Purpose
To describe fundus autofluorescence (FAF) patterns of myopic choroidal neovascularization (CNV) treated with intravitreal ranibizumab and their correlation with visual acuity.
Design
Prospective interventional case series.
Methods
Twenty-seven eyes (27 patients) affected by myopic CNV were enrolled from January 2011 to January 2013. All patients underwent a complete ophthalmologic examination, including best-corrected visual acuity (BCVA) determination and fundus autofluorescence (FAF). The patients underwent ranibizumab injections following a pro re nata treatment regimen. The main outcome measure was the identification of the FAF patterns of myopic CNV over a 12-month follow-up. The secondary outcome was the correlation of the FAF patterns with the BCVA.
Results
At baseline 17 eyes (63%) showed a hyperautofluorescent pattern and 10 eyes (37%) a patchy pattern. BCVA changed from 0.48 ± 0.23 (logMAR) to 0.30 ± 0.32 at the 12-month examination ( P = .027) in the hyper-FAF subgroup. In the subgroup showing the patchy pattern, the BCVA declined slightly from 0.51 ± 0.27 to 0.56 ± 0.37 ( P = .53). The 14 eyes preserving the hyper-FAF pattern during the follow-up had a final BCVA of 0.20 ± 0.17, whereas the 9 eyes maintaining the patchy pattern showed a final BCVA of 0.60 ± 0.37 ( P = .002). The atrophic area of the retinal pigment epithelium assessed on the basis of FAF increased from 1.27 ± 2.80 mm 2 to 1.83 ± 3.74 mm 2 at the 12-month examination ( P = .016). The mean atrophic area increased by 0.37 mm 2 in the hyper-FAF subgroup and by 0.90 mm 2 in the patchy FAF subgroup.
Conclusions
Two main patterns were identified on FAF in myopic CNV and were related to the prognostic evolution, the hyperautofluorescent CNV being associated with a greater visual gain and fewer atrophic changes over a 12-month follow-up.
The fundus autofluorescence (FAF) of eyes affected by pathologic myopia may be very complex owing to the various alterations typical of the condition. The FAF pattern of choroidal neovascularization (CNV) associated with pathologic myopia has seldom been investigated. Generally speaking, the FAF of myopic CNV is most frequently characterized by a hyperautofluorescence signal, probably owing to the double retinal pigment epithelium (RPE) layer enveloping the type 2 CNV typical of pathologic myopia.
In addition, the progressive atrophic changes, related both to the natural history of the myopic CNV and to treatments, can lead to an increased deterioration of the RPE. Nonetheless, no study has specifically focused on the correlation between the FAF response and anti–vascular endothelial growth factor (VEGF) treatment of myopic CNV.
The aim of the present study is to describe the FAF changes of subfoveal myopic CNV treated with intravitreal ranibizumab and its relationship with the visual outcomes.
Methods
The study is a prospective interventional case series. Patients consecutively referred to the Ophthalmology Department of the Vita-Salute University in Milan from January 2011 to January 2013 for subfoveal CNV secondary to pathologic myopia were prospectively recruited. Each patient was carefully informed about the purpose of the research and provided signed consent. The research adhered to the tenets of the Declaration of Helsinki and the institutional review board of Ospedale San Raffaele approved the study.
The inclusion criteria were as follows: spherical equivalent refractive error of −6.0 diopters or more (an eye that had a spherical equivalent under −6.0 diopters was eligible if there were chorioretinal abnormalities consistent with pathologic myopia, such as lacquer cracks, chorioretinal atrophy, and posterior staphyloma, and if the axial length of the eye was at least 26.5 mm); naïve subfoveal CNV with active dye leakage on fluorescein angiography (FA); and baseline best-corrected visual acuity (BCVA) between 20/400 and 20/32. The exclusion criteria included intraocular surgery of any kind within 6 months of the day of injection; any ocular disease able to confound a proper clinical examination, including vitreoretinal traction and myopic foveoschisis; ocular hypertension or glaucoma; uncontrolled systemic hypertension; peripheral vascular disease; a history of thromboembolism, ischemic heart disease, or stroke; and pregnancy.
Each patient underwent a complete monthly ophthalmologic examination, including BCVA assessment on ETDRS charts, slit-lamp examination, tonometry, dilated fundus evaluation, fluorescein angiography, spectral-domain optical coherence tomography (SD OCT), and FAF. FAF was obtained using a confocal scanning laser ophthalmoscope (Heidelberg Retinal Angiograph 2; Heidelberg Engineering, Heidelberg, Germany), with an excitation wavelength of 488 nm and a barrier filter of 500 nm. A series of 50–90 images were averaged to obtain a high-quality picture.
