Purpose
To estimate 4-year incidence and progression of early and advanced age-related macular degeneration (AMD).
Design
Population-based cohort study.
Methods
A comprehensive ophthalmologic examination including stereoscopic fundus photography was performed on adult Latinos at baseline and follow-up. Photographs were graded using a modified Wisconsin Age-Related Maculopathy Grading System. For estimations of incidence and progression of AMD, the Age Related Eye Disease Study Scale was used. Main outcome measures are incidence and progression of early AMD (drusen type, drusen size, and retinal pigmentary abnormalities) and advanced AMD (exudative AMD and geographic atrophy).
Results
A total of 4658 of 6100 subjects (76%) completed the follow-up examination. The 4-year incidence of early AMD was 7.5% (95% CI: 6.7, 8.4) and advanced AMD was 0.2% (95% CI: 0.1, 0.4). Progression of any AMD occurred in 9.2% (95% CI: 8.3, 10.1) of at-risk participants. Incidence and progression increased with age. Incidence of early AMD in the second eye (11.2%) was higher than incidence in the first eye (6.9%). Baseline presence of soft indistinct large drusen ≥250 μm in diameter was more likely to predict the 4-year incidence of pigmentary abnormalities, geographic atrophy, and exudative AMD than smaller or hard or soft distinct drusen.
Conclusions
Age-specific incidence and progression of AMD in Latinos are lower than in non-Hispanic whites. While incident early AMD is more often unilateral, the risk of its development in the second is higher than in the first eye. Older people and those with soft indistinct large drusen had a higher risk of developing advanced AMD compared to those who were younger and did not have soft indistinct large drusen.
Latinos represent the largest of minority populations in the United States. According to the 2004 US Census, 35.6 million people, or 12.5% of the nation’s residents, are Latino. This proportion is expected to increase to 20.1% by the year 2030. The Latino population has unique demographic, socioeconomic, and ocular health characteristics that influence the development of eye disease and its subsequent impact on quality of life.
Various population-based longitudinal studies of eye disease have been conducted on non-Hispanic whites, including the Beaver Dam Eye Study in Wisconsin and the Blue Mountains Eye Study in Australia. Other studies such as the Barbados Incidence Study of Eye Disease have focused on a population of African origin. Although these studies reported data on the incidence and progression of age-related macular degeneration (AMD), given the wide variation in the prevalence and incidence of AMD it would be inaccurate to generalize findings from these studies to people of other ancestries. As observed in cross-sectional studies, there were differences in the prevalence of both early and advanced AMD between non-Hispanic whites and Latinos. Findings from the baseline Los Angeles Latino Eye Study (LALES) reported that age-specific prevalence of early AMD was lower in Latinos (who have 40% European ancestry and 45% Native American ancestry) than the non-Hispanic whites of Beaver Dam, Wisconsin but higher than those from Blue Mountains, Australia. On the other hand, age-specific prevalence of advanced AMD was marginally lower in Latinos compared to the non-Hispanic white populations of both studies. Most interestingly, the prevalence of large drusen, a well-recognized predictor of incident advanced AMD in non-Hispanic whites, was considerably higher in Latinos than in whites. Thus, it is of considerable interest to determine if the incidence and progression of early and advanced AMD in Latinos is comparable to those of non-Hispanic whites.
In this report we describe the 4-year incidence and progression of early and advanced AMD, as well as specific AMD lesions, in a population-based cohort of Latinos.
Methods
Study Population
The Los Angeles Latino Eye Study is a population-based cohort study of eye disease in self-identified Latinos aged 40 years and older living in 6 census tracts in the city of La Puente, Los Angeles County, California. Latinos (Hispanics, Hispanic Americans, and Latino Americans) are individuals who are born into or have descended from a Spanish-speaking community, regardless of race. In the United States they are a heterogeneous group, with the majority of Mexican ancestry (66%). Baseline examination was performed from 2000 to 2003 with 4-year follow-up examination from 2004 to 2008. At baseline, 6357 of 7789 eligible participants (82%) completed an in-home questionnaire and a clinical ophthalmic examination. Details of the study design, methods, and baseline data have been reported elsewhere. The study protocol was approved by the Institutional Review Board (IRB)/Ethics Committee at the University of Southern California and adhered to the recommendations of the Declaration of Helsinki. Written, informed consent was obtained from all participants.
