Purpose
To compare the efficacy of as-needed or variable dosing of intravitreal bevacizumab to continuous fixed-interval dosing in the management of neovascular age-related macular degeneration (AMD).
Design
Prospective, open-label, randomized clinical study.
Methods
One hundred twenty eyes of 120 patients with treatment-naïve subfoveal neovascular AMD participated in this study at the American University of Beirut and Hotel Dieu de France Retina Clinics. Eyes were randomized (1:1) to fixed-interval dosing (every 4 to 6 weeks) or variable dosing with intravitreal bevacizumab (1.25 mg/0.05 mL). Best-corrected visual acuity (BCVA) and central retinal thickness (CRT) using optical coherence tomography (OCT) were measured at baseline and at each follow-up visit. Presence or recurrence of fluid on OCT was the main indicator for retreatment in variable dosing. Main outcome measure was improvement in BCVA and CRT at 12 months.
Results
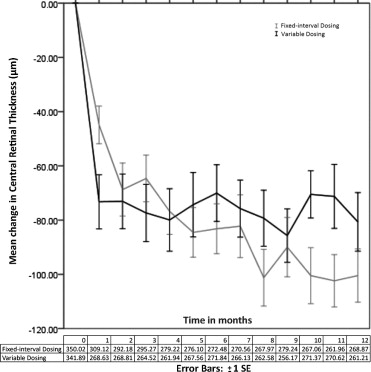
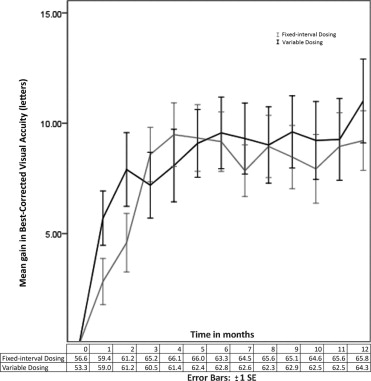
Compared to baseline, variable dosing had a mean improvement in BCVA of 11.0 letters after 12 months vs 9.2 letters for fixed-interval dosing ( P = .81). Similarly, CRT decreased after 12 months by 80.7 μm for variable dosing vs 100.5 μm for fixed-interval dosing ( P = .37). The average number of injections over 12 months was higher for fixed-interval dosing than variable dosing (9.5 vs 3.8 injections, P < .001).
Conclusions
Fixed-interval and variable dosing regimens of intravitreal bevacizumab improved visual acuity and anatomic outcomes after 12 months in eyes with neovascular AMD. However, variable dosing had a reduced treatment burden. Larger trials are needed to confirm these results.
Therapy against vascular endothelial growth factor (VEGF) has revolutionized the treatment of neovascular age-related macular degeneration (AMD). In 2006, the US Food and Drug Administration approved ranibizumab, an Fab antibody fragment against all isoforms of VEGF-A, for the treatment of neovascular AMD. In the MARINA and ANCHOR trials, monthly intravitreal ranibizumab was able to stabilize visual acuity in over 90% of cases and improve visual acuity in about 30% to 40% of cases. The PrONTO study showed it is possible to stabilize and improve visual acuity with variable dosing based mostly on optical coherence tomography (OCT) while reducing the total number of injections over 12 months.
Bevacizumab, a humanized full-length antibody against all isoforms of VEGF-A, has been used off-label for the management of neovascular AMD since 2005. Studies showed that intravitreal bevacizumab was safe, and currently different dosing regimens are used for treatment of neovascular AMD. In the recently published CATT trial, the analysis between as-needed bevacizumab and monthly bevacizumab or as-needed bevacizumab and monthly ranibizumab was inconclusive. This study addresses the efficacy of as-needed bevacizumab treatment compared to fixed-interval treatment in the management of neovascular AMD.
Methods
The study was conducted at the Medical Centers of the American University of Beirut and Hotel Dieu de France. Patients with treatment-naïve neovascular AMD were enrolled if they met the following criteria: 1) age 50 years or older; 2) subfoveal choroidal neovascularization (CNV) attributable to AMD diagnosed by fluorescein angiography (FA); 3) presence of subretinal fluid, cystic maculopathy, or central retinal thickness >250 μm on OCT; 4) best-corrected vision, using ETDRS charts, between 20/40 and 20/400 (Snellen equivalent); 5) CNV less than 5400 μm in greatest linear dimension; and 6) ability to understand and sign a consent form. Patients were excluded if there was: 1) presence of subfoveal scarring or hemorrhage; 2) media opacity that would prevent good-quality retinal imaging; 3) history of uveitis, vitrectomy, diabetic retinopathy, or other condition that may affect vision; and 4) thromboembolic event less than 6 months prior to enrollment. All CNV lesion types were included except for retinal angiomatous proliferation (RAP) and polypoidal choroidal vasculopathy (PCV) since they may respond differently to treatment. Eligible eyes were randomized into either the continuous fixed-interval dosing group or variable dosing group in a 1:1 ratio using a randomization program (GraphPad StatMate, version 1.01i; GraphPad Software Inc, San Diego, California, USA). If both eyes of the same patient were eligible, then the eye with the worse visual acuity was enrolled. To be able to detect a difference of at least 5 letters in mean visual acuity using the independent t test with 80% power and an alpha level of 5%, assuming a standard deviation of 10 letters, 60 eyes were needed in each group.
Baseline Evaluation
Baseline evaluation included best-corrected visual acuity (BCVA), slit-lamp examination of the anterior segment, dilated fundus examination, FA, and OCT. Eyes with clinical, fluorescein angiographic, or OCT characteristics suggestive of RAP or PCV had indocyanine green angiography to more accurately ascertain these 2 lesion types and exclude them from the study. Visual acuity testing was done according to the Early Treatment Diabetic Retinopathy Study (ETDRS) refraction protocol using ETDRS charts at 4 meters with an illuminated cabinet (Precision Vision, La Salle, Illinois, USA).
One-millimeter central retinal thickness (CRT) was determined by spectral-domain OCT (Cirrus; Carl Zeiss Meditec, Dublin, California, USA) using a macular cube (512 × 128 lines). The internal limiting membrane and retinal pigment epithelium (RPE) lines were identified automatically by the algorithms of the OCT machine, which then calculated the thickness between these layers. The boundaries were confirmed by 1 of 3 investigators (Z.F.B., A.R.S., H.I.S.) and corrected using the “edit layers” function if the algorithms incorrectly traced the actual boundaries. If the patient had difficulty fixating, the scan pattern in the fundus viewport was manually centered at the fovea. Only good-quality examinations with a signal strength of 6/10 or better were retained. The “Repeat Set-up” option was used to acquire subsequent OCT. Fluid in the macula was defined as intraretinal cysts or subretinal fluid. A fluid-free macula was considered if there was absence of intraretinal or subretinal fluid on OCT.
Initial and Follow-up Treatment
Patients randomized to fixed-interval dosing and variable dosing received the first and subsequent intravitreal bevacizumab (Avastin; Genentech, South San Francisco, California, USA) injections based on a standard protocol. At each follow-up, BCVA, slit-lamp examination, dilated fundus examination, and OCT were performed. The response to treatment on OCT was described as: 1) complete response if there was total absence of fluid in the macula; 2) partial response if there was a decrease of at least 50% in CRT with persistent macular fluid; 3) minimal response if there was a decrease of less than 50% in CRT with persistent macular fluid; and 4) no response if there was no change or increase in macular fluid. FA was repeated at the discretion of the treating physician.
Patients enrolled in the fixed-interval dosing group received bevacizumab injections every 6 weeks unless the eye was not showing a complete response after the 2 initial consecutive injections. If there was evidence of fluid on OCT, 6 weeks after the second injection, then the third injection was administered and treatment interval was reduced to every 4 weeks. Once a fluid-free macula was achieved, the treatment interval could be extended back to 6 weeks at the discretion of the treating physician.
Patients in the variable dosing group were seen 6 weeks after the initial treatment. If there was a complete response, the patient was followed every 6 weeks. If there was persistent fluid in the macula on OCT, a second bevacizumab injection was given and follow-up made 6 weeks later. If there was evidence of any fluid in the macula on OCT 6 weeks after the second injection, a third injection was administered and further follow-up and treatment were conducted every 4 weeks until the macula was fluid-free. However, if there was persistent fluid on OCT after 3 additional consecutive injections, the treating physician had the option to stop injections and observe. In this case the patient would have received a total of 6 bevacizumab injections over a period of 24 weeks. Based on personal observation and other studies, eyes with some persistent fluid in the macula can show stability in BCVA. Figure 1 summarizes the initial treatment protocol for the variable dosing group.
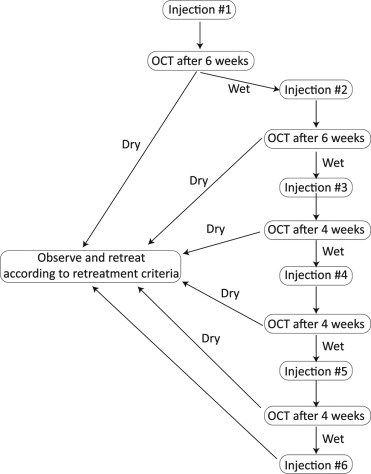
Follow-up was carried out every 6 weeks in the variable dosing group once the macula was dry on OCT or it was decided to observe persistent fluid in the macula. However, this interval could be reduced to every 4 weeks if deemed necessary by the treating physician. Retreatment in the variable dosing group was based on the following criteria: 1) recurrence or presence of any fluid in the macula on OCT in a previously dry macula; 2) an increase in OCT central retinal thickness of at least 50 μm from the lowest recorded value, especially for eyes with persistent macular fluid; 3) visual acuity loss of at least 5 letters with OCT evidence of fluid in the macula; 4) new macular hemorrhage; or 5) new area of classic CNV. Persistent and stable serous retinal pigment epithelial detachment (PED) on OCT was not a retreatment criterion.
Once the 2-year results of the PrONTO trial were available in July 2009, retreatment criteria for variable dosing was revised to include, in addition to those previously mentioned, any OCT changes suggestive of an active CNV. This included the appearance or increase in size of a retinal cyst or subretinal fluid. Although it was not our intention to achieve total resolution of a PED already present at baseline, the appearance of a new PED or increase in size of a previously stable PED became a retreatment criterion even in the absence of retinal fluid. These amendments to the retreatment criteria occurred less than 2 months after patient recruitment commenced. High-definition 5-line raster scan centered on the fovea was also obtained for further qualitative assessment of the central macular area. In addition, the treating physician had the option to do high-definition scans to any suspicious areas of the macula that were not clear on the cube scan. In December 2010, OCT software was upgraded to version 4.5, which allowed “macular change analysis.” This made possible side-by-side and line-to-line comparison between different OCT studies, allowing more precise qualitative analysis.
Treating Physicians and Masking
The 3 treating physicians (Z.F.B., H.I.S., A.R.S.) are retina specialists experienced in the management of neovascular AMD with anti-VEGF therapy using the same treatment protocol as this study. Treating physicians were not masked to the treatment regimen of patients under their care and no sham injections were employed. However, visual acuity examiners were masked to treatment regimen and patients were instructed not to share this information with the examiner. Each patient was assigned a unique identification number on all study documents and imaging modalities. The physician reviewing OCT images or other material to be recorded in the study was masked to that particular patient’s identity and treatment regimen and in no way could be involved in the treatment of that patient.
Adverse Events
Severe ocular adverse events were defined as sudden loss of 30 letters or more in BCVA, endophthalmitis, vitreous hemorrhage, retinal detachment, cataract formation or progression, intraocular inflammation, and intraocular pressure persistently above 21 mm Hg. Severe systemic adverse events were defined as elevation of arterial blood by more than 10 mm Hg, stroke, myocardial ischemic event, and peripheral thrombotic events.
Statistical Analysis
Sample characteristics were summarized for the overall sample and for each group using frequency distribution for sex and lesion type, while mean and standard deviation were used for age. To ensure that randomization yielded comparable groups, these characteristics were compared between the 2 study arms using χ 2 test for sex and lesion type and independent t test for age.
The main outcome measures, improvement in BCVA and CRT, were analyzed using linear regression where the dependent variable was BCVA at 12 months (or CRT value at 12 months) and the independent variables were baseline BCVA (or CRT) and dosing regimen. We also performed the independent t test and the Wilcoxon rank sum test to compare relative change in BCVA and CRT between the 2 groups. BCVA and CRT were compared between baseline and 12 months within each group using the paired t test and Wilcoxon signed rank test.
The number of eyes avoiding loss of 15 letters and those that gained at least 15 letters was compared between the 2 groups using the χ 2 test or Fisher exact test for cell counts less than 5. The average number of injections was compared between the 2 groups using Wilcoxon rank sum test.
All analyses were done using Statistical Package for Social Sciences (SPSS, version 17; SPSS Inc, Chicago, Ilinois, USA). A P value of .05 or less was considered statistically significant.
Results
Demographics
Recruitment for the study started in May 7, 2009 and was completed by October 12, 2009, with 60 patients randomized to each group. All patients completed the 12 months of the study and were able to make scheduled visits with no greater than a 7-day delay. The majority of the participants of both groups combined were female (78/120 patients, 65.0%) and had an average age of 76.8 years. There were no significant differences in sex distribution ( P = .25) or mean age ( P = .60) between the 2 groups. The most frequent lesion type was pure occult (50/120 eyes, 41.7%), followed by minimally classic (46/120 eyes, 38.3%). Lesion type did not significantly differ between the 2 groups ( P = .17).
Visual Acuity and Central Retinal Thickness
At baseline, mean BCVA was 56.6 and 53.3 letters in the fixed-interval and variable dosing groups, respectively ( t test, P = .21; Wilcoxon, P = .14). At 12 months, mean BCVA increased by 11.0 letters from baseline, to 64.3 letters, in the variable dosing group ( t test and Wilcoxon, P < .001). The fixed-interval dosing group achieved a mean BCVA of 65.8 letters at 12 months, which was a 9.2-letter increase compared to baseline ( t test and Wilcoxon, P < .001). Increase in mean BCVA by 12 months did not differ significantly between the 2 groups (regression analysis, P = .81; t test, P = .37; Wilcoxon, P = .50). Figures 2 and 3 illustrate the gain in BCVA over the course of the study.
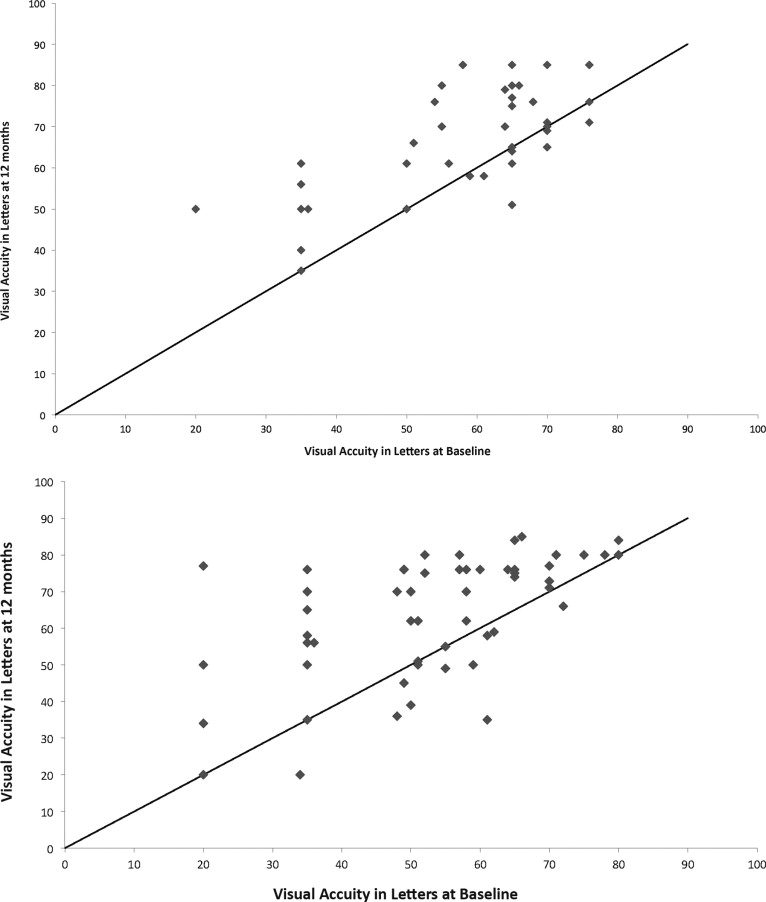
Sixty of 60 eyes (100%) in fixed-interval dosing and 59 of 60 eyes (98.3%) in variable dosing avoided losing at least 15 letters of BVCA ( P = .99). Twenty-four of the 60 eyes (40%) in the variable dosing group gained at least 15 letters vs 21 of 60 eyes (35%) for fixed-interval treatment ( P = .57).
Baseline mean CRT was 350.0 μm for fixed-interval dosing and 341.9 μm for variable dosing ( t test, P = .31; Wilcoxon, P = .25). Compared to baseline, mean CRT at 12 months decreased by 80.7 μm to 261.2 μm for the fixed-interval group ( t test and Wilcoxon, P < .001) and by 100.5 μm to 268.9 μm for the variable dosing group ( t test and Wilcoxon, P < .001). Decrease in mean CRT at 12 months did not differ significantly between the 2 groups (regression analysis, P = .37; t test, P = .13; Wilcoxon, P = .26). Figure 4 shows the mean decrease in CRT over 12 months.