Endoscopic pituitary surgery has been gaining wide acceptance as the first-line treatment of most functional pituitary adenomas. This technique has many advantages over traditional procedures, and growing evidence supports its use for endocrine control of functioning tumors. This article reviews data on the different modalities of treatment of functioning pituitary adenomas and compares the results. Endoscopic pituitary surgery controls tumor growth and endocrinopathy as well as or better than other treatment modalities. Complication rates are low and patient recovery is fast. Furthermore, surgery provides a means of achieving prompt decompression of neurologic structures and endocrine remission.
| EBM Question | Level of Evidence | Grade of Recommendation |
|---|---|---|
| Does endoscopic surgery improve control of functional tumors? | 4 | C |
With rapid advances in technology and endoscopic techniques in the 1980s and early 1990s, endoscopic sinus surgery expanded beyond the realms of the paranasal sinuses and into the skull base. In the early 1990s, endoscopic approaches to the skull base were described. Jankowski and colleagues reported on the endoscopic approach to the sella in 3 patients in 1992. Sethi and Pillay at the Singapore General Hospital reported on a series of 40 patients using an endonasal transsphenoidal technique for the management of sellar lesions. A series of 45 cases of endoscope-assisted pituitary surgery was reported by the Pittsburgh group in 1996, and in 1999, Cappabianca and colleagues reported a series of purely endoscopic approaches to the sella. Many proponents of the endoscopic approach reported superior panoramic vision and improved appreciation of the relationships of the structures surrounding the sella turcica compared with the operating microscope. These advantages facilitated further developments in techniques and reconstruction to allow surgery in structures beyond the sella. The concept of extended approaches to the skull base evolved, enabling the endoscopic surgeon to manage larger tumors with suprasellar extension and local invasion.
The ability of endoscopic pituitary surgery to control hormonal dysfunction holds great appeal because it may reduce the risks associated with other surgical approaches and addresses many of the disadvantages of radiotherapy and medical therapy. After the initial reports of successful endoscopic surgery of pituitary tumors, comparative studies emerged, supporting several advantages over the microsurgical approach. These advantages include reduced blood loss and analgesia requirements. Shorter hospital admissions have also been reported, and intraoperative complications were reduced in several reports.
Epidemiology
Pituitary adenomas are the most common neoplasms of the pituitary gland. These tumors are more common in females and increase in incidence with age. Although rare in children, these adenomas have been reported to be present in almost 30% of the older population, although most are asymptomatic. Pituitary adenomas account for approximately 5% to 15% of symptomatic intracranial tumors. Although most cases are sporadic, a small proportion is related to syndromes such as the multiple endocrine neoplasia type I syndrome, an autosomal dominant syndrome of pituitary adenomas, parathyroid hyperplasia, and pancreatic tumors.
Adenomas that secrete hormone products account for approximately 90% of pituitary adenomas, according to a report from 1995. This number is now likely to be much lower with the widespread use of imaging, which now detects incidental tumors as small as 2 mm in 10% to 20% of the population. Many functional tumors do not produce hormone products in significant quantities to cause clinically manifest endocrinopathy.
Pathophysiology
Pituitary adenomas are classified according to their secretory hormone products. They are also arbitrarily classified into microadenomas (<10 mm in size) and macroadenomas (≤10 mm) ( Fig. 1 ). Secretory hormones include prolactin, growth hormone (GH) ( Fig. 2 ), corticotropin, and, rarely, thyrotropin (TSH). With the exception of incidentalomas, secretory adenomas often present as endocrinopathies and are microadenomas at diagnosis. However, nonfunctional tumors usually present as macroadenomas when they are large enough to produce compressive symptoms (see Fig. 1 ).
Pathophysiology
Pituitary adenomas are classified according to their secretory hormone products. They are also arbitrarily classified into microadenomas (<10 mm in size) and macroadenomas (≤10 mm) ( Fig. 1 ). Secretory hormones include prolactin, growth hormone (GH) ( Fig. 2 ), corticotropin, and, rarely, thyrotropin (TSH). With the exception of incidentalomas, secretory adenomas often present as endocrinopathies and are microadenomas at diagnosis. However, nonfunctional tumors usually present as macroadenomas when they are large enough to produce compressive symptoms (see Fig. 1 ).
Clinical features
Prolactin-Secreting Adenomas
In women who have not had a recent pregnancy, galactorrhea is the most common endocrinologic sequelae of prolactinomas. This condition may be accompanied by amenorrhea (Forbes-Albright syndrome). Men present with sexual dysfunction, infertility, obesity, and gynecomastia.
ACTH-Secreting Adenomas
In patients who are not treated with steroids, Cushing syndrome (the clinical manifestations of adrenal glucocorticoid hypersecretion) is most commonly caused by a pituitary adenoma. This scenario, termed Cushing disease, is more common in females. Presenting features include weight gain, widening of the face (moon facies), mood alterations, diabetes, and hypertension.
GH-Secreting Adenomas
GH-secreting adenomas present with acromegaly in adults. There is coarseness of facial features with enlargement of hands and feet, along with hirsutism, diabetes, hypertension, and hypertrophic arthropathy. As the dysmorphic features develop gradually over years or decades, patients tend to present with larger tumors than those with other secretory adenomas.
TSH-Secreting Tumors
TSH-secreting tumors account for less than 3% of pituitary adenomas. They present with thyrotoxicosis.
Visual Symptoms
Nonfunctional adenomas tend to present as macroadenomas causing visual symptoms. However, neural compression can also be a feature of functional adenomas. Classically, there is compression of the optic chiasm producing bitemporal visual field deficits. As the adenoma compresses the chiasm from below, the superior bitemporal quadrants tend to be affected more than the inferior quadrants. Long-standing compression of optic axons may lead to reduced visual acuity, and monocular deficits may result from compression of the lateral aspect of the chiasm or compression of only one optic nerve. Ophthalmoplegia may result from cavernous sinus involvement, with the third cranial nerve being the most commonly affected.
Pituitary Apoplexy
Sudden enlargement of a pituitary adenoma resulting from hemorrhage or infarction may result in headache, altered consciousness, and ophthalmoplegia, with or without visual loss. This condition may be fatal and usually has no identifiable precipitants. Known predisposing factors include head trauma, radiation treatment, bromocriptine, pregnancy, surgery, and anticoagulation.
Investigation
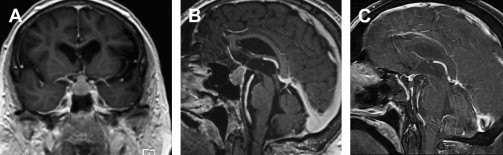
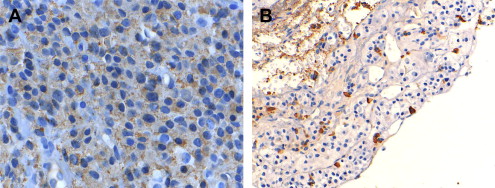
Investigations for pituitary adenomas include a comprehensive visual assessment, hormone analysis, and radiology. These investigations are important to characterize the tumor and to establish a baseline from which the results of treatment can be measured. Secretory tumors may not produce clinical endocrinopathy and are thus diagnosed on biochemical analysis. Prolactin-secreting tumors are diagnosed when serum levels are 10 times more than the normal limit. The level of prolactin is increased during pregnancy and lactation and when a tumor (functional or not) compresses the pituitary stalk and reduces the dopaminergic inhibition of prolactin secretion. Final histopathologic tests can distinguish between hyperplasia and true prolactinomas ( Fig. 3 ). High-field magnetic resonance imaging with and without gadolinium is the imaging modality of choice, and a computed tomographic scan may be helpful in preoperative planning ( Fig. 4 ). Pituitary adenomas often are very large ( Fig. 5 ) and display bone erosion ( Fig. 6 ).
Management
As well as the control of hormonal secretion, the goals of treatment of pituitary adenomas include decompression of the optic nerve and other neurologic structures by reduction of tumor size, total resection in some cases, and the access to tissue for a histologic diagnosis. Other goals of surgery include minimization of complications from intervention and prevention of disease recurrence. Not all these goals are sufficiently addressed by all treatment modalities, so treatment choices are made by balancing the priorities and the perceived risks and disadvantages (see Fig. 5 ).
Treatment of pituitary adenomas is pursued more vigilantly than many other benign tumors because the location of the pituitary complex predisposes tumors in that region to produce neurologic complications that may not be reversible, particularly visual deficits. Because most adenomas are functional, treatment is necessary for control of hormonal dysfunction (see Fig. 4 ). The risk of pituitary apoplexy adds impetus for treatment.
Prognosis and natural history
The natural history of pituitary adenomas is not well understood. Karavitaki and colleagues reported on a series of nonfunctioning tumors observed over an average of 42 months. The investigators found that of the 16 microadenomas and 24 macroadenomas, the 48-month probability of enlargement was 19% for microadenomas and 44% for macroadenomas. Of the tumors that demonstrated growth, 57% had associated deterioration of vision.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


