Purpose
To evaluate 4-year treatment results of neovascular age-related macular degeneration with ranibizumab using a variable dosing regimen.
Design
Retrospective, single-center chart review.
Methods
This was a retrospective single-center study that included 855 patients with neovascular age-related macular degeneration receiving treatment with ranibizumab during a 4-year period. Included in the study were patients with a minimum follow-up of 15 months and all patients who terminated treatment regardless of follow-up.
Results
A total of 1321 patients were treated over the 4-year period, and 855 patients were eligible for inclusion. Of those, 456 patients were still receiving active treatment, whereas 399 patients had discontinued treatment. Overall treatment results showed a significant decrease in vision from 53.2 Early Treatment Diabetic Retinopathy Study letters (range, 1 to 85 letters) to 50.5 letters (range, 1 to 87 letters; P < .001). Mean follow-up was 23.3 months (range, 4 to 48 months). The reason for discontinuing treatment in 181 patients was no signs of activity, whereas 113 patients were judged to be nontreatable. Thirty-six patients declined further treatment for various reasons.
Conclusions
This report shows that when follow-up extends beyond 2 to 3 years, visual acuity does seem to decrease. Our data show that different responder groups can be identified: bad or nonresponders (approximately 15% of all patients) and good responders (approximately 21% of all patients). These 2 groups in general can be identified within the first 2 years of treatment, whereas the third group of regular responders (approximately 64% of all patients) require continuous monitoring and treatment for years.
Age-related macula degeneration (AMD) is the most common cause of blindness in the Western world. The prognosis for neovascular AMD has changed dramatically since the introduction of anti–vascular endothelial growth factor treatment. Hallmark randomized clinical trials have shown a marked effect of monthly injections with ranibizumab (Lucentis; Genetech, South San Francisco, California, USA) compared with placebo, and when the trials were extended to 24 months, there was a sustained treatment effect. However, because other studies have reported that pro re nata (PRN) treatment can obtain comparable treatment results and because monthly injections are not feasible, many retinal clinics, including ours, have adhered to a clinician-determined retreatment strategy, in which treatment is administered when there are signs of activity indicating choroidal neovascularization (CNV) on clinical follow-up.
Numerous reports have shown that anti–vascular endothelial growth factor treatment is effective in maintaining or improving visual acuity (VA) in real-world clinical practice using the PRN treatment strategy, but very few of those reports, and none with a long follow-up period, have given a detailed account of the cause of termination of treatment and how many patients obtain complete inactivation of the CNV. This information is highly relevant to the clinician when discussing treatment prognosis with the patient.
One recent study reported the causes of discontinuing treatment after 1 year among 101 (21.4%) of a total of 471 patients. Of the 471 patients included in the study, 1.2% declined further treatment, and in 7.2% of cases, the physician decided that no further follow-up was necessary, although it is unclear whether this was because of inactivation of the lesion or because the lesion was judged to be untreatable. In 13% of cases, the cause was ascribed to “other reasons.”
Treatment of neovascular AMD with ranibizumab has been performed at our department since 2007. In Denmark, treatment with Lucentis is covered by the Public National Health Insurance, which is a privilege extended to all citizens and is financed from general taxation. Herein, we present the results from a 4-year period, including a detailed account of the patients who stopped treatment. Our findings suggest that relatively few patients stop treatment because of inactivation, and that most patients should be considered as having chronically active disease.
Methods
This was a single-center study of 1321 patients with neovascular AMD who received treatment with ranibizumab in the 4-year period from April 2007 to April 2011. Included in the review of treatment results are patients with a minimum of 12 months of follow-up from the assessment visit after the third injection (minimum of 15 months of follow-up from initiation of treatment). All patients who terminated treatment in the 4-year period were included in the study, regardless of length of follow-up. Of the 1321 eyes, a total of 855 met the inclusion criteria ( Table 1 ).