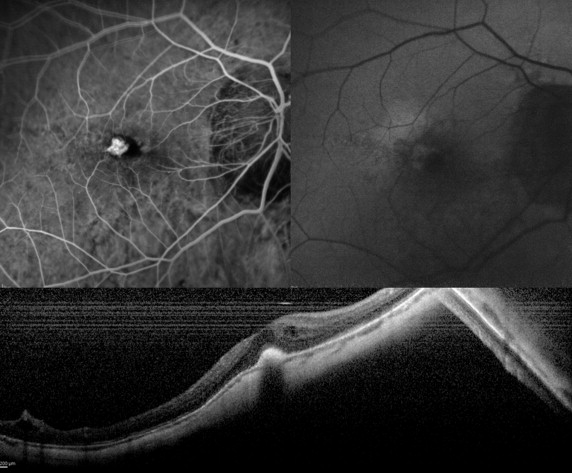
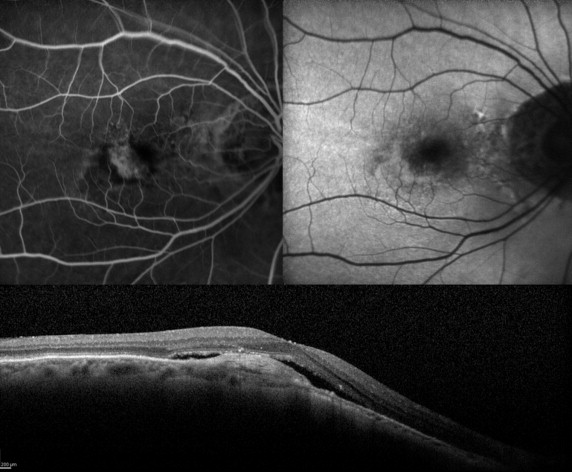
The FAF patterns of the CNV were divided into 2 main categories. The hyper-FAF pattern was characterized by an increased FAF signal corresponding to the area of the CNV, as identified on FA. The patchy FAF pattern was characterized by a variable combination of increased and decreased signals over the CNV area, as outlined by FA. In cases of CNV associated with retinal/subretinal hemorrhage, only the unmasked area of the CNV could be assessed by means of FAF.
Areas of low FAF signal corresponding to atrophic changes in the RPE were measured using the instrument’s image analysis software. Different types of atrophy were distinguished: enlargements of pre-existing atrophies and newly developed atrophies. BCVA was assessed on EDTRS charts by an examiner unaware of the purpose of the study. Two examiners well trained in FAF evaluation and masked to the purpose of the investigation (R.S. and P.I.) provided an independent appraisal of the FAF images.
FA and OCT examinations were carried out by means of HRT and SD OCT, respectively (Spectralis; Heidelberg Engineering). The study protocol required an initial ranibizumab injection and a subsequent monthly follow-up with pro re nata treatment regimen over a 12-month follow-up. Additional treatments were performed on the basis of the detection of leakage on FA and/or fluid on SD OCT.
The primary outcome measure was the identification of the FAF patterns of myopic CNV over a 12-month follow-up. The secondary outcomes included the correlation between each FAF pattern and the BCVA changes over the follow-up, and the correlation with the number of injections for each FAF pattern.
Statistical analyses were performed using SPSS Statistics Version 20 (IBM, Armonk, New York, USA). The Gaussian distribution of continuous variables was verified with the Kolmogorov-Smirnov test. Subgroup comparison of the mean BCVA, central foveal thickness (CFT), RPE atrophic area, and number of injections was performed using Student t test. Pearson correlation analysis examined the correlation between the number of injections and RPE atrophic area changes. In all analyses, P values <.05 were considered statistically significant.
Results
Twenty-seven patients with subfoveal myopic CNV (27 eyes) were recruited for the study. The mean age was 58.30 ± 15.7 years. Fourteen patients were female and 13 male.
In 8 eyes (30%) the CNV was associated with retinal/subretinal hemorrhage along its border. At baseline the mean BCVA was 0.49 ± 0.24 logMAR (approximately corresponding to 20/60 Snellen equivalent), whereas the mean CFT was 359 ± 106 μm. At the end of the follow-up the mean BCVA was 0.40 ± 0.21 logMAR overall (approximately corresponding to 20/50 Snellen equivalent) and the mean CFT was 346 ± 92 μm.
The main clinical data are listed in Tables 1 and 2 . Overall, the FAF pattern of the CNV at baseline turned out to be hyperautofluorescent in 17 eyes (63%), whereas 10 eyes (37%) showed a patchy pattern ( Figures 1 , 2 , and 3 ). BCVA changed from 0.48 ± 0.23 at baseline to 0.30 ± 0.32 at the 12-month examination ( P = .027) in the subgroup with CNV with hyper-FAF pattern at baseline. On the other hand, BCVA slightly declined from 0.51 ± 0.27 at baseline to 0.56 ± 0.37 at the end of the follow-up ( P = .53) in the subgroup showing a CNV patchy pattern. At the end of the follow-up 3 eyes starting with hyper-FAF of the CNV at baseline changed to a patchy pattern, whereas a single eye with patchy pattern at baseline changed to the hyper-FAF pattern.
| Baseline BCVA (logMAR) | Final BCVA (logMAR) | |
|---|---|---|
| Total | 0.49 | 0.40 |
| Hyper-FAF, 17 eyes (63%) | 0.48 | 0.30 |
| Patchy FAF, 10 eyes (37%) | 0.51 | 0.56 |
| Baseline Fundus Autofluorescence Pattern | Number of Patients (%) | Baseline Mean Atrophy Area (mm 2 ) | Final Mean Atrophy Area (mm 2 ) |
|---|---|---|---|
| Hyper-FAF | 17/27 (63%) | 1.33 | 1.70 |
| Patchy FAF | 10/27 (37%) | 1.16 | 2.06 |