Fig. 8.1
Various indications for endothelial keratoplasty. Upper row (L–R) – Fuchs endothelial corneal dystrophy; pseudophakic corneal edema (posterior chamber intraocular lens in the anterior chamber). Lower row (L–R) – Congenital hereditary endothelial dystrophy; iridocorneal endothelial (ICE) syndrome
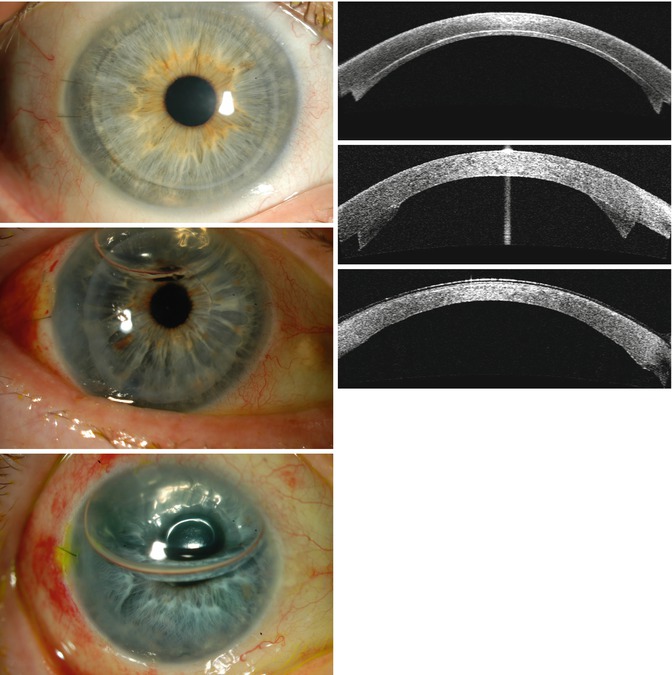
Fig. 8.2
Slit lamp and anterior segment optical coherence tomography images of 3 types of endothelial keratoplasty. First row: Descemet stripping automated endothelial keratoplasty (DSAEK). Second row: Descemet membrane automated endothelial keratoplasty (DMAEK), with residual air bubble 1 day after surgery. Third row: Descemet membrane endothelial keratoplasty (DMEK), with residual air bubble and bandage contact lens in place 1 day after surgery
8.2 Surgical Techniques
8.2.1 DLEK
The DLEK technique initially involved creation of a 9.0 mm scleral wound and a deep corneoscleral lamellar pocket down to about 75–85 % corneal depth along the entire length of the wound. The stromal dissection was completed to the limbus for 360° using semisharp dissectors to create a deep lamellar corneal pocket. The posterior stromal disc was trephined and excised. The donor corneoscleral tissue was mounted onto an artificial anterior chamber, and anterior stromal tissue was resected either manually or using a microkeratome, leaving approximately 150 μ of the posterior stroma. The donor tissue was trephined to the same diameter as the recipient bed, inserted into the anterior chamber, and attached using an air bubble. The scleral wound was closed with several interrupted sutures. The procedure was later performed through a 5–6 mm incision by folding the graft for insertion.
Although not ideal, DLEK was the first successful EK procedure. It certainly had several advantages over PK because there were no anterior corneal incisions and suturing of the graft. So, the visual recovery was better and the suture-related complications as seen with PK were prevented. The surgical difficulties involved with the procedure led to exploration of alternative approaches.
8.2.2 DSEK
The procedure comprises 3 steps: (a) preparation of a posterior lamellar graft, (b) removal of the host Descemet membrane and dysfunctional endothelium, and (c) insertion of the graft into the anterior chamber and positioning using air tamponade.
(a)
Donor preparation: The donor cornea is mounted on an artificial anterior chamber for lamellar dissection; then it is placed on a tissue-cutting block and trephined to the desired diameter (usually 8–9 mm) from the endothelial side. Methods of lamellar dissection include:
Manual dissection (Fig. 8.3, first row)—An initial 4–5 mm curvilinear incision is made at the limbus to a depth of 300–350 μm with a guarded diamond/Bevers’s blade. Short and long curved dissecting blades are used to extend the lamellar dissection 360° to reach to the limbus.
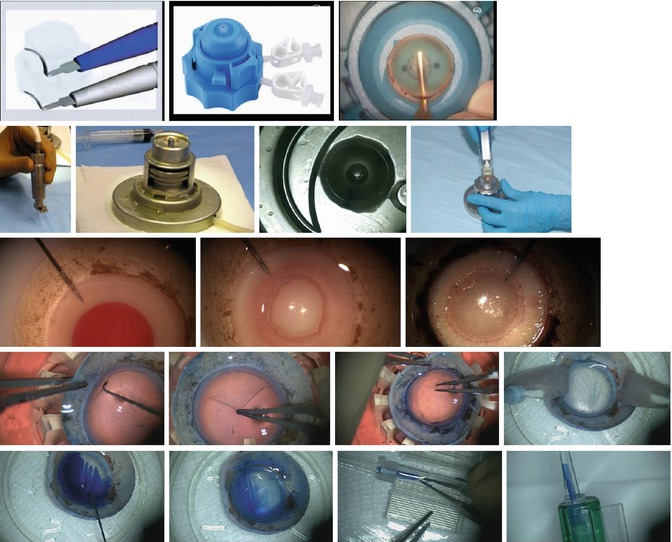
Fig. 8.3
Donor tissue preparation: equipment and methods. Upper row (L–R): Descemet stripping endothelial keratoplasty (DSEK): manual dissectors (DORC, the Netherlands); Barron disposable artificial anterior chamber (Katena Products); manual dissection with donor cornea mounted on artificial anterior chamber. Second row (L–R): Descemet stripping automated endothelial keratoplasty (DSAEK): microkeratome (Moria); reusable artificial anterior chamber (Moria); microkeratome-assisted donor dissection; Microkeratome (Gebauer, Germany). Third row (L–R): Descemet membrane automated endothelial keratoplasty (DMAEK) graft preparation: air is injected via a needle inserted through the peripheral scleral rim; this creates a big bubble; the big bubble is enlarged with more air to separate the Descemet membrane (DM) from the posterior stroma. Fourth row (L–R): Descemet membrane endothelial keratoplasty (DMEK) graft preparation: peripherally scored DM is separated from underlying stromal tissue circumferentially using a microfinger; DM is peeled in four quadrants leaving it attached at the center; final peel to free the center of the tissue; scrolled donor Descemet membrane and endothelium. Fifth row (L–R): DMEK graft insertion: trypan blue is being used to stain the DM scroll to improve visualization; the stained DM scroll; the tissue is being loaded into the cartridge of an intraocular lens inserter; the DM scroll within the inserter
Microkeratome dissection (Fig. 8.3, second row)—The donor dissection plane is created with a microkeratome. The microkeratome heads can be chosen (usually 300–350 μm) according to the desired plane of lamellar dissection. Microkeratome dissection produces a smoother and more regular dissection plane compared to manual dissection. Numerous eye banks have microkeratomes and provide pre-dissected tissue.
Femtosecond (FS) laser dissection (FS–DSEK)—The feasibility of using a femtosecond laser to create lamellar cuts was assessed, but the regularity of deep lamellar ablations required for posterior lamellar grafts was suboptimal and failed to produce encouraging results (Mehta et al. 2008; Cheng et al. 2008).
(b)
Stripping of the host Descemet membrane (Fig. 8.4, first row—first): Host DM is removed within an area corresponding to the graft diameter or slightly smaller. DM stripping is necessary in Fuchs endothelial dystrophy to remove the guttae; however, this step may be optional in conditions where the DM is optically clear and devoid of any structural alterations, such as in failed PK and pseudophakic corneal edema (Nottage and Nirankari 2012; Chaurasia et al. 2011a, b; Kobayashi et al. 2008).
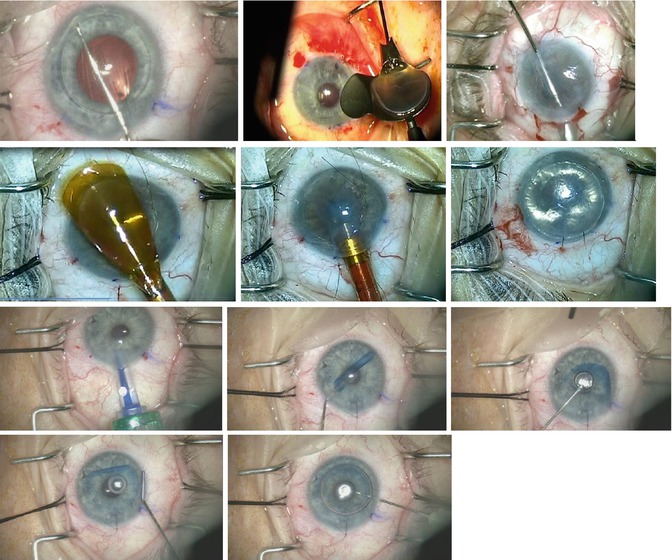

Fig. 8.4
Recipient preparation, graft insertion, and positioning. First row (L–R): Descemet membrane scoring; loading a DSEK graft into a Busin glide (Moria); pull-through method of graft insertion using a Busin glide and intraocular forceps. Second row (L–R): loading a DSEK graft into an EndoSerter (Ocular Systems, Winston-Salem, NC); graft insertion with EndoSerter; air tamponade. Third row (L–R): DMEK graft insertion; short bursts of balanced salt solution unfold the scrolled tissue; partial unfold achieved. Fourth row (L–R): unwrapping the scrolled DMEK donor tissue over a posterior air bubble (the air bubble helps anchor the graft in the correct position); final air injection to press the donor tissue against the recipient cornea
(c)
Graft insertion and positioning: The graft is inserted through a 3–5 mm incision using devices such as forceps, glides, and inserters. Currently used donor insertion instruments include:
Forceps—The donor tissue is folded into a 60/40 configuration (with the endothelium facing inward and protected with a small amount of viscoelastic) and inserted into the anterior chamber using atraumatic non-coapting forceps (e.g., Charlie II, Goosey, Kelman forceps) (Melles et al. 2002a; Price and Price 2005, 2006b).
Sheets glide—This method can be helpful in eyes with a shallow anterior chamber and predisposed to iris prolapse. The anterior chamber is maintained using an anterior chamber (AC) maintainer. A Sheets intraocular lens (IOL) glide is inserted halfway into the chamber, which serves to keep the iris behind. The donor graft is placed onto the glide with the endothelial side down protected with a generous amount of viscoelastic. An intraocular forceps is inserted through the site opposite to the main incision. The donor edge is grasped with the forceps and pulled inside the anterior chamber (pull-through method) (Mehta et al. 2007). Alternatively, the graft may be inserted through the main incision using a Sinskey IOL dialer (push-in method) without the use of an AC maintainer.
Busin glide (Fig. 8.4, first row—second)—This reusable funnel glide (Moria, Inc., Antony, France) curls the graft into a cylindrical shape to minimize endothelial trauma during insertion (Busin et al. 2008). The edge of the graft is grasped and pulled into the anterior chamber with an intraocular forceps introduced through the opposite site incision.
Suture pull–through—In this method (Macsai and Kara-Jose 2007), a10-0 Prolene suture is passed through a 5 mm superior lamellar incision and across the AC to exit through the cornea approximately 1 mm beyond the edge of stripped DM. The donor endothelium is coated with viscoelastic, and the second arm of the suture is passed through the periphery of the donor lenticule, entering from the endothelial side and exiting from the stromal side. It is then passed through the incision, across the AC and through the cornea 1 mm central to the edge of stripped DM. The donor lenticule is gently folded in half with the suture at the leading edge, and the anterior edge of the incision is lifted as both ends of the suture are pulled to guide the graft into the eye. The graft unfolds as the AC is filled with air, and it is secured by tying off the suture, which helps minimize the risk of graft dislocation.
Injectors/Inserters (Fig. 8.4, second row—first, second) – Several single-use devices have been designed to deliver the graft with minimal endothelial trauma (Khor et al. 2011; Kuo et al. 2008). Adoption has been limited by the cost and the good results surgeons have obtained with the other methods described above.
After the graft is inserted, it is positioned and attached to the host posterior stroma using an air bubble (Fig. 8.4, second row—third). Complete air fill in the anterior chamber is maintained for 10–12 min, followed by a partial air-fluid exchange to avoid risk of raised intraocular pressure (IOP) secondary to pupillary block. Alternatively, a prophylactic inferior iridectomy may be made prior to graft insertion to prevent pupillary block due to air. Postoperatively, supine positioning is maintained for 15–20 min or longer.
8.2.3 DMEK
The procedure involves isolation of donor DM and endothelium followed by inserting, unwrapping, and positioning in the proper orientation. Just as in DSEK, the central host DM is removed before inserting the donor tissue.
(a)
Donor preparation (Fig. 8.3, fourth row): Donor DM can be isolated by direct peeling (Lie et al. 2008) or by injection of air to create a big bubble (Venzano et al. 2010). Direct peeling has a higher success rate with less endothelial cell loss (Yoeruek et al. 2012). Giebel and Price described a direct peeling method called submerged cornea using backgrounds away (SCUBA) that has a success rate of almost 99 % (Giebel and Price 2009; Guerra et al. 2011a). The donor corneoscleral rim is submerged with the endothelial side up in a viewing chamber filled with corneal storage solution, or it can be placed on a cutting block. The DM is lightly scored 1–2 mm inside the trabecular meshwork using a Y-hook. Trypan blue staining improves visualization of the scored edge, which is then lifted circumferentially with a microfinger (Moria Inc.). The edge of the DM is grasped with a blunt forceps. DM is partially peeled in four quadrants, leaving the center part attached. The membrane is floated back into position and the donor is trephined lightly into the stroma. The donor is placed back in the viewing chamber for a final peel to detach DM centrally. The detached DM spontaneously forms a scroll with the endothelium on the outside. The DM scroll is placed either in the storage medium or used immediately for transplantation.
The DM thickens with age, and so grafts from older donors generally scroll less tightly which makes graft unwrapping easier during surgery. Therefore, donor tissue over 40 years of age is preferable for DMEK. As with DSEK, DMEK donor tissue can be prepared several days before the surgical procedure (Feng et al. 2013a).
(b)
Graft insertion, unfolding, and positioning: The DM scroll can be loaded into a glass pipette or IOL cartridge and injector and inserted through a 2.8-mm-wide corneal incision. Various types of IOL cartridges are suitable for graft delivery including Softec (Lenstec, Inc, St. Petersburg, FL, USA); Carl-Zeiss inserters (Jena, Germany) and Viscoject (Medicel AG, Wolfhalden, Switzerland) (Fig. 8.3, fifth row). Graft adhesion is reported to be better with the use of IOL cartridges that form a closed system without any addition of viscoelastic (Chaurasia et al. 2014b).
Several graft unfolding maneuvers are available (Fig. 8.4, third and fourth row). Liarakos et al. (2013) described 4 standard and 3 auxiliary techniques for unfolding the graft in the anterior chamber depending upon the orientation and how tightly the DM is curled. Essentially, the DM scroll in the anterior chamber is opened using short quick bursts of BSS. A portable slit beam or slit beam or optical coherence tomography attachment on the operating microscope can be used to confirm the graft orientation (Burkhart et al. 2013). After the scroll is partially unwrapped, a small air bubble is injected under the donor to secure the orientation (Guerra et al. 2011a). The recipient corneal surface is stroked to center and unfold the graft completely, followed by air fill in the anterior chamber. The patients are advised to keep supine position for 60 min to allow for donor adherence.
8.2.4 Hybrid Techniques
Hybrid techniques have been developed to combine the optical outcomes of DMEK with the easier handling of DSEK. Studeny described transplantation of a posterior corneal lamellae consisting of endothelium and DM centrally with a stromal supporting rim (DMEK-S), and McCauley et al. described a partially automated variation (DMAEK) (Studeny et al. 2010; McCauley et al. 2009). The bare central endothelium and DM provide excellent optical outcomes, comparable with those of successful DMEK patients, while the stromal rim fixes the thin, fragile central part, helping to maintain its shape and prevent scrolling.
The donor tissue is dissected as in DSEK. Then the DM is detached from the posterior stroma using a big bubble technique (Fig. 8.3, third row). An incision is made through the stromal bed overlying the big bubble, and the disk of the posterior stroma is excised within the area of the detached DM. The donor tissue is inserted into the eye using the pull-through technique with Busin glide, and air is injected to attach the donor against recipient stroma. Donor insertion and positioning is easier than DMEK, because the tissue unfolds spontaneously, but the donor tissue preparation is more complicated and the rate of tissue loss is higher, which has limited the adoption.
8.2.5 Ultrathin DSAEK
Some surgeons have reported better visual acuity and faster visual recovery with thinner endothelial grafts (Taravella et al. 2013). Busin et al. described a microkeratome-assisted double-pass method for obtaining ultrathin posterior lamellar grafts (<100 μ) (Busin et al. 2013). The first pass is done with a 300 or 350 μm microkeratome head to debulk the cornea. The thickness of the residual bed determines the selection of the microkeratome head for the second pass, which is necessarily made going the opposite direction to help avoid perforation because the blade goes deepest at the beginning of the pass. Nomograms, which take into account donor thickness, corneal storage medium, pressurization of the artificial anterior chamber, and cutting speed, have been devised to help select the appropriate microkeratome head size to obtain thin EK donor tissue with either single- or double-pass techniques (Taravella et al. 2013). The risk of tissue loss from perforation is higher with ultrathin grafts compared with standard DSEK grafts.
8.3 Surgical Considerations with Ocular Comorbidities
EK is most straightforward in an eye with a normal anterior segment and a stable posterior chamber IOL. Because of the well-known advantages of DSEK over PK, the indications for EK have expanded to eyes with anterior segment complexity such as those with pupillary abnormalities, peripheral anterior synechiae, glaucoma-filtering procedures, glaucoma tube shunts, prior PK, or anterior chamber IOL. While DSEK can be attempted successfully in all mentioned conditions, the decision for DMEK is more reserved because the DM graft is delicate and requires more manipulation in comparison to DSEK. When deciding about the type of EK, the advantage of one procedure needs to be balanced with the technical ease of the procedure in an individual case scenario.
8.3.1 Aphakic Eyes with Complete or Partial Aniridia
In eyes with aphakia with complete or partial aniridia, a potential concern is intraoperative or postoperative graft detachment, which may result in graft dislocation into the posterior segment. Several options are available to manage this situation depending upon the extent of iris abnormality (Price et al. 2007). A posterior chamber IOL (sulcus/scleral fixated) with or without iris reconstruction/pupilloplasty can be planned simultaneously or a few weeks before the DSEK procedure. A Sheets IOL glide can be used during graft insertion to direct the graft and avoid posterior migration. A temporary fixation suture through the recipient cornea and the endothelial graft can be placed at the end as a measure to prevent posterior dislocation of the graft (Macsai and Kara-Jose 2007). In eyes with large iris defects, the host DM stripping should be avoided or performed cautiously to prevent the fragments from falling into the posterior cavity. Prolonged air tamponade and supine positioning may be additional measures to promote graft adhesion.
In aniridic eyes with an artificial iris implant, the graft can slide between the edge of the implant and the wall of the eye and fall into the posterior segment. To prevent this from happening, air should be injected under the graft before it is released from the insertion forceps, and a temporary fixation suture should be used to hold the graft in place (Price et al. 2007). Alternatively, the graft can be inserted with a suture pull-through technique and the pulling sutures can be used to affix the graft until adherence is confirmed in the postoperative period (Macsai and Kara-Jose 2007).
8.3.2 Phakic Eyes
In eyes with significant cataract, a triple procedure (cataract surgery with DSEK or DMEK) is the preferred approach. It is also advantageous with DSEK because cataract extraction deepens the anterior chamber and facilitates unfolding the graft. In patients with endothelial disease and clear crystalline lens, one may contemplate endothelial keratoplasty alone (Tsui et al. 2011). It is prudent to avoid iatrogenic damage to the clear lens and the endothelial graft by avoiding anterior chamber fluctuations.
As with PK, following EK, the rate of cataractogenesis is accelerated as a result of intraocular manipulations during surgery and the postoperative use of steroids. The probability of cataract progression requiring extraction is significantly associated with the age of the patient (Price et al. 2010).
8.3.3 Prior Glaucoma Filtering/Tube Surgery
Here, there may be difficulties in achieving an air fill in the anterior chamber as the injected air finds its way into the subconjunctival space through the ostium and so may require several attempts. Also, after obtaining adequate air tamponade, IOP needs be strictly monitored to avoid extremes of high pressure for prolonged periods which can be detrimental to the already compromised optic nerve. Rarely in cases where the air just escapes easily and the pressure cannot be increased enough to firm the eye, a few drops of viscoelastic can be placed over the end of the tube or ostium of the filter to block the flow. Viscoelastic should not be used until the graft is in place to prevent it from coating the graft interface. Once the patient sits up, the viscoelastic will fall away from superior tubes and ostia; so postoperative IOP spikes should not be an issue.