| No. of Patients | Gender (Female/Male) | Mean Age (Range), y | Mean Starting Visual Acuity (Range), ETDRS Letters | Mean Visual Acuity at Latest Follow-up (Range), ETDRS Letters | Mean Follow-up (Range), mos | Mean No. of Injections with Ranibizumab (Range) | |
|---|---|---|---|---|---|---|---|
| All patients included | 855 | 537/318 | 77.3 (54 to 98) | 53.2 (1 to 85) | 50.5 (1 to 87) | 23.3 (4 to 48) | 8.7 (1 to 35) |
| Patients in active treatment | 456 | 291/165 | 76.0 (55 to 98) | 56.9 (1 to 85) | 57.8 (1 to 87) | 29.5 (15 to 48) | 11.3 (2 to 35) |
| Patients stopped treatment (total) a | 399 | 246/153 | 78.7 (54 to 96) | 48.9 (1 to 85) | 42.3 (1 to 87) | 16.1 (4 to 47) | 5.6 (1 to 22) |
| No signs of activity | 181 | 116/65 | 77.7 (54 to 95) | 50.8 (1 to 85) | 52.4 (5 to 87) | 18.6 (5 to 47) | 5.2 (1 to 17) |
| Did not want to receive further injections | 36 | 24/12 | 79.7 (61 to 96) | 50.3 (20 to 72) | 43.3 (8 to 75) | 11.7 (4 to 38) | 5.1 (2 to 18) |
| Judged nontreatable | 113 | 70/43 | 79.1 (60 to 95) | 43.8 (1 to 80) | 21.8 (1 to 69) | 15.1 (4 to 41) | 6.8 (2 to 22) |
| Other reasons | 69 | 36/33 | 80.3 (60 to 92) | 52 (5 to 82) | 48 (1 to 82) | 13.8 (4 to 45) | 6.1 (2 to 19) |
a The patients who terminated treatment are divided into the following subgroups: no signs of activity, did not want to receive further treatment, and judged nontreatable, and other reasons comprising death and the patient moving to another region.
Patients suspicious of neovascular AMD were examined with funduscopy, spectral-domain optical coherence tomography (Spectralis HRA-OCT; Heidelberg Engineering, Heidelberg, Germany), and fundus autofluorescence. Best-corrected visual acuity (BCVA) was determined using the Early Treatment Diabetic Retinopathy Study (ETDRS) chart. All patients underwent fluorescein angiography (FA), indocyanine green angiography (ICGA), or both. All patients with clinical signs of active neovascular AMD and a BCVA of more than 20 letters were offered treatment with ranibizumab. In some cases, treatment was offered although BCVA was fewer than 20 letters. Findings considered signs of activity were retinal hemorrhages, leakage on FA, and presence of subretinal or intraretinal fluid, or both, on OCT. Definite signs of active CNV under a pigment epithelial detachment (PED) was detected using ICGA and also was considered an indication for treatment, whereas only serous PEDs were monitored. All initial data including clinical findings were recorded in a database for treatment follow-up. The treatment initially was given as a loading dose using a series of 3 monthly injections. Very few patients received fewer injections initially. The first clinical follow-up took place 4 to 6 weeks after the third injection. At the follow-up, consultation measurement of BCVA using the ETDRS chart, funduscopy, and OCT was performed. At follow-up visits, FA and ICGA were not performed routinely and generally were reserved to cases with unusual treatment responses or unexpected morphologic changes. All the findings were recorded in the database. If there were signs of activity, the examining physician assessed the number of injections needed (1 to 3 injections). Minimal signs of activity (limited amount of fluid) could have indicated the need for only 1 additional injection, whereas extensive signs of activity resulted in 3 injections. If no signs of activity were detected, the patient was invited to a follow-up visit after 4 to 6 weeks. We terminated treatment if there were no signs of activity after 6 months after the last injection or if BCVA had deteriorated after 3 to 6 injections. No distinct BCVA threshold was defined, and the decision to terminate treatment always was individualized. All patients discontinuing treatment were recommended to attend regular follow-up visits at the referring clinic every 3 to 6 months from termination of treatment. Facilities at referring clinics vary, but many have the capacity to perform OCT. It is easy for the referring clinics to obtain a second opinion at the treating clinic, despite limited suspicion of reactivation. If reactivation occurred, the patients were included in the group of patients receiving active treatment, and registration in the database was continued. Treatment, monitoring and decisions regarding retreatment or discontinuation of treatment followed the Danish National guidelines set forward by the Danish Ophthalmological Society. All doctors involved in treating patients with AMD were instructed to follow these guidelines under the supervision of the retinal specialists and chief physicians (T.L.S. and H.K.) at the department. There has been no formal quality control of this or control of reproducibility regarding interpretation of OCT, FA, or ICGA results.
Statistical Analysis
Because the data were normally distributed, the data are reported as means and ranges. Differences among groups were calculated using a 1-way analysis of variance, differences between 2 groups were calculated using the independent samples t test, and the paired-sample t test was used to calculate changes over time. SPSS software for Windows version 19 (SPSS Inc, Chicago, Illinois, USA) was used, and a P level of less than .05 was considered significant.
Results
Patients
The 855 eyes included in the review were divided into 2 groups, one with 456 patients currently in active treatment and another with 399 patients who have discontinued treatment ( Table 1 ). Overall treatment results show a significant decrease in vision from 53.2 letters (range, 1 to 85 letters) to 50.5 letters (range, 1 to 87 letters; P < .001, paired-sample t test). The mean number of injections was 8.7 (range, 1 to 35). Mean duration of follow-up was 23.3 months (range, 4 to 48 months).
Patients Currently in Active Treatment
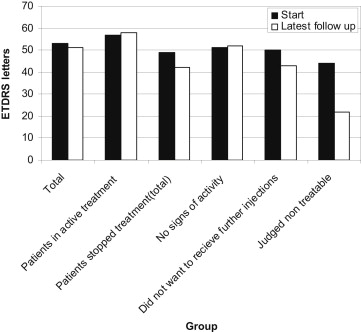
The clinical characteristics and treatment results for patients currently in active treatment are given in Table 1 . In total, 291 women and 165 men had a follow-up longer than 15 months from initiation of treatment. Thirty-four patients were referred again during the 4-year period because of reactivation. The mean length of follow-up was 29.5 months (range, 15 to 48 months). The mean age was 76 years, the youngest patient being 55 years of age and the oldest patient being 98 years of age. The mean BCVA before treatment was 56.9 letters (range, 1 to 85 letters), and at the latest follow up consultation, the mean visual acuity was 57.8 letters (range, 1 to 87 letters), a small and nonsignificant increase in vision ( P = .232, paired-sample t test; Table 1 and Figure ). The mean number of injections was 11.3 (range, 2 to 35). Treatment results are given in more detail in Tables 2 and 3 . Eighty-one (18%) of 456 patients currently in active treatment gained a minimum of 15 letters, whereas 66 (14%) of 456 patients experienced a loss of a minimum 15 letters. At baseline, 6 (1%) of 456 patients had a BCVA of fewer than 20 letters, whereas 51 (11%) of 456 patients of had a BCVA of more than 70 letters. At the latest follow-up, 10 (2%) of 456 patients in this group had a BCVA of fewer than 20 letters, whereas 116 (25%) of 456 patients had a BCVA of more than 70 letters ( Table 3 ). Seven patients in active treatment had a total follow-up of 48 months, whereas 122 patients in active treatment had a follow-up of between 36 and 48 months.
| Minimum 15-Letter Gain | Minimum 15-Letter Loss | |
|---|---|---|
| All patients included (n = 855) | ||
| No. of patients | 142 (17%) | 198 (23%) |
| Mean no. of injections | 8.2 | 8.1 |
| Patients in active treatment (n = 456) | ||
| No. of patients | 81 (18%) | 66 (14%) |
| Mean no. of injections | 11.0 | 11.4 |
| Total patients stopping treatment a (n = 399) | ||
| No. of patients | 61 (15%) | 132 (33%) |
| Mean no. of injections | 4.4 | 6.4 |
| No signs of activity (n = 181) | ||
| No. of patients | 43 (24%) | 36 (20%) |
| Mean no. of injections | 4.4 | 5.9 |
| Did not want to receive further injections (n = 36) | ||
| No. of patients | 4 (11%) | 13 (36%) |
| Mean no. of injections | 5.3 | 5.4 |
| Judged nontreatable (n = 113) | ||
| No. of patients | 3 (3%) | 67 (59%) |
| Mean no. of injections | 4.0 | 7.1 |
| Other reasons (n = 69) | ||
| No. of patients | 11 (16%) | 16 (23%) |
| Mean | 6.0 | 6.2 |
a The patients who terminated treatment were divided into the following subgroups: no signs of activity, did not want to receive further treatment, and judged nontreatable, and other reasons comprising death and the patient moving to another region.
| BCVA < 20 Letters | BCVA > 70 Letters | |||
|---|---|---|---|---|
| Start | Latest Follow-up | Start | Latest Follow-up | |
| All patients (n = 855) | ||||
| No. of patients | 23 (3%) | 90 (11%) | 68 (8%) | 171 (20%) |
| Patients in active treatment (n = 456) | ||||
| No. of patients | 6 (1%) | 10 (2%) | 51 (11%) | 116 (25%) |
| Total patients stopping treatment a (n = 399) | ||||
| No. of patients | 17 (4%) | 80 (20%) | 17 (4%) | 55 (14%) |
| No signs of activity (n = 181) | ||||
| No. of patients | 4 (2%) | 17 (9%) | 7 (4%) | 42 (23%) |
| Did not want to receive further injections (n = 36) | ||||
| No. of patients | 0 (0%) | 2 (6%) | 1 (3%) | 5 (14%) |
| Judged nontreatable (n = 113) | ||||
| No. of patients | 11 (10%) | 54 (48%) | 3 (3%) | 0 (0%) |
| Other reasons (n = 69) | ||||
| No. of patients | 2 (3%) | 7 (10%) | 6 (9%) | 8 (9%) |
a The patients who terminated treatment were divided into the following subgroups: no signs of activity, did not want to receive further treatment, and judged nontreatable, and other reasons comprising death and the patient moving to another region.
