens and the posterior iris at the pupil leads to blockage of aqueous humor flow from the posterior chamber to the anterior chamber. This mechanism leads to increased posterior chamber pressure, forward movement of the peripheral iris, and subsequent obstruction of the TM. Predisposed eyes have a narrow anterior chamber angle recess, anterior iris insertion of the iris root, or short axial length. Risk factors include increased age, East Asian descent, female gender, hyperopia, and family history.
May be precipitated by topical mydriatics or, rarely, miotics, systemic anticholinergics (e.g., antihistamines and antidepressants), accommodation (e.g., reading), or dim illumination. Fellow eye has similar anatomy.
Angle crowding as a result of an abnormal iris configuration including high peripheral iris roll or plateau iris syndrome angle closure. SEE 9.13, PLATEAU IRIS.
Etiology of Secondary Angle Closure
PAS pulling the angle closed: Causes include uveitis, inflammation, argon laser trabeculoplasty. SEE 9.5, CHRONIC ANGLE CLOSURE GLAUCOMA.
Neovascular or fibrovascular membrane pulling the angle closed: SEE 9.14, NEOVASCULAR GLAUCOMA.
Membrane obstructing the angle: Causes include endothelial membrane in iridocorneal endothelial syndrome (ICE) and posterior polymorphous corneal dystrophy (PPMD), and epithelial membrane in epithelial downgrowth (often follows penetrating and nonpenetrating trauma). SEE 9.15, IRIDOCORNEAL ENDOTHELIAL SYNDROME.
Lens-induced narrow angles: Iris–TM contact as a result of a large lens (phacomorphic), small lens with anterior prolapse (e.g., microspherophakia), small eye (nanophthalmos), or zonular loss/weakness (e.g., trauma, advanced pseudoexfoliation, Marfan syndrome).
Aphakic or pseudophakic pupillary block: Iris bombé configuration secondary to occlusion of the pupil by the anterior vitreous or fibrous adhesions. May also occur with anterior chamber intraocular lenses.
Topiramate and sulfonamide-induced angle closure: Usually after increase in dose or within first 2 weeks of starting medication. Usually bilateral angle closure due to supraciliary effusion and ciliary body swelling with subsequent anterior rotation of the lens–iris diaphragm. Myopia is induced secondary to anterior displacement of ciliary body and lens along with lenticular swelling.
Choroidal swelling: Following extensive retinal laser surgery, placement of a tight encircling scleral buckle, retinal vein occlusion, and others.
Posterior segment tumor: Malignant melanoma, retinoblastoma, ciliary body tumors, and others. SEE 11.36, CHOROIDAL NEVUS AND MALIGNANT MELANOMA OF THE CHOROID.
Hemorrhagic choroidal detachment: SEE 11.27, CHOROIDAL EFFUSION/DETACHMENT.
Aqueous misdirection syndrome. SEE 9.17, AQUEOUS MISDIRECTION SYNDROME/MALIGNANT GLAUCOMA.
Developmental abnormalities: Axenfeld–Rieger syndrome, Peters anomaly, persistent fetal vasculature, and others. SEE 8.12, DEVELOPMENTAL ANTERIOR SEGMENT AND LENS ANOMALIES/DYSGENESIS.
Differential Diagnosis of Acute IOP Increase With an Open Angle
Inflammatory open angle glaucoma: SEE 9.7, INFLAMMATORY OPEN ANGLE GLAUCOMA.
Traumatic (hemolytic) glaucoma: Red blood cells in the anterior chamber. SEE 3.6, HYPHEMA AND MICROHYPHEMA.
Pigmentary glaucoma: Characteristic angle changes, (4+ posterior TM band); vertical pigment deposition on endothelium; pigment cells floating in the anterior chamber; radial iris transillumination defects (TIDs); pigment line on the posterior lens capsule or anterior hyaloid face. SEE 9.10, PIGMENT DISPERSION SYNDROME/PIGMENTARY GLAUCOMA.
Pseudoexfoliation glaucoma: Grayish-white flaky proteinaceous material deposited throughout anterior segment structures and TM (usually irregular pigment most prominent inferiorly). Classically occurs in patients of European descent. Iris TIDs along pupillary margin often present. SEE 9.11, PSEUDOEXFOLIATION SYNDROME/EXFOLIATIVE GLAUCOMA.
Glaucomatocyclitic crisis (Posner–Schlossman syndrome): Recurrent IOP spikes in one eye, mild cell, and flare with or without fine keratic precipitates (KP). SEE 9.8, GLAUCOMATOCYCLITIC CRISIS/POSNER–SCHLOSSMAN SYNDROME.
Retrobulbar hemorrhage or inflammation. SEE 3.10, TRAUMATIC RETROBULBAR HEMORRHAGE.
Carotid–cavernous fistula: SEE 7.7, MISCELLANEOUS ORBITAL DISEASES.
Work-Up
History: Risk factors including hyperopia or family history? Precipitating events such as being in dim illumination, receiving dilating drops? Retinal problem? Recent laser treatment or surgery? Medications (e.g., topical adrenergics or anticholinergics, oral topiramate, or sulfa medications)?
Slit lamp examination: Look for KP, posterior synechiae, iris atrophy or neovascularization (NV), a mid-dilated and sluggish pupil, a swollen lens, anterior chamber cells and flare or iridescent particles, and a shallow anterior chamber. Glaukomflecken (small anterior subcapsular lens opacities) and atrophy of the iris stroma indicate prior attacks. Always carefully examine the other eye and compare.
Measure IOP.
Gonioscopy of both anterior chamber angles. Corneal edema can be cleared by using topical hyperosmolar agents (e.g., glycerin). Gonioscopy of the involved eye after IOP reduction is essential in assessment of the persistence and extent of angle closure; also needed to evaluate for the presence of NV.
Careful examination of the fundus looking for signs of central retinal vein occlusion, hemorrhage, optic nerve cupping, or spontaneous arterial pulsations which may indicate an exacerbation of IOP elevation. If cupping is pronounced or if there are spontaneous arterial pulsations, treatment is more urgent.
When secondary angle closure glaucoma is suspected, B-scan US or US biomicroscopy (UBM) may be helpful.
Treatment
Depends on etiology of angle closure, severity, and duration of attack. Severe, permanent damage may occur within several hours. If visual acuity is hand motion or better, IOP reduction is usually urgent; therapeutic intervention should include all topical glaucoma medications, systemic (preferably intravenous) CAI, and in some cases intravenous osmotic agent (e.g., mannitol) as long as not contraindicated. SEE 9.14, NEOVASCULAR GLAUCOMA, 9.16, POSTOPERATIVE GLAUCOMA, AND 9.17, AQUEOUS MISDIRECTION SYNDROME/MALIGNANT GLAUCOMA.
Compression gonioscopy is essential to determine if the trabecular blockage is reversible and may break an acute attack.
Topical therapy with β-blocker ([e.g., timolol 0.5%] caution with asthma or COPD), α-2 agonist (e.g., brimonidine 0.1%), prostaglandin analog (e.g., latanoprost 0.005%), and CAI (e.g., dorzolamide 2%) should be initiated immediately. In urgent cases, three rounds of these medications may be given, with each round being separated by 15 minutes.
Topical steroid (e.g., prednisolone acetate 1%) may be useful.
Systemic CAI (e.g., acetazolamide 250 to 500 mg i.v. or two 250-mg tablets p.o. in one dose if unable to give i.v.) if reduction in IOP is urgent or if IOP is refractory to topical therapy. Do not use in sulfonamide-induced angle closure.
Recheck the IOP in 1 hour. If IOP reduction is urgent or refractory to therapies listed above, repeat topical medications and give osmotic agent (e.g., mannitol 1 to 2 g/kg i.v. over 45 minutes [a 500 mL bag of mannitol 20% contains 100 g of mannitol]). Contraindicated in congestive heart failure, renal disease, and intracranial bleeding.
