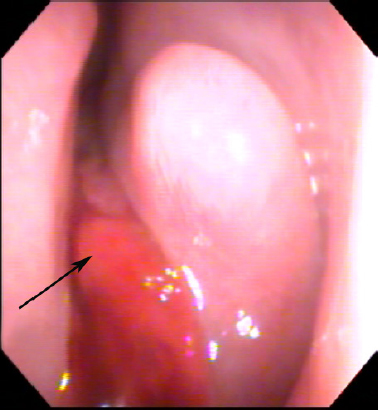
30 Endoscopic and Endoscopic-Assisted Skull Base Surgery for Anterior Skull Base Malignancy: Management Rationale Sinonasal and skull base malignancies (SSBM) are relatively rare and comprise approximately 1 to 3% of all head and neck cancers.1,2 Proximity of the tumor to vital neurovascular structures and insidious growth patterns result in advanced stage presentation. This can complicate the management of these patients, and necessitates a comprehensive evaluation. The vast majority of cases involve squamous cell carcinoma (SCC). Adenocarcinoma, adenoid cystic carcinoma, sinonasal undifferentiated carcinoma (SNUC), esthesioneuroblastoma, and other histopathologies comprise the remainder of cases.3–5 Although the use of an open transfacial or craniofacial approach is a time-honored method, endoscopic methods have been gaining favor for several reasons including but not limited to: increased experience in endoscopic surgery, growing understanding of the surgical outcomes, and the pursuit of reduced patient morbidity. The knowledge gap of our understanding of effectiveness of open approaches and those using endoscopic methods has been closing steadily. It is important to note that while endoscopic techniques are growing, being able to execute an open approach or collaborate with a colleague who can perform them is important for patient care. In the end, the goal is curing or reducing the patient’s cancer burden with the least amount of morbidity. Failure of local control has been established as a primary cause of death,6,7 underscoring the importance of complete extirpation. The exact approach used depends on the location and degree of spread of the primary tumor and involvement of vital structures. For instance, the anterior craniofacial approach is used for tumors involving the sinonasal cavity and anterior cranial fossa. Treatment using open approaches alongside appropriate adjuvant therapies has been successful in achieving variable 5-year survival rates, in some studies ranging from 40 to 70%.8–11 However, open procedures can have undesirable results in terms of postoperative appearance. These patients may suffer loss of function because of alterations of the facial contour, nerve sacrifice, and loss of velopharyngeal competence, among other deficits. Endoscopic approaches have been used to reduce potential morbidities associated with transfacial and other access-related incisions (e.g., oral cavity, and oropharynx) while providing other enhancements to surgical access. Continued refinements in equipment and technique, as well as ongoing outcomes reporting, have allowed endoscopic surgeons to help create new branch points in the surgical paradigm for SSBM. A thorough history and physical examination in a patient with sinonasal neoplasm can provide vital clues regarding the extent of the lesion. Nasal obstruction, epistaxis, anosmia, recurrent sinusitis, and facial pain and pressure are common symptoms and may allow the lesion to masquerade as a benign pathology. Endoscopic examination of the nasal cavity is essential (Fig. 30.1), but may be obscured by the presence of edema or secretions. The tumor bulk will also obscure the extent of involvement. Oral and oropharyngeal fullness can indicate mass effect from the tumor and extension beyond the sinonasal cavity. Trismus is another sign of an advanced lesion, and can signify involvement of the pterygoid fossa, infratemporal fossa, or beyond. Careful palpation of the cervical lymph nodes is also important. The likelihood of cervical lymphadenopathy at presentation varies depending on the pathology but can be as high as approximately 20%, as for esthesioneuroblastoma.12,13 Figure 30.1 Example of endoscopic examination of sinonasal malignancy. Endoscopic examination of the nasal cavity is important in the assessment of sinonasal malignancies. Image shows the clinical examination of a patient with esthesioneuroblastoma (arrow) extending into the left nasal cavity from the anterior skull base. Ophthalmological examination should be undertaken in patients with possible or known orbit involvement. Proptosis, decreased visual acuity, reduced ocular motility, epiphora, diplopia, pupil asymmetry, or visual field defects may indicate involvement of the orbit and visual system. These findings can help determine whether preservation of the orbit and its contents is feasible, modify patient counseling, and fine-tune the surgical approach. It can also help determine whether the approach should be open, endoscopic, or endoscopic-assisted. Involvement of the orbit is not necessarily an indication to routinely sacrifice the orbit, as this varies based on patient and disease characteristics.14 Neurological examination should be thorough and tailored for the patient. Cranial nerve function should be carefully assessed as deficits may indicate extent of the tumor including the possibility of perineural spread, a poor prognostic sign. Depending on the extent, a full neurological assessment may be necessary, including understanding of the handedness of the patient. With transcranial involvement, early involvement of a neurosurgical colleague in the evaluation and management of the patient will make treatment planning more effective. Imaging is necessary for staging and determining the extent of a lesion. Understanding local, regional, and distant involvement is critical to patient management. Baseline studies are useful to understand a patient’s response to treatment. Of the imaging modalities, computed tomography (CT) scan is excellent for delineating bony changes surrounding a neoplasm and assessment of orbital involvement. The use of intravenous contrast permits assessment of tumor vascularity and involvement of the carotid vasculature. Magnetic resonance imaging (MRI) provides superior soft tissue characterization compared with CT, but poor for bony architecture; this is especially useful to understand dural and transcranial involvement, as well as perineural extension. In addition, T2-weighted imaging can differentiate entrapped secretions from tumor to better delineate disease extent. Positron emission tomography (PET) is employed at many centers for staging of regional and distant metastasis. Alternately, this can be accomplished with whole-body CT or MRI. Tissue diagnosis is essential to management of a sinonasal malignancy. Biopsy with biting forceps under endoscopic vision will generally provide appropriate tissue for pathological analysis. Endoscopic biopsy in the office setting—using local anesthetic and decongestion—should be considered if the patient is amenable to an office-based biopsy and the tumor does not appear to be vascular; vascular tumors can bleed uncontrollably and can be difficult to manage in an office setting. If a vascular tumor is suspected, or the patient is not amenable to an office-based biopsy, then the biopsy should be performed in the operating room where bleeding can be controlled and the patient can be kept comfortable. Depending on the clinical picture, imaging may be obtained first to assess the vascularity and aid in decision making for tissue diagnosis. Having the patient perform a Valsalva maneuver while observing the mass for engorgement can help assess for intracranial or venous connection, and may further help determine the safest place to perform a biopsy. If doubt remains, fine-needle aspiration of the mass may be performed, but success with this method depends on sampling and expertise of the pathologist reading the needle aspirate. There are many histopathological varieties of sinonasal malignancy. With the exception of esthesioneuroblastoma, they share a staging system set forth by the American Joint Committee on Cancer (Table 30.1).15 Recent revisions to the primary lesion staging criterion emphasize the importance of determining the extent of invasion and involvement of adjacent structures. In multiple series, SCC represents the most common variety of sinonasal malignancy.1,16 Numerous environmental agents have been described as risk factors including aflatoxin, nickel, chromium, and aromatic hydrocarbons.17 Cervical metastases are present in approximately 10% of patients at initial diagnosis and are an independent prognostic indicator.1,18,19 SCC may also arise from inverted papillomas of the sinonasal regions; such lesions demonstrate more aggressive local growth and a tendency for distant metastatic spread.20 This histopathology comprises approximately 10% of sinonasal malignancies rendering it the second most common variety.4 Chronic exposure to various dust particles of wood, leather, and textiles is an associated risk factor. Sinonasal adenocarcinoma is characterized by aggressive local growth and infrequent metastasis. SNUC was previously classified as a form of high-grade epithelial malignancy with possible neuroendocrine differentiation but is now considered a distinct carcinoma of uncertain histogenesis. The tumor often presents with a rapid onset of symptoms relative to the slow and insidious nature of other sinonasal malignancies. Grossly, SNUC is characterized by a high degree of local invasiveness with involvement of multiple anatomic subsites. Histologically, there is a dense hypercellular proliferation, a high mitotic rate, and a lack of squamous or glandular differentiation.21 There is no specific characteristic immunohistochemical staining pattern, but these studies are crucial to rule out other entities on the differential diagnosis, namely, esthesioneuroblastoma.
Patient Evaluation
History and Physical Examination
Imaging
Biopsy
Pathology
Squamous Cell Carcinoma
Adenocarcinoma
Sinonasal Undifferentiated Carcinoma
Primary Tumor (T) | |
TX | Primary tumor cannot be assessed. |
T0 | No evidence of primary tumor. |
Tis | Carcinoma in situ. |
Maxillary Sinus | |
T1 | Tumor limited to maxillary sinus mucosa with no erosion or destruction of bone. |
T2 | Tumor causing bone erosion or destruction including extension into the hard palate and/or middle meatus, except extension to posterior wall of maxillary sinus and pterygoid plates. |
T3 | Tumor invades any of the following: bone of the posterior wall of the maxillary sinus, subcutaneous tissues, floor or medial wall of orbit, pterygoid fossa, ethmoid sinuses. |
T4a | Moderately advanced local disease. Tumor invades anterior orbital contents, skin of cheek, pterygoid plates, infratemporal fossa, cribriform plate, sphenoid or frontal sinuses. |
T4b | Very advanced local disease. Tumor invades any of the following: orbital apex, dura, brain, middle cranial fossa, cranial nerves other than maxillary division of trigeminal nerve (V2), nasopharynx, or clivus. |
Nasal Cavity and Ethmoid Sinuses | |
T1 | Tumor restricted to any one subsite,a with or without bony invasion. |
T2 | Tumor invading two subsites in a single region or extending to involve an adjacent region within the nasoethmoidal complex, with or without bony invasion. |
T3 | Tumor extends to invade the medial wall or floor of the orbit, maxillary sinus, palate, or cribriform plate. |
T4a | Moderately advanced local disease—tumor invades any of the following: anterior orbital contents, skin of nose or cheek, minimal extension to anterior cranial fossa, pterygoid plates, sphenoid or frontal sinuses. |
T4b | Very advanced local disease—tumor invades any of the following: orbital apex, dura, brain, middle cranial fossa, cranial nerves other than V2, nasopharynx, or clivus. |
Adapted from reference 15.
Note: This system does not apply to nonepithelial tumors such as those of lymphoid tissue, soft tissue, bone, cartilage, and mucosal melanoma.
Esthesioneuroblastoma
This uncommon malignancy is thought to arise from the neuroepithelium of the olfactory bulb and is located at the interface of sinonasal cavity and the anterior skull base. The most widely used classification scheme is the Kadish staging system22 with stage A lesions confined to the nasal cavity, stage B lesions involving the paranasal sinuses, and stage C lesions extending to include regions outside of the sinonasal cavities. The value of the Kadish staging system in prognostication was confirmed by multiple reports and it is highly utilitarian in its simplicity.
Esthesioneuroblastoma can occur at any age but demonstrates a bimodal distribution with peak incidence in the second and sixth decades of life, without a gender predilection.23 The tumor demonstrates an insidious growth pattern often resulting in presentations at advanced stages with extensive local destruction and tumor involvement. Approximately 5 to 8% of cases have cervical lymphadenopathy at the time of diagnosis and 20 to 25% of patients eventually develop regional metastatic disease. Note that 62% of esthesioneuroblastoma cervical metastases occur 6 months or more after primary treatment.13 This pattern of delayed regional metastasis may be because of lymphatic micrometastases undetected at the time of initial staging. The ideal treatment of the clinically negative neck in patients with esthesioneuroblastoma is under investigation.
Other Pathologies
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree