Purpose
To compare the diurnal and nocturnal effects of latanoprostene bunod 0.024% solution with timolol maleate 0.5% solution on intraocular pressure (IOP) and ocular perfusion pressure.
Design
Prospective, open-label randomized crossover trial.
Methods
Twenty-five patients (aged 43–82 years) with ocular hypertension or early primary open-angle glaucoma were enrolled. Baseline IOP and blood pressure were measured in a sleep laboratory every 2 hours in the sitting and supine positions during the 16-hour diurnal/wake period and in the supine position during the 8-hour nocturnal/sleep period. Subjects were randomly assigned to bilateral treatments of latanoprostene bunod at 8 PM or timolol at 8 AM and 8 PM. The second laboratory recording occurred after the 4-week treatment. Subjects were crossed over to the comparator treatment for 4 weeks before the third laboratory recording. Mean IOP and calculated ocular perfusion pressure were compared for the diurnal and nocturnal periods.
Results
Twenty-one subjects completed the study. Both treatments reduced diurnal sitting and supine IOP compared to baseline by 2.3–3.9 mm Hg (all P < .001) with no statistically significant difference between the 2 treatments. Nocturnal IOP under latanoprostene bunod treatment was 2.5 ± 3.1 mm Hg (mean ± SD) less than baseline ( P = .002) and 2.3 ± 3.0 mm Hg less than timolol treatment ( P = .004). Latanoprostene bunod treatment resulted in greater diurnal sitting and supine ocular perfusion pressures compared with baseline ( P ≤ .006) and greater nocturnal ocular perfusion pressure compared with timolol treatment ( P = .010).
Conclusions
During the nocturnal period, latanoprostene bunod caused more IOP reduction and more increase of ocular perfusion pressure than timolol.
Lowering intraocular pressure (IOP) may delay the onset of glaucoma in ocular hypertensive patients and slow the disease progression in patients with existing glaucoma. Whereas the highest IOP in patients with ocular hypertension and primary open-angle glaucoma frequently occur outside the diurnal/wake period, currently available topical medications to treat these patients have shown variable IOP-lowering efficacies during the nocturnal/sleep period compared to their diurnal efficacies. Prostaglandin analogues (including latanoprost, travoprost, and bimatoprost) applied once daily in the evening were shown to be effective in lowering IOP for 24 hours, but with less nocturnal efficacy than the diurnal efficacy. Timolol, a β-adrenergic antagonist, applied in gel form once daily in the morning, had very limited IOP reduction during the nocturnal period compared to its diurnal efficacy.
Latanoprostene bunod (Bausch & Lomb, Bridgewater, New Jersey, USA) is a new nitric oxide–donating prostaglandin F 2α analogue with unique biological properties. In situ, latanoprostene bunod is rapidly metabolized to latanoprost acid, a prostaglandin agonist, and butanediol mononitrate, a nitric oxide (NO)-donating moiety. Latanoprost acid (as the active moiety of latanoprost 0.005% [Xalatan; Pfizer, New York, NY]) is reported to reduce IOP by primarily increasing uveoscleral outflow. In contrast, NO released from the NO-donating moiety of latanoprostene bunod may lower IOP by increasing the trabecular meshwork outflow. Nitric oxide is also a biological messenger for other physiological functions, including vasodilation and, on systemic administration, the reduction of blood pressure.
A recent report showed that latanoprostene bunod was well tolerated and efficacious in lowering IOP in patients with primary open-angle glaucoma or ocular hypertension. There was a dose-dependent reduction in diurnal IOP over 28 days at concentrations of 0.006% to 0.04%, reaching a maximum effect with the 0.024% and 0.04% doses. Statistically significantly greater IOP reductions were observed with latanoprostene bunod 0.024% and 0.04% compared with latanoprost 0.005%. To date, IOP-lowering efficacy of latanoprostene bunod in subjects with ocular hypertension or primary open-angle glaucoma has not been reported outside the office hours. Effects of latanoprostene bunod on systemic blood pressure and calculated ocular perfusion pressure (based on mean arterial blood pressure and IOP) are also unknown outside the office hours. The present study evaluated the 24-hour effects of latanoprostene bunod 0.024%, which is considered the optimal IOP-lowering concentration during office hours, on IOP and ocular perfusion pressure after 4 weeks of once-daily treatment in patients with ocular hypertension or primary open-angle glaucoma. These effects were compared with the treatment effects of timolol maleate 0.5% ophthalmic solution given twice daily for 4 weeks in the same patients.
Methods
This randomized crossover clinical trial ( http://www.clinicaltrials.gov ; NCT01707381 ) adhered to the Declaration of Helsinki for research involving human subjects and was registered as a clinical trial. Study protocol was approved prospectively by the University of California, San Diego’s Institutional Review Board of Human Research Protections Program and in accordance with the Health Insurance Portability and Accountability Act. The study was conducted from November 2012 through December 2013. Study subjects were recruited consecutively from patients with bilateral early-stage open-angle glaucoma or ocular hypertension seen at the University’s Hamilton Glaucoma Center with predefined inclusion and exclusion criteria. Criteria for the diagnosis of early glaucomatous changes and ocular hypertension have been described previously. Patients with open-angle glaucoma had abnormalities in optic discs or visual fields (or both). Patients with ocular hypertension had normal optic discs and visual fields. Eligible patients for this study were 40–90 years old with untreated IOP of at least 22 mm Hg in 1 eye and not higher than 36 mm Hg in both eyes, determined using Goldmann tonometer (Haag-Streit, Mason, Ohio, USA) during office hours. Subjects were fully informed about this study, and informed consents were obtained.
Each subject’s medical history was reviewed. Candidates who had previous glaucoma surgery in either eye, had a history of ocular trauma, or had a sleep disorder or irregular sleep schedule were excluded. Each subject had an examination including slit-lamp biomicroscopy, gonioscopy, Goldmann applanation tonometry, and dilated funduscopy. Individuals with ocular inflammation, narrow iridocorneal angle, severe cardiovascular or diabetic condition, or use of a medication that may interact with the safety or efficacy of an NO-donating compound were excluded. Other routine systemic medications used by the subjects were documented, and enrolled subjects with a change of systemic medication during the study period were excluded.
Either the enrolled subjects were untreated with glaucoma medication or their glaucoma medications were washed out for 4 weeks followed by a check of IOP eligibility. A washout period of 4 weeks had been used for the previous study of latanoprostene bunod and its dose–IOP response relationship. Before the 24-hour laboratory recording, subjects were instructed to maintain a daily 8-hour regular sleep schedule for 1 week. Individual sleep schedule was verified using a wrist monitor for light exposure and arm movements (Actiwatch; Mini Mitter, Sunriver, Oregon, USA) and a wake/sleep log. Subjects were asked to abstain from alcohol for 3 days and regular coffee for 1 day before reporting to the laboratory approximately at 2 PM. Laboratory conditions and general experimental procedures have been described previously. The 8-hour sleep time for each subject in the laboratory was adjusted close to the recorded bedtime in previous week and this 8-hour period was referred to as the nocturnal/sleep period. Clock times for the IOP measurements were also individualized accordingly. Subjects were encouraged to continue normal indoor activities in the laboratory. Food and water were available, and meal times were not regulated.
Measurements of IOP and blood pressure/heart rate were taken by experienced researchers. Intraocular pressure was measured using a calibrated pneumatonometer (Reichert, Depew, New York, USA). Topical proparacaine 0.5% was used as the local anesthetic. Every plot of IOP measurement by the pneumatonometer was evaluated according to commonly accepted standards. Blood pressure and heart rate were measured immediately before the IOP measurements using an automated arm monitor (Accutorr Plus; Datascope, Montvale, New Jersey, USA). The cuff was mounted over the brachial artery, level with the heart, and removed after the measurement. Two readings were taken and, if the systolic or diastolic pressure differed by more than 5 mm Hg between the 2 readings, the third reading was added. The average of 2–3 readings was recorded for the systolic pressure, diastolic pressure, and heart rate at each clock time point.
Before bedtime, measurements were taken every 2 hours (eg, at 2 PM, 4 PM, 6 PM, and 8 PM). Subjects were instructed to sit for 5 minutes before the measurements of blood pressure/heart rate and IOP, and then lie in bed for 5 minutes before the supine measurements of blood pressure/heart rate and IOP. Lights in individual sleep rooms were turned off according to each individual’s sleep times (between 9:30 PM and 11:30 PM). Supine measurements only were taken first at 30 minutes after the lights were turned off (eg, 10 PM) and every 2 hours later. Subjects were awakened, if needed, and the measurements were taken immediately. A dim room light was used to assist with the measurements. Some sleep disturbance due to the measurement procedure was unavoidable. The disturbance may affect nocturnal blood pressure and IOP levels, even though the influence on IOP was probably minor. However, we assumed that every 24-hour laboratory session would be similarly affected for each individual. Room lighting was restored after the 8-hour nocturnal/sleep period, and subjects were awakened. Actual sleep length for each individual was not determined. Measurements of blood pressure/heart rate and IOP were taken again first at 30 minutes after the lights were turned on (eg, at 6 AM) and every 2 hours thereafter. Timings of the measurements were documented using infrared camera recording.
After baseline 24-hour sleep laboratory data were collected, subjects were randomly assigned to receive latanoprostene bunod 0.024% ophthalmic solution in both eyes once daily at approximately 8 PM or timolol maleate 0.5% ophthalmic solution twice daily at approximately 8 AM and 8 PM. Test agents were provided by Bausch & Lomb (Bridgewater, New Jersey, USA). The first treatment period lasted for 4 weeks before the second 24-hour sleep laboratory recording. Subjects were then crossed over to the alternate therapy for 4 weeks before the third 24-hour sleep laboratory recording. During the second and third laboratory recordings, subjects self-administered the test agent under supervision.
Per the study protocol, IOP data from 1 eligible eye of each subject were used for data analyses. If both eyes were eligible, the eye with a higher baseline IOP (Goldmann tonometer) prior to the first 24-hour laboratory recording was chosen for analysis. The right eye was chosen if both eyes had the same IOP value. Mean arterial blood pressure was calculated as the diastolic pressure plus one third of the difference between the systolic and the diastolic pressures. Ocular perfusion pressure in the sitting and supine body positions were calculated, using formulas based on the mean arterial blood pressure and IOP, adjusted for the height of the eye over the heart :
Sitting ocular perfusion pressure = 95/140 × mean arterial blood pressure − IOP
Supine ocular perfusion pressure = 115/130 × mean arterial blood pressure − IOP
Means of IOP, mean arterial blood pressure, ocular perfusion pressure, and heart rate were calculated for the 16-hour diurnal/wake period (8 readings) and the 8-hour nocturnal/sleep period (4 readings). For data presentation, laboratory data were aligned as if each subject had a nocturnal/sleep period from 9:30 PM to 5:30 AM. Statistical comparisons of study parameters were performed among the baseline, the latanoprostene bunod treatment, and the timolol treatment using the 1-way repeated-measures analysis of variance. The criterion for statistical significance was P < .05. Post hoc Bonferroni t test was used for all 3 possible paired comparisons. Normal distribution of test data was confirmed using the Kolmogorov-Smirnov test. Comparison of IOP at a single clock time point was not performed because its value in assessing IOP-lowering therapy is limited.
Subjects were instructed to report significant or unusual ocular and systemic reactions to the test agents during the study period. An eye examination was performed after each 24-hour laboratory recording and when adverse events of test agent had occurred. The severity of an adverse event was evaluated by a glaucoma specialist and could lead to patient withdrawal from the study.
Results
Twenty-five patients, 18 female and 7 male, with an age of 60.3 ± 10.6 years (mean ± standard deviation; range, 43–82 years) were enrolled. There were 20 white (4 Hispanic ethnicity), 4 black, and 1 American Indian patients. Twenty-one patients had a diagnosis of ocular hypertension and 4 patients had primary open-angle glaucoma. Nineteen patients were not using any glaucoma medication at enrollment. Six patients were being treated with a glaucoma medication(s) at enrollment: 4 with latanoprost, 1 with bimatoprost, and 1 with latanoprost and timolol. Treated patients underwent 4-week washout. The mean of the last office-hour Goldmann IOP reading before the first laboratory recording was 23.2 ± 1.6 mm Hg (range, 21–28 mm Hg) for all the enrolled subjects under no glaucoma medication. The mean central corneal thickness was 556.3 ± 27.9 μm (range, 511–598 μm).
The first (baseline) 24-hour laboratory recordings were obtained from all 25 enrolled subjects, but only 21 subjects completed the entire study protocol that involved 2 additional 24-hour laboratory recordings ( Supplemental Figure ; Supplemental Material available at AJO.com ). One subject who was under the treatment with latanoprostene bunod missed the scheduled second 24-hour laboratory recording and was withdrawn administratively. Three subjects withdrew from the study before the second laboratory recordings; 2 subjects had significant bradycardia under the timolol treatment and 1 subject had nausea and hyperhidrosis under the latanoprostene bunod treatment. The recorded adverse events from all enrolled subjects during the study period are summarized in Table 1 .
| Latanoprostene Bunod 0.024% (N = 46 Eyes or 23 Subjects) | Timolol 0.5% (N = 46 Eyes or 23 Subjects) | |
|---|---|---|
| Ocular | ||
| Punctate keratitis | 1 | 3 |
| Instillation site erythema | 1 | 0 |
| Instillation site irritation | 0 | 1 |
| Nonocular | ||
| Dizziness | 0 | 1 |
| Dyspnea | 0 | 1 |
| Nausea | 1 | 1 |
| Hyperhidrosis | 1 | 0 |
| Blood pressure decreased | 0 | 1 |
| Heart rate decreased | 0 | 2 |
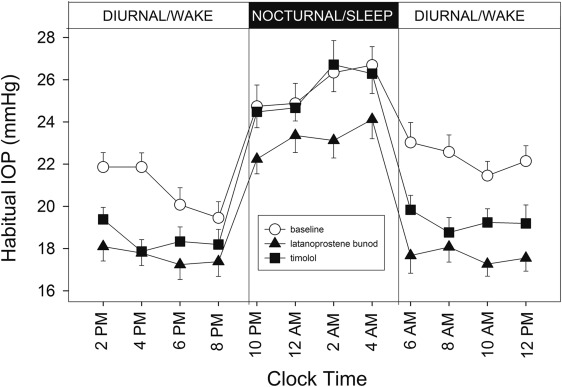
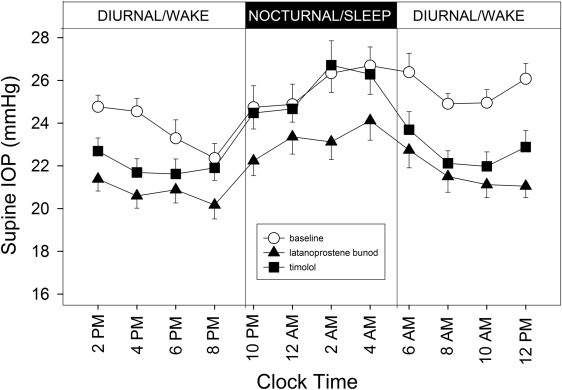
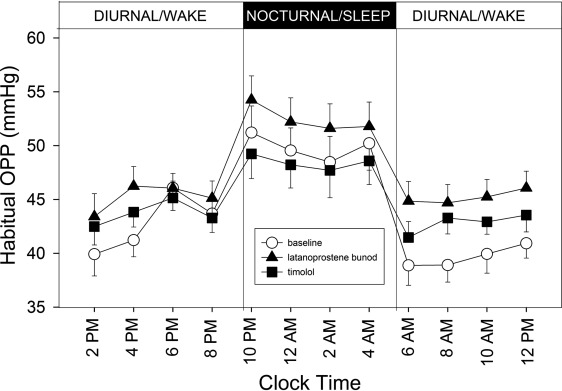
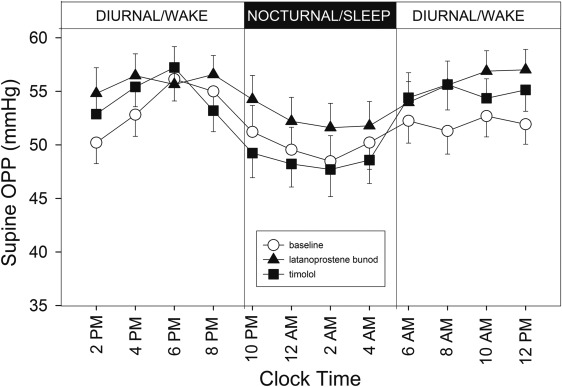
Figure 1 shows the 24-hour profiles of habitual IOP, sitting during the diurnal/wake period and supine during the nocturnal/sleep period, for the 21 subjects who completed the study. Coincidently with postural changes, IOP increased during the transition from the diurnal to the nocturnal period and IOP decreased during the transition from the nocturnal to the diurnal period. Mean IOP under the latanoprostene bunod treatment was consistently observed to be the lowest among the 3 experimental conditions during the 16-hour diurnal period. During the 8-hour nocturnal period, mean baseline IOP and mean IOP under the timolol treatment were similar. In contrast, mean IOP under the latanoprostene bunod treatment was notably lower than the baseline IOP as well as IOP under the timolol treatment. Figure 2 presents the 24-hour supine IOP profiles for the baseline, the latanoprostene bunod treatment, and the timolol treatment. For supine IOP during the diurnal period, IOP under the latanoprostene bunod treatment was also the lowest among the 3 experimental conditions. Profiles of 24-hour ocular perfusion pressure for the habitual body positions and for the supine position are presented as Figures 3 and 4 , respectively. For most time points, ocular perfusion pressures after the latanoprostene bunod treatment were the highest.