 NOTE: Prior to initiation of systemic CAIs or osmotic agents, renal function should be tested.
NOTE: Prior to initiation of systemic CAIs or osmotic agents, renal function should be tested.
When acute angle closure glaucoma is the result of:
Phakic pupillary block or angle crowding: Historically, pilocarpine, 1% to 2%, every 15 minutes for two doses was recommended but has fallen out of favor by some physicians due to adverse effects such as headache, accommodative spasm, associated increased risk for uveitis and retinal detachment, and potential for miosis-induced angle closure.
Aphakic or pseudophakic pupillary block or secondary closure of the angle: Do not
use pilocarpine. Consider a mydriatic and a cycloplegic agent (e.g., cyclopentolate 1% to 2%, and phenylephrine 2.5% every 15 minutes for four doses) when laser or surgery cannot be performed because of corneal edema, inflammation, or both.
Topiramate- or sulfonamide-induced secondary angle closure: Do not use CAIs in sulfonamide-induced angle closure. Immediately discontinue the inciting medication. Consider cycloplegia to induce posterior rotation of the ciliary body (e.g., atropine 1% b.i.d. or t.i.d.). Consider hospitalization and treatment with intravenous hyperosmotic agents and intravenous steroids (methylprednisolone 250 mg i.v. every 6 hours) for cases of markedly elevated IOP unresponsive to other treatments. Cases involving large ciliochoroidal or choroidal effusions may benefit from intravenous corticosteroids, as inflammation may play a role in their formation. Peripheral iridotomy (PI) or iridectomy and miotics are not indicated.
In phacomorphic glaucoma, the lens should be removed as soon as the eye is quiet and the IOP controlled, if possible. SEE 9.12.4, PHACOMORPHIC GLAUCOMA.
Address systemic problems such as pain and vomiting.
For pupillary block (all forms) or primary angle crowding: If the IOP decreases significantly, definitive treatment with laser (YAG) PI or surgical iridectomy is performed once the cornea is clear and the anterior chamber is quiet, typically 1 to 5 days after attack.
 NOTE: If affected eye is too inflamed initially for laser PI, perform laser PI of the fellow eye first. An untreated fellow eye has a 40% to 80% chance of acute angle closure in 5 to 10 years. Repeated angle closure attacks with a patent PI may indicate plateau iris syndrome. SEE 9.13, PLATEAU IRIS.
NOTE: If affected eye is too inflamed initially for laser PI, perform laser PI of the fellow eye first. An untreated fellow eye has a 40% to 80% chance of acute angle closure in 5 to 10 years. Repeated angle closure attacks with a patent PI may indicate plateau iris syndrome. SEE 9.13, PLATEAU IRIS.
Patients are discharged on a regimen of maintenance dose IOP-lowering drops and oral medications (described above). Close monitoring with IOP measurement each day is necessary immediately after an angle closure attack. Once the IOP has been reasonably reduced, follow-up frequency is guided by overall clinical response and stability.
 NOTE: If IOP does not decrease after two courses of maximal medical therapy, a laser (YAG) PI should be considered if there is an adequate view of the iris. If IOP still does not decrease after more than one attempt at laser PI, then a laser iridoplasty or surgical PI is needed. If all preceding treatments are unsuccessful or refractory, a guarded filtration procedure should be considered.
NOTE: If IOP does not decrease after two courses of maximal medical therapy, a laser (YAG) PI should be considered if there is an adequate view of the iris. If IOP still does not decrease after more than one attempt at laser PI, then a laser iridoplasty or surgical PI is needed. If all preceding treatments are unsuccessful or refractory, a guarded filtration procedure should be considered.
For secondary angle closure: Treat the underlying problem. Consider argon laser gonioplasty to open the angle, particularly in plateau iris syndrome or nanophthalmos. Goniosynechiolysis can be performed for chronic angle closure of <6 months’ duration. Systemic steroids may be required to treat serous choroidal detachments secondary to inflammation.