Interview and Examination Procedures
All eligible participants of the baseline LALES examination were invited to return for a home interview and a clinical examination. Similar questionnaire and examination procedures were used for both baseline and follow-up studies. Trained ophthalmologists and technicians performed a comprehensive ocular examination using standardized protocols, which included 30-degree stereoscopic color fundus photographs of Diabetic Retinopathy Study field 1 (centered on the optic disc), field 2 (centered on the macula), and a modified field 3 (nonstereoscopic, temporal to and including the fovea) on all participants.
Age-Related Macular Degeneration Grading
A modification of the Wisconsin Age-Related Maculopathy Grading System (WARMGS) was used to perform grading of individual AMD lesions by masked graders at the Wisconsin Ocular Epidemiology Reading Center. Detailed descriptions of all grading procedures and definitions were previously reported. In brief, a lesion-by-lesion evaluation of each fundus photograph taken at the follow-up study was performed to determine maximum drusen size, type, area, and retinal pigmentary abnormalities. Each eye was graded independent of the contralateral eye. Any discrepancies between 2 initial graders were adjudicated by a senior grader using standardized edit rules. All data from the detailed grading were checked for progression or regression of AMD lesions using a custom program. For eyes that had changes in lesion severity by 2 or more steps between the baseline and 4-year follow-up examinations, a longitudinal review was conducted through side-by-side comparison of photographs from both examination periods. Graders were masked to the year the photographs were taken.
Definitions of Age-Related Macular Degeneration
Definitions of AMD component lesions, which include specific drusen size, drusen types, and retinal pigmentary abnormalities, have been described in detail elsewhere. In this study, we present incidence and progression for maximum drusen size (<63 μm, ≥63 μm to <125 μm, ≥125 μm to <250 μm, and ≥250 μm), drusen type in increasing severity (hard distinct, soft distinct, and soft indistinct/reticular), increased retinal pigment, retinal pigment epithelial (RPE) depigmentation, and signs of geographic atrophy and exudative AMD.
Various classifications were used to define the presence of early and advanced AMD. In this study we present data based on 2 systems, the WARMGS and the Age-Related Eye Disease Study (AREDS). The WARMGS defined early AMD as the absence of signs of advanced AMD and the presence of 1) soft indistinct or reticular drusen or 2) hard distinct or soft distinct drusen with pigmentary abnormalities (RPE depigmentation or increased retinal pigment). With respect to drusen type, any soft indistinct or reticular drusen is considered early AMD, as well as any other type with pigmentary abnormality. With respect to drusen size, any drusen size with a pigmentary abnormality is considered early AMD. Any drusen size or type with evidence of geographic atrophy and/or exudative lesion is considered late AMD.
Advanced AMD was defined as the presence of either 1) geographic atrophy or 2) exudative AMD. Exudative AMD was defined as presence of any of the following exudative lesions: 1) pigment epithelial detachment or age-related retinal detachment, 2) subretinal hemorrhage, 3) subretinal scar (subretinal fibrous scar), or 4) laser treatment for exudative AMD. Population-based studies that used this definition include the Beaver Dam Eye Study, the Blue Mountains Eye Study, and the Los Angeles Latino Eye Study for baseline data.
The AREDS Research Group developed a more detailed severity scale for AMD that allowed for the classification of risk categories and the tracking of AMD development along an 11-step scale. Stereoscopic fundus photographs were graded in detail and scored for increasing severity of drusen characteristics (size, type, area), pigmentary abnormalities (increased pigment, RPE depigmentation, geographic atrophy), and retinal abnormalities (RPE detachment, serous or hemorrhagic sensory retinal detachment, subretinal hemorrhage, subretinal fibrous tissue). With this scale, early AMD was defined as steps 4, 5, 6, 7, or 8 while advanced AMD was defined as steps 9, 10, or 11, with steps 1, 2, or 3 signifying presence of questionable lesions or other characteristics not amounting to AMD.