In the eyes with glaucoma drainage devices, it is important to ensure that mechanical contact between the graft and tube is avoided by proper trimming and repositioning of the tube, if required.
8.3.4 Vitrectomized Eyes
Eyes with prior vitrectomy and associated iris/zonular defects may have difficulties in graft adhesion as air may escape into the vitreous cavity increasing the risk of appositional angle closure/graft detachments. Similar to other situations, prolonged tamponade may facilitate in avoiding these problems.
8.3.5 Failed Prior PK
DSEK under a failed graft can successfully restore the graft clarity and avoid repeat PK. However, in situations where the refractive result of the prior PK was unsatisfactory, it would be better to consider a repeat PK rather than EK.
As mentioned earlier, the DM may be left intact in a failed graft if it does not show any abnormalities (Price and Price 2006c; Nottage and Nirankari 2012). This prevents the weakening of the graft–host junction that may occur inadvertently during the stripping maneuver. In eyes with prior therapeutic PK, the DM may be hazy and require removal for optimal results (Chaurasia et al. 2014a). If stripping of DM is planned, it should be made internal to the graft–host junction or even in a small area overlying the pupil to avoid disrupting the incision.
The graft can be oversized, undersized, or same sized. Oversizing provides the advantage of a larger endothelial cell reserve, while undersizing avoids the need for the EK graft to conform to the irregularity at the graft-host junction that may interfere with the graft attachment process.
One of the most remarkable findings in a series of 60 eyes with DSEK after failed PK was that neither neovascularization nor the number of previous graft failures increased the risk of graft failure. The only preoperative characteristic associated with increased risk of graft failure was previous filtration surgery, either trabeculectomy or tube shunt (Anshu et al. 2011).
8.3.6 Iridocorneal Endothelial (ICE) Syndrome
These eyes may have a very shallow anterior chamber because of broad peripheral synechiae (Price and Price 2007). In addition, they may have undergone a prior glaucoma filtering surgery/drainage surgery for IOP control. Extensive synechiolysis may be required for deepening the anterior chamber. Postoperatively, frequent follow-up and aggressive control of IOP are needed for graft survival (Chaurasia et al. 2013).
8.3.7 Pediatric Endothelial Keratoplasty
DSEK can be more challenging in pediatric eyes as compared with adults. The main reasons for performing EK in pediatric eyes are for failed graft, congenital hereditary endothelial dystrophy, and pseudophakic corneal edema. Surgical challenges involve insertion and unfolding of the donor tissue in the small anterior chamber of a child, avoiding trauma to the crystalline lens, postoperative positioning, and anesthesia issues (Ramappa et al. 2012). Pediatric eyes can have a positive vitreous pressure which can make the surgical maneuvers difficult. Discussing with the anesthetist the need for hypotensive anesthesia during graft insertion and use of an anterior chamber maintainer during the surgery helps in maintaining the anterior chamber during the surgery.
8.4 Surgical Outcomes
8.4.1 Visual Acuity
When compared with PK, the visual recovery is remarkably rapid, occurring within a few weeks of EK, and mean visual outcomes continue to further improve for up to several years afterward. The average Snellen corrected distance visual acuity (CDVA) reported after DSEK has ranged from 20/30 to 20/60 in different studies, with variable follow-up periods (Lee et al. 2009; Li et al. 2012b). Several factors may interfere with complete visual recovery: graft folds, thickness irregularity, centration, interface haze, and residual anterior abnormalities in the host cornea.
DMEK eliminates any thickness variation or stromal interface and thereby results in better and faster visual recovery with fewer higher order aberrations from the posterior surface of the cornea (Rudolph et al. 2012). Most patients achieve 20/25 or better vision within several months with DMEK (Giebel and Price 2009; Guerra et al. 2011a). Like DMEK, DMAEK also provides superior visual recovery with high rates of 20/25 or better vision (Taravella et al. 2013). The relationship between DSEK graft thicknesses and visual acuity has been debated (Shinton et al. 2012; Seery et al. 2011). While some believe that thinner grafts are associated with better vision, others have failed to establish this association. Thinner, well-centered, and planar grafts may induce fewer higher order optical aberrations and contribute to superior visual results. Busin et al. have reported excellent visual outcomes with ultrathin DSAEK (Busin et al. 2013).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