Follow-Up
After definitive treatment, patients are reevaluated in weeks to months initially, and then less frequently. Visual fields and stereo disc photographs are obtained for baseline purposes.
Cardiovascular status and electrolyte balance must be considered when contemplating osmotic agents, CAIs, and β-blockers.
The corneal appearance may worsen when the IOP decreases.
Worsening vision or spontaneous arterial pulsations are signs of increasing urgency for pressure reduction.
Since one-third to one-half of first-degree relatives may have occludable angles, patients should be counseled to alert relatives to the importance of screening.
Angle closure glaucoma may be seen without an increased IOP. The diagnosis should be suspected in a patient who had episodes of pain and reduced acuity and is noted to have:
An edematous, thickened cornea.
Normal or markedly asymmetric pressure in both eyes.
Shallow anterior chambers in both eyes.
Occludable anterior chamber angle in the fellow eye.
9.5 Chronic Angle Closure Glaucoma
Symptoms
Usually asymptomatic, although patients with advanced disease may present with decreased vision or visual field loss. Intermittent eye pain, headaches, and blurry vision may occur.
Signs
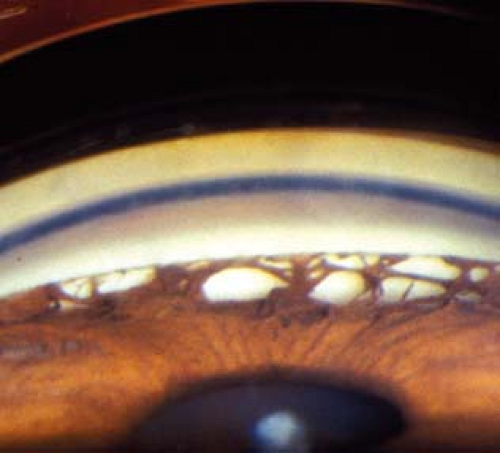
(See Figure 9.5.1.)
Critical. Gonioscopy reveals broad bands of PAS in the angle. The PAS block visualization of the underlying structures of the angle. Glaucomatous optic nerve and visual field defects.
Other. Elevated IOP.
Etiology
Prolonged acute angle closure glaucoma or multiple episodes of subclinical attacks of acute angle closure resulting in PAS, often in superior angle.
Previous uveitic glaucoma with development of PAS, often in inferior angle.
Regressed NV of the anterior chamber angle resulting in the development of PAS.
Previous laser to the TM (ALT), typically seen with small peaked PAS.
Previous flat anterior chamber from surgery, trauma, or hypotony that resulted in the development of PAS.
Work-Up
History: Presence of symptoms of previous episodes of acute angle closure? History of proliferative diabetic retinopathy, retinal vascular occlusion, or ocular ischemic syndrome? History of trauma, hypotony, uveitis, or laser treatment?
Complete baseline glaucoma evaluation. SEE 9.1, PRIMARY OPEN ANGLE GLAUCOMA.
Treatment
SEE 9.1, PRIMARY OPEN ANGLE GLAUCOMA.
Laser trabeculoplasty contraindicated in CACG prior to PI and can induce greater scarring of the angle.
If patient has angle closure with pupillary block, perform laser PI to prevent further angle closure episodes. The TM may have sustained enough damage that the IOP will still be elevated despite a patent iridotomy, necessitating continued use of medications to lower the IOP.
Laser iridoplasty may be performed (and repeated) to decrease the formation of new PAS. May not be effective, or may serve as only a temporary measure. If iridoplasty fails and other medical therapy has been maximized, the patient may need additional surgery. ECP may be attempted in cases with early glaucoma, but trabeculectomy or tube shunt is usually indicated in more advanced cases. MIGS usually contraindicated due to closed angle.
9.6 Angle Recession Glaucoma
Symptoms
Usually asymptomatic. Late stages have visual field or acuity loss. A history of hyphema or trauma to the glaucomatous eye can often be elicited. Glaucoma due to the angle recession (not from the trauma that caused the angle recession) usually takes 10 to 20 years to develop. Typically unilateral.