For this report, we present the incidence and progression of early and advanced AMD as defined by the WARMGS and the AREDS severity scale. Progression, regression, and disappearance are also reported for individual AMD lesions, following definitions outlined in the WARMGS. All definitions are summarized in Table 1 .
| Incidence | Progression | Regression | Disappearance | |||
|---|---|---|---|---|---|---|
| Baseline | Follow-up | Baseline | Follow-up | |||
| Age-Related Eye Disease Study (AREDS) 11-step severity scale | ||||||
| Early AMD | ≤Step 3 | Step 4, 5, 6, 7, or 8 | ≥2-step increase | — | — | — |
| Advanced AMD | ≤Step 8 | Step 9, 10, or 11 | — | — | — | — |
| Wisconsin age-related maculopathy grading system definitions | ||||||
| Early AMD | No early or advanced AMD lesions in both eyes | Either 1) any soft indistinct or reticular drusen, or 2) retinal pigmentary abnormalities with any type of drusen, in either eye | ≥2-step increase from Level 1–3, or ≥1-step increase from Level 4 | — | — | — |
| Advanced AMD | No advanced AMD lesions in both eyes | Exudative AMD or geographic atrophy in either eye | — | — | — | — |
| Drusen size |
|
| ≥2 additional involved subfields at follow-up without change in drusen size from baseline | ≥2 fewer involved subfields at follow-up without change in drusen size from baseline |
|
|
| Drusen type |
|
| ≥2 additional involved subfields at follow-up without change in drusen type from baseline | ≥2 fewer involved subfields at follow-up without change in drusen type from baseline |
|
|
| Increased retinal pigment | No increased retinal pigment in any subfield | Increased retinal pigment in any subfield | Either 1) ≥2 additional involved subfields at follow-up without change in maximum score from baseline, or 2) increase in the maximum score at follow-up | Either 1) ≥2 fewer involved subfields at follow-up without change in maximum score from baseline, or 2) decrease in the maximum score at follow-up | Increased retinal pigment in at least 1 subfield | No increased retinal pigment in all subfields |
| RPE depigmentation | No RPE depigmentation in any subfield | Definite RPE depigmentation in any subfield | Either 1) ≥2 additional involved subfields at follow-up without change in maximum score from baseline, or 2) increase in the maximum score at follow-up | Either 1) ≥2 fewer involved subfields at follow-up without change in maximum score from baseline, or 2) decrease in the maximum score at follow-up | Definite RPE depigmentation in at least 1 subfield | No RPE depigmentation in all subfields |
| Geographic atrophy | No geographic atrophy | Definite geographic atrophy | Increase of ≥2 disc areas, or movement of lesion towards the center | Decrease of ≥2 disc areas, or movement of lesion away from the center | Definite geographic atrophy | No geographic atrophy |
| Exudative AMD | No exudative AMD | Exudative AMD | Increase of ≥2 disc areas, or movement of lesion towards the center | Decrease of ≥2 disc areas, or movement of lesion away from the center | Exudative AMD | No exudative AMD |
Definitions of Incidence and Progression of Age-Related Macular Degeneration
In our study we present the 4-year incidence of early and advanced AMD and 4-year incidence of specific AMD lesions using 2 mutually exclusive at-risk cohorts of participants. Our first cohort refers to persons who did not have any evidence of AMD at baseline in both eyes, thus being at risk of developing AMD in either or both eyes at follow-up. This definition of incidence in the 1st eye is usually referred to as person-specific incidence in population-based studies of AMD. Our second cohort refers to people who had only 1 eye without evidence of AMD at baseline while the contralateral eye had some evidence of AMD at baseline. Thus in this cohort of participants, the disease-free eye was at risk of developing AMD at follow-up, and any presence of AMD at the follow-up assessment was considered to be incidence in the 2nd eye for that person.
To enable a closer comparison of incidence data between LALES and other population-based studies with similar photography and grading protocols, annual incidences are reported using the WARMGS classification for AMD. Similar age-group stratification was also used to allow uniformity across studies.
For estimations of the 4-year progression of AMD, the AREDS scale was used. Person-specific progression is reported by concatenating the score given for each eye, therefore defining overall severity using the score from the more affected eye. In the 11-step AREDS severity scale, progression was defined as a 2-or-more-step increase in concatenated score from baseline to the 4-year follow-up examination, and estimated for persons with gradable fundus photographs at both time points. The cohort at risk for progression was defined as having step 9 or less (no AMD or early AMD) at baseline.
Data and Statistical Analysis
All clinical and grading data were entered into a central database with internal automated quality control checks. The Statistical Analysis System (version 9.1, SAS Institute Inc, Cary, North Carolina, USA) was used for tabulations and statistical analyses, conducted at the 0.05 significance level. Age at baseline was categorized into 5 groups (40–49 years, 50–59 years, 60–69 years, 70–79 years, and 80+ years) for all analyses.
Sociodemographics and clinical characteristics between participants and nonparticipants, and between subjects with gradable and ungradable photographs were compared using the χ 2 test for categorical variables and the Student t test for continuous variables. Associations between age groups and incidence and progression estimates were tested by the Mantel-Haenszel test of trend. Differences in incidence and progression rates for all AMD lesions between right and left eyes were tested using the McNemar test. The crude overall incidence and progression rates were age-adjusted to the LALES study cohort using direct standardization methods. Results were also annualized to enable comparison across population-based studies. Inter-grader and intra-grader agreements were assessed in a random sample of eyes with quadratic weighted κ statistics.
Results
Of the 6100 living eligible participants identified, 4658 (76%) participated in the 4-year follow-up study. Mean follow-up period was 4.3 ± 0.03 years. Mean age of participants was 54.7 ± 10.5 years, 60% were female, and 76% were born outside of the United States. In this study, the country of origin was identified as Mexico in 64%, US in 24%, El Salvador in 5%, Guatemala in 2.5%, and Nicaragua 1%. Other countries were identified in 3.5% of participants. Living eligible participants who did not participate at the 4-year examination were mostly male (44% vs 40%; P < .01), not married (33% vs 29%; P = .01), and without health insurance coverage (46% vs 33%; P < .001) compared to those who participated.
Of those who completed the ophthalmologic examination (n = 4658), fundus photographs gradable for AMD lesions in at least 1 eye were available for 4029 participants ( Figure ). Six hundred twenty-nine participants did not complete fundus photography because of various reasons. More common reasons included refusal (n = 36), poor fixation (n = 74), poor dilation (n = 109), cataract (n = 114), movement (n = 253), and poor view (n = 51). Sixty-eight of the 98 participants who had completed photography did not produce gradable photographs in both eyes because of presence of other non-AMD conditions obscuring AMD lesions (eg, diabetic macular edema) and 30 had significant media opacities or poor camera focus. Of the 3931 follow-up participants with gradable fundus photograph, 3908 had gradable fundus photographs in at least 1 eye from their baseline examination, thus making this the analysis cohort.
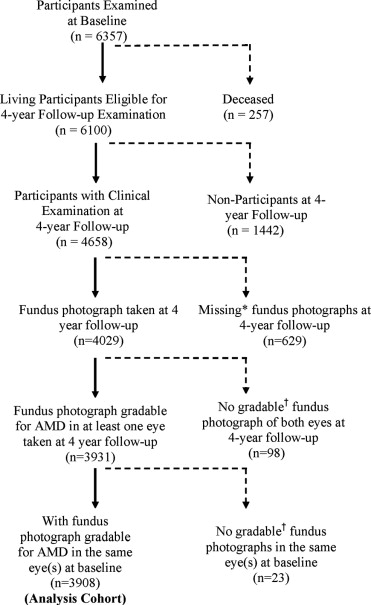
Living eligible participants who were not included in this analysis, compared to those who were included, were less educated (32% vs 35%; P = .03), were not married (34% vs 29%; P < .001), did not have health insurance coverage (41% vs 33%; P < .001), had fewer comorbidities (37% vs 41%; P = .001), reported worse visual health status (61% vs 57%; P = .01), and had a higher proportion with history of eye disease (14% vs 12%; P = .01), cataract (11% vs 9%, P = .001), and diabetic retinopathy (3% vs 2%, P < .001). Living eligible participants were not included in this analysis because of either missing or nongradable fundus photographs at baseline or follow-up examinations. The intra-grader agreement for AMD lesions was 100% and the inter-grader agreement was 89.7% with a quadratic weighted κ of 0.82 (95% CI: 0.63, 1.0).
Incidence of Age-Related Macular Degeneration
The 4-year incidence of early AMD in at least 1 eye for people without signs of drusen or other AMD lesions in both eyes at baseline was 6.9% (95% CI: 6.0, 7.8) ( Table 2 ). The incidence was higher in the second eye, with 62 out of 554 people (11.2%) developing signs of early AMD after 4 years, given the presence of AMD lesions in the contralateral eye at baseline. Incidence of early AMD in either eye (combined first and second eye) was 7.5%. Incidence in both the first and either eye increased significantly with age (test of trend, P < .001 for both.
| Age at Baseline (Years) | Incidence in 1st Eye | Incidence in 2nd Eye | Incidence in Either Eye | ||||||
|---|---|---|---|---|---|---|---|---|---|
| N | n | % (CI) | N | n | % (CI) | N | n | % (CI) | |
| Early AMD | |||||||||
| 40–49 | 1311 | 50 | 3.8 (2.8, 5.0) | 166 | 16 | 9.6 (5.4, 15.2) | 1477 | 66 | 4.5 (3.5, 5.7) |
| 50–59 | 1034 | 66 | 6.4 (5.0, 8.0) | 175 | 13 | 7.4 (4.0, 12.4) | 1209 | 79 | 6.5 (5.2, 8.1) |
| 60–69 | 591 | 72 | 12.2 (9.7, 15.1) | 122 | 16 | 13.1 (7.7, 20.4) | 713 | 88 | 12.3 (10.0, 15.0) |
| 70–79 | 198 | 25 | 12.6 (8.3, 18.1) | 81 | 16 | 20.0 (11.7, 30.1) | 279 | 41 | 14.7 (10.8, 19.4) |
| 80+ | 21 | 4 | 19.0 (5.4, 41.9) | 10 | 1 | 10.0 (0.3, 44.5) | 31 | 5 | 16.1 (5.5, 33.7) |
| P < .001 | P = .05 | P < .001 | |||||||
| Overall | 3155 | 217 | 6.9 (6.0, 7.8) | 554 | 62 | 11.2 (8.7, 14.1) | 3711 | 279 | 7.5 (6.7, 8.4) |
| Advanced AMD | |||||||||
| 40–49 | 1447 | 0 | N/A | 56 | 0 | N/A | 1503 | 0 | N/A |
| 50–59 | 1196 | 1 | 0.1 (0.0, 0.5) | 57 | 0 | N/A | 1253 | 1 | 0.1 (0.0, 0.4) |
| 60–69 | 719 | 1 | 0.1 (0.0, 0.8) | 48 | 0 | N/A | 767 | 1 | 0.1 (0.0, 0.7) |
| 70–79 | 267 | 4 | 1.5 (0.4, 3.8) | 51 | 0 | N/A | 318 | 4 | 1.3 (0.3, 3.2) |
| 80+ | 38 | 2 | 5.3 (0.6, 17.7) | 8 | 0 | N/A | 46 | 2 | 4.3 (0.5, 14.8) |
| P < .001 | P < .001 | ||||||||
| Overall | 3667 | 8 | 0.2 (0.1, 0.4) | 220 | 0 | N/A | 3887 | 8 | 0.2 (0.1, 0.4) |
a AREDS (Age-Related Eye Disease Study) 11-step severity scale: Incidence of early AMD defined as presence of step 4, 5, 6, 7, or 8 characteristics in at least 1 eye that had characteristics of step 3 or less at baseline. Incidence of advanced AMD defined as presence of step 9, 10, or 11 characteristics in at least 1 eye that had characteristics of step 8 or less at baseline.
Incidence of advanced AMD remained low in this cohort of Latinos, with only 8 out of 3667 at-risk participants (0.2%) showing signs of exudative AMD, geographic atrophy, or photocoagulation for AMD in at least 1 eye after 4 years of follow-up ( Table 2 ). Advanced AMD was seen only in people who were 50 years or older at baseline. Incidence estimates increased with age ( P < .001). No incidence of advanced AMD occurred in the second eye.
Drusen Characteristics, Pigmentary Abnormalities, Geographic Atrophy, and Exudative Age-Related Macular Degeneration
Table 3 shows the 4-year incidence of various AMD lesions stratified by occurrence in the first and second eye. Incidence was reported in over half of participants at risk for drusen <63 μm in diameter (61.2% in the first eye, 57.4% in the second eye). Incidence was less frequent for larger-sized drusen than those of smaller size. Incidence of soft distinct drusen was higher than that of soft indistinct drusen (incidence in the first eye, 10.3% vs 2.2%).
| Incidence in 1st Eye | Incidence in 2nd Eye | Incidence in Either Eye | |||||||
|---|---|---|---|---|---|---|---|---|---|
| N | n | % (CI) | N | n | % (CI) | N | n | % (CI) | |
| Drusen size (diameter) a | |||||||||
| <63 μm | 152 | 93 | 61.2 (53.0, 69.0) | 787 | 452 | 57.4 (53.9, 60.9) | 939 | 547 | 57.9 (54.7, 61.1) |
| ≥63 μm, <125 μm | 2282 | 420 | 18.4 (16.8, 20.1) | 1058 | 216 | 20.4 (18.0, 23.0) | 3340 | 636 | 19.0 (17.7, 20.4) |
| ≥125 μm, <250 μm | 3152 | 203 | 6.4 (5.6, 7.4) | 584 | 53 | 9.1 (7.0, 11.7) | 3736 | 256 | 6.9 (6.1, 7.7) |
| ≥250 μm | 3577 | 57 | 1.6 (1.2, 2.1) | 303 | 14 | 4.6 (2.5, 7.6) | 3880 | 71 | 1.8 (1.4, 2.3) |
| Drusen type | |||||||||
| Soft distinct | 2829 | 290 | 10.3 (9.2, 11.4) | 749 | 87 | 11.6 (9.4, 14.1) | 3578 | 379 | 10.5 (9.5, 11.6) |
| Soft indistinct | 3459 | 77 | 2.2 (1.8, 2.8) | 372 | 26 | 7.0 (4.6, 10.1) | 3831 | 103 | 2.7 (2.2, 3.3) |
| Any pigment abnormality | 3553 | 80 | 2.3 (1.8, 2.8) | 320 | 11 | 3.4 (1.7, 6.1) | 3873 | 91 | 2.3 (1.9, 2.9) |
| Increased retinal pigment | 3145 | 101 | 3.2 (2.6, 3.9) | 525 | 14 | 2.7 (1.5, 4.4) | 3670 | 118 | 3.1 (2.6, 3.7) |
| RPE depigmentation | 3419 | 66 | 1.9 (1.5, 2.4) | 330 | 9 | 2.7 (1.3, 5.1) | 3749 | 75 | 1.9 (1.5, 2.4) |
| Geographic atrophy | 3702 | 5 | 0.1 (0.0, 0.3) | 149 | 0 | N/A | 3851 | 5 | 0.1 (0.0, 0.3) |
| Pure geographic atrophy | 3882 | 3 | 0.1 (0.0, 0.2) | 24 | 0 | N/A | 3906 | 3 | 0.1 (0.0, 0.2) |
| Exudative AMD | 3707 | 5 | 0.1 (0.0, 0.3) | 146 | 0 | N/A | 3853 | 5 | 0.1 (0.0, 0.3) |
a Incidence refers to the presence of drusen of a specific size at follow-up when at baseline there was no evidence of drusen of that size in the same eye although drusen of a different size may be present.
The 4-year incidence of pigmentary abnormalities and signs of advanced AMD were relatively low in this population. Only 3.2% had increased retinal pigment and 1.9% RPE depigmentation occurring in the first eye ( Table 3 ). Overall only 5 people developed exudative AMD while 3 developed pure geographic atrophy in at least 1 eye. Incidence in the second eye was marginally higher than incidence in the first eye for presence of RPE depigmentation (2.7% vs 1.9%, respectively).
Table 4 shows the incidence of various AMD lesions stratified according to baseline drusen type and drusen size for participants who were at risk for that specific lesion. There was no statistically significant difference in the incidence estimates between right and left eyes; thus results are reported for the right eye only. Presence of soft indistinct drusen at baseline was associated with a higher incidence of RPE depigmentation (13.7%), increased retinal pigment (29.1%), pure geographic atrophy (1.3%), and exudative AMD (1.4%) as compared to other drusen types. Larger drusen (diameter ≥250 μm) was also associated with a higher incidence of pigmentary abnormalities (21.6%) and pure geographic atrophy (1.5%) than eyes with smaller drusen present at baseline.
| Four-Year Incidence of AMD Lesions | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RPE Depigmentation | Increased Retinal Pigment | Any Pigmentary Abnormalities | Geographic Atrophy | Pure Geographic Atrophy | Exudative Macular Degeneration | |||||||||||||
| N | n | % | N | n | % | N | n | % | N | n | % | N | n | % | N | n | % | |
| Baseline drusen type a | ||||||||||||||||||
| None or hard indistinct | 549 | 2 | 0.4 | 534 | 3 | 0.6 | 551 | 2 | 0.4 | 559 | 0 | N/A | 579 | 0 | N/A | 559 | 0 | N/A |
| Hard distinct | 2674 | 11 | 0.4 | 2581 | 22 | 0.9 | 2684 | 9 | 0.3 | 2702 | 0 | N/A | 2726 | 0 | N/A | 2703 | 0 | N/A |
| Soft distinct | 368 | 16 | 4.4 | 317 | 21 | 6.6 | 380 | 15 | 4.0 | 418 | 0 | N/A | 426 | 0 | N/A | 417 | 1 | 0.2 |
| Soft indistinct | 73 | 10 | 13.7 | 55 | 16 | 29.1 | 83 | 16 | 19.3 | 146 | 3 | 2.1 | 149 | 2 | 1.3 | 147 | 2 | 1.4 |
| Baseline drusen size (maximum diameter in μm) | ||||||||||||||||||
| None or questionable | 549 | 2 | 0.4 | 534 | 3 | 0.6 | 551 | 2 | 0.4 | 559 | 0 | N/A | 579 | 0 | N/A | 559 | 0 | N/A |
| <63 | 2297 | 5 | 0.2 | 2221 | 12 | 0.5 | 2302 | 3 | 0.1 | 2308 | 0 | N/A | 2328 | 0 | N/A | 2308 | 0 | N/A |
| ≥63 to <125 | 573 | 12 | 2.1 | 536 | 24 | 4.5 | 582 | 15 | 2.6 | 626 | 0 | N/A | 635 | 0 | N/A | 927 | 1 | 0.2 |
| ≥125 to <250 | 200 | 14 | 7.0 | 162 | 19 | 11.7 | 212 | 11 | 5.2 | 266 | 2 | 0.8 | 271 | 1 | 0.4 | 267 | 2 | 0.8 |
| ≥250 | 45 | 6 | 13.3 | 34 | 4 | 11.8 | 51 | 11 | 21.6 | 66 | 1 | 1.5 | 67 | 1 | 1.5 | 65 | 0 | N/A |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