Patients Discontinuing Treatment
Clinical characteristics and the cause of discontinuation of treatment of the patients who stopped treatment are summarized in Table 1 . A total of 399 patients (246 women and 153 men) with a mean age of 78.7 years (range, 54 to 96 years) discontinued treatment in the 4-year period. In 181 (45%) of 399 patients discontinuing treatment, there were no signs of activity (denoted as having no signs of activity), 36 (9%) of 399 patients did not want to receive further treatment, and 113 (28%) of 399 patients were judged nontreatable by the treating physician (denoted as being judged nontreatable). Of 399 patients discontinuing treatment, 69 (17%) could not complete follow-up primarily because of death (n = 56) or because they moved to another region of the country (n = 13). There was a significant difference among the 3 groups regarding the BCVA at baseline ( P = .003, analysis of variance). The mean visual acuity at the initiation of treatment was 50.8 letters (range, 1 to 85 letters) in the group showing no signs of activity and was 50.3 letters (range, 20 to 72 letters) in the group of patients who did not want further treatment, whereas the mean visual acuity was only 43.8 letters (range, 1 to 80 letters) in the group judged nontreatable (significantly lower compared with the group with no signs of activity ( P = .001, t test) and almost significantly lower than the group of patients who did not want further treatment ( P = .06, t test). At the end of follow-up, the BCVA was 52.4 letters (range, 5 to 87 letters) in the group showing no signs of activity, a nonsignificant change from baseline ( P = .295, paired-sample t test), 43.3 letters (range, 8 to 75 letters) in the group who did not want further treatment, an almost significant decrease from baseline ( P = .092, paired-sample t test), and 21.8 letters (range, 1 to 69 letters) in the group judged nontreatable, a significant decrease compared with baseline ( P < .0001, paired-sample t test; Table 1 and Figure ).
Tables 2 and 3 show in more detail the treatment results of the different groups. In the group with no signs of activity, 43 (24%) of 181 patients experienced a gain of 15 letters or more, whereas 36 (20%) of 181 patients experienced a loss of 15 letters or more. In the group of patients who did not want to received further treatment, 4 (11%) of 36 patients experienced an increase of minimum 15 letters, whereas 13 (36%) of 36 patients lost a minimum of 15 letters after treatment.
In the group of patients who did not want to receive any further treatment, none of the patients started of treatment with a BCVA of fewer than 20 letters and only 2 (6%) of 36 patients identified fewer than 20 letters on the ETDRS chart at the last follow-up. One patient (3%) of 36 patients had a BCVA of more than 70 letters at initiation of therapy, whereas 5 (14%) of 36 patients had a BCVA of more than 70 letters at the end of follow-up. In the group judged nontreatable, 11 (10%) of 113 patients had a BCVA of fewer than 20 letters at initiation of therapy, increasing to 54 (48%) of 113 patients after treatment. Three (3%) of 113 patients in this group had a BCVA of more than 70 letters at inclusion, whereas no patient in this group could identify more than 70 letters on the ETDRS chart at the last follow-up consultation.
Only 3 (3%) of 113 patients in the group judged nontreatable gained 15 letters or more, whereas 67 (59%) of 113 patients in this group had lost 15 letters or more at the end of follow-up. Four (2%) and 17 (9%) of the 181 patients in the group with no signs of activity had a BCVA of fewer than 20 letters at the initiation of therapy and at the last clinical follow-up, respectively. However, 7 (4%) and 42 (23%) of the 181 patients had a BCVA of more than 70 letters at inclusion and at the last follow-up, respectively.
The reasons that the patients gave for not wanting to receive further treatment and the BCVA of the individual patients are summarized in the Supplemental Table (available at AJO.com ). Of the 36 patients who did not want to receive further treatment, 23 patients (64%) did not give a specific reason for their decision, although they were asked to offer one. Three (8%) of the 36 patients explained that they were generally too tired and could not report to the department so often, and 2 (6%) other patients stopped treatment because of other diseases. Three (8%) of the 36 patients did not show up 3 times consecutively and did not respond to the department attempts to contact them, and 2 (6%) of 36 patients patients believed that the treatment had no effect. One patient (3%) thought that the treatment had made the visual acuity even worse. One patient (3%) stopped because he had experienced corneal abrasions in connection with the last 3 injections, and 1 patient (3%) did not want further treatment because he did not like the treating physician. During the 4-year period, 157 (18%) of the 855 patients included in the study discontinued treatment within the first year of treatment and 174 (20%) patients discontinued treatment during the second year of treatment, whereas 55 (6%) and 13 (2%) patients discontinued treatment during the third and fourth year of treatment, respectively.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


