Fundus Autofluorescence
Miki Sawa
R. Theodore Smith
Lawrence A. Yannuzzi
Retinal pigment epithelium (RPE) cells are essential to maintain normal photoreceptor function. The RPE has the important task of phagocytosing spent photoreceptor outer segment discs. However, in vivo imaging of the RPE is difficult due to low contrast and strong absorption by melanin pigments. Indirect information about the RPE cell layer can be obtained using fluorescein and indocyanine green angiography. However, functional assessment of the RPE is incomplete even if these techniques are performed. Recently, fundus autofluorescence (FAF) has been studied as a noninvasive test of RPE function based on the fluorescence characteristics of a curious waste material, lipofuscin, which deposits in the RPE over time (1, 2, 3, 4, 5, 6, 7, 8 and 9).
In vivo recording of FAF was described by Delori (1), who showed that FAF arose from lipofuscin in the RPE. Lipofuscin is derived in large part from phagocytosis of outer segment discs containing bisretinoid byproducts of light absorption. A number of retinoid-derived lipofuscin pigments have now been described (2,3,7 and 9), a major one being the molecule A2E (3), that is formed through a series of chemical reactions from two molecules of all-trans-retinal and one molecule of phosphatidylethanolamine. Precursors of A2E, specifically dihydroA2PE and A2PE, all of which are autofluorescent, form in the outer segments prior to phagocytosis by the RPE (4,7 and 9). A2E and its precursors are potentially susceptible to oxidative damage and capable of entering into photo-oxidative reactions with neighboring molecules (8). RPE in the macula contains a higher concentration of lipofuscin than nonmacular RPE (5). With aging, lipofuscin accumulates normally in the lysosomal compartment (1,5). It is also known to accumulate abnormally in various monogenetic and complex retinal diseases and is associated with photoreceptor degeneration (10). Lipofuscin is photoreactive and can produce a variety of reactive oxygen species and other radicals, may induce apoptosis of the RPE, and mediates blue light-induced RPE apoptosis (3,8).
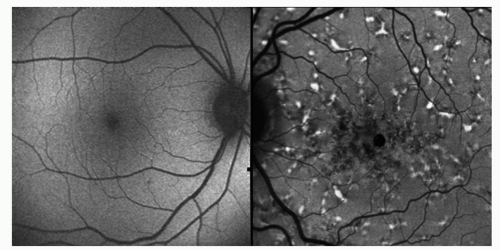
The emission of lipofuscin has a broad band ranging from 500 nm to 750 nm (1). The optimal excitation is 630 nm. The intensity of FAF parallels the amount and distribution of lipofuscin (1). The confocal scanning laser ophthalmoscope using the excitation wavelength 488 nm and a barrier filter of 500 nm is able to provide FAF imaging demonstrating AF distribution in vivo (10,11). Spaide et al. demonstrated FAF photography using a fundus camera-based system with a band pass filter for the excitation light of 580 nm and a barrier filter of 695 nm to avoid autofluorescence (AF) from the crystalline lens (12).
In the normal FAF pattern, diffuse AF is most intense between 5 degrees and 15 degrees from the fovea. The optic disc and retinal blood vessels have a low (dark) autofluorescent signal. Abnormal AF is classified into increased FAF (hyperautofluorescence) or decreased FAF (hypoautofluorescence) in comparison to the normal AF distribution or to the surrounding autofluorescent pattern. Decreased FAF is thought to indicate low lipofuscin concentration and can be due to RPE atrophy or RPE cell loss, or blocking effect due to overlying material such as hemorrhage. Increased FAF is thought to indicate excessive lipofuscin (Fig. 7-1). This manifestation of abnormal metabolism may result from high turnover of photoreceptor outer segments or an intrinsic defect in the ability of the RPE to recycle metabolites (10).
SPECIFIC DISEASES
Age-related Macular Degeneration, Geographic Atrophy, and Choroidal Neovascularization
The sub-RPE deposits known as drusen and accumulation of debris in Bruch’s membrane are believed to play an important
role in the pathogenesis of age-related macular degeneration (AMD). Drusen are a representative clinical manifestation of early AMD (13). In the earlier stages of the disease, large soft drusen and focally increased AF (FIAF) are spatially closely linked (See Fig. 7-2) (14, 15 and 16



role in the pathogenesis of age-related macular degeneration (AMD). Drusen are a representative clinical manifestation of early AMD (13). In the earlier stages of the disease, large soft drusen and focally increased AF (FIAF) are spatially closely linked (See Fig. 7-2) (14, 15 and 16
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree