Purpose
To evaluate subfoveal choroidal thickness in patients with central retinal vein occlusion (CRVO) using enhanced depth imaging optical coherence tomography.
Design
Retrospective observational study.
Methods
We measured bilateral subfoveal choroidal thickness, averaged for 100 scans, in 36 patients (mean age, 66 ± 15 years; 26 women and 10 men) with unilateral CRVO by using the enhanced depth imaging methods of the Spectralis optical coherence tomography system. Twenty-two patients were treated with intravitreal bevacizumab (1.25 mg/0.05 mL), and subfoveal choroidal thickness was measured before and after treatment. Statistical analysis was performed to compare subfoveal choroidal thickness of CRVO and fellow eyes and to compare subfoveal choroidal thickness before and after intravitreal bevacizumab.
Results
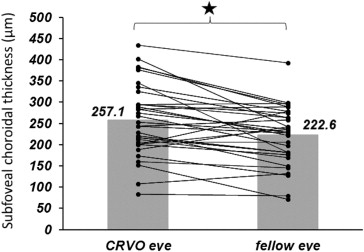
Mean subfoveal choroidal thickness measured in 36 eligible eyes of 36 patients was 257.1 ± 83.2 μm, which was significantly greater than that in fellow eyes (222.6 ± 67.8 μm; P < .01, paired t test). There was strong correlation between CRVO eyes and fellow eyes ( r = 0.79, P < .01). Mean subfoveal choroidal thickness after intravitreal bevacizumab was 227.7 ± 65.1 μm, which was significantly thinner than that before intravitreal bevacizumab therapy (266.9 ± 79.0 μm; P < .01, paired t test).
Conclusions
Subfoveal choroidal thickness of CRVO eyes was significantly greater than that of fellow eyes and decreased significantly after intravitreal bevacizumab treatment. Enhanced depth imaging optical coherence tomography can be used to evaluate choroidal involvement in CRVO and may assist noninvasive diagnosis and management of this disease.
Central retinal vein occlusion (CRVO) is a common cause of unilateral visual loss, and macular edema is a frequent cause of visual loss. Although it is well known that central macular thickness increases because of severe macular edema in CRVO, we could find no report on subfoveal choroidal thickness in patients with CRVO. Spaide and associates used spectral-domain optical coherence tomography (OCT) and developed a method termed enhanced depth imaging (EDI) OCT, which enables in vivo cross-sectional imaging of the choroid and measurement of the thickness of the choroid. Subsequently, the subfoveal choroidal thickness of patients with various diseases, such as central serous chorioretinopathy, macular hole, age-related macular degeneration, high myopia, and Vogt-Koyanagi-Harada disease, was reported. Further, certain reports noted changes in subfoveal choroidal thickness after treatment. Because macular edema in patients with CRVO seems to be related closely to vascular endothelial growth factor (VEGF) levels in the vitreous, inhibiting VEGF seems to be a reasonable therapeutic approach. Although many studies have reported that central macular thickness in CRVO decreases after anti-VEGF treatment, such as intravitreal bevacizumab therapy, we could find no reports of changes in choroidal thickness in CRVO patients after treatment. The aim of this study was to compare the choroidal thickness in the macular area between eyes with CRVO and unaffected fellow eyes and to investigate how subfoveal choroidal thickness changes after intravitreal bevacizumab treatment.
Methods
A retrospective analysis was performed for consecutive patients examined with unilateral CRVO at our retinal outpatient department in the Department of Ophthalmology of the University of Nagasaki from July 2010 through May 2012. The clinical examination for diagnosis of CRVO included measurement of best-corrected visual acuity, slit-lamp biomicroscopy with a contact lens or noncontact lens, indirect ophthalmoscopy, digital fluorescein angiography, and indocyanine green angiography (Heidelberg Retinal Angiography, Heidelberg Engineering, Heidelberg, Germany). We obtained best-corrected visual acuity measurements for analysis by using a Japanese standard decimal visual chart and the logarithm of the minimal angle of resolution scale. Patients with macular edema were treated with intravitreal bevacizumab (1.25 mg/0.05 mL). The intravitreal bevacizumab treatment followed the tenets of the Declaration of Helsinki, and approval was obtained from the Ethics Committee of Nagasaki University School of Medicine. Subfoveal choroidal thickness was measured using the Heidelberg Spectralis (Heidelberg Engineering) according to the EDI OCT technique described by Spaide and associates. The camera was positioned close enough to the eye to obtain an inverted image of the choroid. This image was averaged over 100 scans with the automatic averaging and eye-tracking features. The horizontal section going directly through the center of the fovea was selected. The resulting images were viewed and measured using the Heidelberg Eye Explorer software (version 1.6.40; Heidelberg Engineering). The choroid was measured from the outer portion of the hyperreflective line corresponding to the retinal pigment epithelium to the inner surface of the sclera. These measurements were obtained at the subfoveal choroid. Each image was measured by 3 independent observers (Y.M., R.U., E.T.), with discrepancies of more than 20% being resolved by open adjudication with the senior author (E.T.). The visual acuities are stated as decimal equivalents and logarithm of the minimal angle of resolution equivalents.
The data obtained were analyzed with descriptive statistics. Measurements of choroidal thickness and retinal thickness were analyzed using the Student t test. The Spearman rank correlation coefficient was used to evaluate the correlation between choroidal thickness of the CRVO eyes and that of the fellow eyes. For all tests, P < .05 was considered significant.
Results
There were 36 patients with unilateral CRVO; their mean age was 66 ± 15 years (range, 26 to 84 years). Twenty-six patients (72%) were women. The type of CRVO was ischemic in 10 (28%) patients and nonischemic in 26 (72%) patients. These patients had additional systemic diseases: 23 (64%) had hypertension and 8 (22%) had diabetes mellitus without retinopathy. The full thickness of the choroid could be visualized in all 36 eyes. Best-corrected visual acuity (logarithm of the minimal angle of resolution) before treatment ranged from −0.08 to 2.0 (median, 0.40). Twenty-two (61%) patients were treated with intravitreal bevacizumab to decrease macular edema, and subfoveal choroidal thickness was measured 1 month after intravitreal bevacizumab. The baseline demographic and clinical characteristics of the CRVO and fellow eyes are reported in the Table . A negative correlation was found between subfoveal choroidal thickness and axial length in the CRVO eyes ( r = 0.28, P < .05). Compared with fellow-eyes, the CRVO eyes showed significantly greater subfoveal choroidal thickness ( P < .01, paired t test). The mean ± standard deviation subfoveal choroidal thickness was 257.1 ± 83.2 μm in the CRVO eyes and 222.6 ± 67.8 μm in the fellow eyes ( Figure 1 ). A strong correlation was found between subfoveal choroidal thickness in the CRVO eyes and that in the fellow eyes ( r = 0.79, P < .01). The mean subfoveal choroidal thickness was 252.6 μm (ischemic) and 258.8 μm (nonischemic) in the CRVO eyes and 217.6 μm (ischemic) and 224.5 μm (nonischemic) in the fellow eyes. Similarly, CRVO eyes showed significantly greater subfoveal choroidal thickness ( P < 0.01, paired t test) in both the ischemic and nonischemic groups. There was no difference between the ischemic and nonischemic groups. Figure 2 shows EDI OCT images of the choroid of 1 eye with CRVO and that of the unaffected fellow eye. For CRVO eyes, subfoveal choroidal thickness was significantly greater before intravitreal bevacizumab therapy than after therapy ( P < .01, paired t test). The mean ± standard deviation subfoveal choroidal thickness in the CRVO eyes was 266.9 ± 79.0 μm before intravitreal bevacizumab and 227.7 ± 65.1 μm after intravitreal bevacizumab ( Figure 3 ). Similarly, there was no difference between the ischemic and nonischemic groups, and subfoveal choroidal thickness after intravitreal bevacizumab decreased significantly in both the ischemic (210.3 μm from 251.5 μm) and nonischemic (237.6 μm from 275.7 μm) groups ( P < .05, paired t test). Figure 4 shows EDI OCT images of the choroid of 1 eye with CRVO before and after intravitreal bevacizumab treatment.
| CRVO Group (n = 36) | Fellow Eve Group (n = 36) | Intravit real Bevacizumab Group (n = 22) | P Value | |
|---|---|---|---|---|
| Mean age ± SD (y) | 66 ± 15 | 66 ± 15 | 66 ± 14 | NS |
| Gender (female/male) | 26/10 | 26/10 | 16/6 | NS |
| Meant ± SD axial length (mm) | 23.2 ± 1.3 | 23.3 ± 1.2 | 23.1 ± 1.4 | NS |
| Type (ischemic/nonischemic) | 10/26 | 10/26 | 3/14 | NS |
| Systemic disease | ||||
| Hypertension (+/−) | 23/13 | 23/13 | 13/9 | NS |
| Diabetes Mellitus (+/−) | 8/23 | 3/23 | 7/15 | NS |