Purpose
To determine whether early visual acuity response to ranibizumab in diabetic macular edema is associated with long-term outcome.
Design
Post hoc analysis of randomized controlled trial data.
Methods
Pooled data from the ranibizumab plus prompt and deferred laser treatment arms of the Diabetic Retinopathy Clinical Research Network’s Protocol I study were used to explore the relationship between early (week 12) and late (weeks 52–156) visual acuity response (mean change from baseline in best-corrected visual acuity [CFB BCVA]; categorized improvement [<5, 5–9, or ≥10 Early Treatment Diabetic Retinopathy Study (ETDRS) letters] in BCVA).
Results
In the analysis population (340 eyes), <5-, 5- to 9-, and ≥10-letter BCVA improvements occurred in 39.7%, 23.2%, and 37.1% of eyes, respectively, at 12 weeks, and 34.2%, 16.5%, and 49.3% of eyes at 156 weeks. Within each early BCVA response category (<5, 5–9, and ≥10 letters of improvement at 12 weeks), mean CFB BCVA at 52–156 weeks varied by <5 letters from that at 12 weeks. CFB BCVA and <5-letter improvement at 12 weeks showed significant positive and negative association, respectively, with CFB BCVA and ≥10-letter improvement at 52 and 156 weeks. Similar relationships were demonstrated in eyes with baseline BCVA <69 letters, and associations remained significant after multivariate adjustment for potential confounders.
Conclusions
Ranibizumab ± laser therapy resulted in similar rates (∼40%) of suboptimal (<5-letter) and pronounced (≥10-letter) BCVA improvement at 12 weeks. Eyes with suboptimal early BCVA response showed poorer long-term visual outcomes than eyes with pronounced early response (mean improvement 3.0 vs 13.8 letters at 156 weeks).
Multiple randomized clinical trials indicate that intravitreal anti–vascular endothelial growth factor (VEGF) therapy, used alone or in conjunction with focal/grid laser photocoagulation, is more effective than laser photocoagulation alone in improving visual acuity in diabetic macular edema (DME), with ∼30%–70% of patients achieving ≥10-letter improvement and ∼10%–40% of patients achieving ≥15-letter improvement in best-corrected visual acuity (BCVA) after 1 year of treatment. However, even with the intensive treatment schedules (monthly or near-monthly intravitreal injections for the first 12 months) and close patient monitoring typically employed in controlled clinical trials, more than 35% of patients with DME fail to achieve ≥10-letter improvement in BCVA and more than 55% fail to achieve ≥15-letter improvement after 2 years of first-line anti-VEGF therapy. Although anti-VEGF therapy is generally considered suitable first-line therapy for center-involved DME, clearly not all DME patients respond satisfactorily to anti-VEGF agents.
Early identification of those patients who are likely to prove unresponsive or only partly responsive to long-term anti-VEGF therapy would enable more timely consideration of potential changes to their treatment regimens that might prove more effective in improving visual function and/or preventing vision loss. To this end, the EARLY (Early Anti-VEGF Response and Long-term efficacY) program, a series of post hoc analyses of data from the Diabetic Retinopathy Clinical Research Network’s ( DRCR.net ) Protocol I study of ranibizumab plus laser in DME, was initiated to explore the relationship between early and long-term anatomic and visual acuity responses to anti-VEGF therapy. The present analysis assesses the strength of the association between visual acuity outcome after 12 weeks of anti-VEGF therapy (ie, after 3 monthly intravitreal injections) and visual acuity outcomes at 1 and 3 years.
Methods
Study Overview
In Protocol I ( clinicaltrials.gov identifier NCT00445003 ), a large, prospective, multicenter trial in DME patients with baseline BCVA of 78-24 Early Treatment Diabetic Retinopathy Study (ETDRS) letters (approximate Snellen equivalent 20/32 to 20/320) and optical coherence tomography (OCT)-determined central subfield retinal thickness (CRT) ≥250 μm, study eyes were randomized to 1 of 4 treatment arms: (1) sham injection plus prompt (within 7–10 days) focal/grid photocoagulation, (2) intravitreal ranibizumab 0.5 mg plus prompt (within 7–10 days) focal/grid photocoagulation, (3) intravitreal ranibizumab 0.5 mg plus deferred (after ≥24 weeks) focal/grid photocoagulation, or (4) intravitreal triamcinolone 4 mg plus prompt (within 7–10 days) focal/grid photocoagulation. Intravitreal ranibizumab and sham injections were performed every 4 weeks for the first 12 weeks of the study and as needed thereafter; laser retreatment was determined by the extent of central macular edema. Eyes that met prespecified “failure” or “futility” criteria or were withdrawn from study treatment at the investigator’s discretion could be switched to an alternative treatment. Follow-up examinations, including measurements of BCVA and OCT-derived CRT, were performed every 4 weeks for the first year and every 4–16 weeks during the second and third years of the study. Follow-up was planned for 3 years, with the primary outcome being at 1 year. After review of study findings at 2 years, which demonstrated an efficacy advantage in the ranibizumab treatment arms, patients in the sham injection and intravitreal triamcinolone treatment arms were offered the option of switching to ranibizumab treatment for the third year. The study follow-up period was subsequently extended to 5 years.
Visual Acuity Response Analyses
The present analysis is based on 3-year study data from those patients in Protocol I who were randomized to treatment with ranibizumab plus either prompt or deferred laser, and who provided an observed BCVA reading at 12 weeks. To evaluate the strength of the association between early treatment response and long-term outcome, study eyes were separately categorized according to the change from baseline in BCVA (<5-letter, 5- to 9-letter, or ≥10-letter improvement) at 12 weeks. Visual acuity outcomes at subsequent follow-up visits were determined within study cohorts based on these initial BCVA response categories. Missing data owing to missed visits after week 12 were imputed using the last-observation-carried-forward method. BCVA readings obtained after introduction of alternative treatments to the randomly assigned study treatment were included in the analysis. In view of the possibility that the improvement in BCVA might be truncated by a ceiling effect among eyes initiating treatment with good visual acuity, a sensitivity analysis was performed in the subset of eyes with baseline BCVA <69 ETDRS letters (Snellen equivalent ∼20/50). Additional sensitivity analyses were performed in study eyes randomized to treatment with intravitreal ranibizumab plus deferred laser and in eyes categorized according to early BCVA response at other time points, ranging from 4 to 24 weeks after treatment initiation.
Statistical Methods
Intercohort comparisons of baseline characteristics were performed using the Student t test for continuous variables and Pearson χ 2 test for categorical variables. Intercohort comparisons of vision outcomes (change from baseline in BCVA) were performed using analysis of variance. Multiple linear and logistic regression analysis was performed to assess the relationship between early (12-week) and late (52- and 156-week) visual acuity outcomes after controlling for multiple potential confounding factors. Additional covariates of interest included age, sex, baseline BCVA and CRT, proportional CRT response (≥20% vs <20% reduction from baseline) at 12 weeks, cumulative number of ranibizumab injections and laser treatments received at 52 and 156 weeks, and prior receipt of DME treatment. P values were determined using Student t test (linear regression) and Wald’s χ 2 test (logistic regression). Statistical analyses were performed with SAS versions 9.3 and 9.4 (SAS Inc, Cary, North Carolina, USA). A P value of ≤.05 was considered statistically significant.
Results
In total, 375 study eyes were assigned to the ranibizumab plus prompt laser and ranibizumab plus deferred laser treatment arms in the Protocol I study. Of these, 340 study eyes provided observed visual acuity data and 335 study eyes provided observed OCT-derived CRT data at 12 (± 2) weeks (pooled analysis population). Within this overall population, sensitivity analyses were conducted on 212 eyes that had baseline BCVA <69 ETDRS letters.
Visual Acuity Outcomes
Overall best-corrected visual acuity response rate over time
Among the pooled analysis population (n = 340), 135 eyes (39.7%) showed <5-letter improvement in BCVA at 12 weeks, 79 eyes (23.2%) showed 5- to 9-letter improvement, and 126 eyes (37.1%) showed ≥10-letter improvement; within this latter category, 60 eyes (17.6% of total) achieved ≥15-letter improvement. Overall, at study end (156 weeks), 116 eyes (34.2%) showed <5-letter improvement, 56 eyes (16.5%) showed 5- to 9-letter improvement, and 167 eyes (49.3%) showed ≥10-letter improvement from baseline; of this latter group, 98 eyes (28.9% of total) achieved ≥15-letter improvement.
Relationship between best-corrected visual acuity responses at 12 weeks and 1 and 3 years
Within each of the initial BCVA response categories (ie, BCVA improvement <5 letters, 5–9 letters, and ≥10 letters at 12 weeks), the mean BCVA response (BCVA change from baseline) of study eyes at 52 weeks onward did not vary by more than 5 ETDRS letters from the observed mean BCVA response at 12 weeks ( Figure 1 ). Across the 3 response categories, intercohort differences in mean BCVA response (BCVA change from baseline) were statistically significant ( P < .001) at each 4-week time point. Marked intersubject variation in BCVA response was evident within each initial response category, and a limited initial BCVA response (<5-letter improvement [deterioration, no change, or 1- to 4-letter improvement] at 12 weeks) did not entirely preclude later development of a pronounced BCVA response during long-term treatment. However, development of the response was slow.
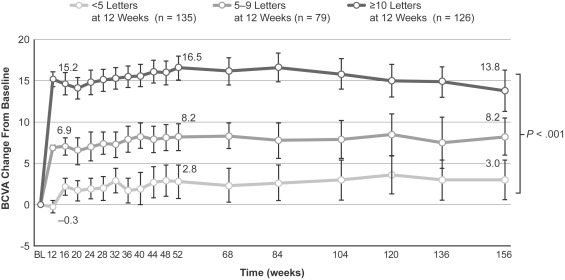
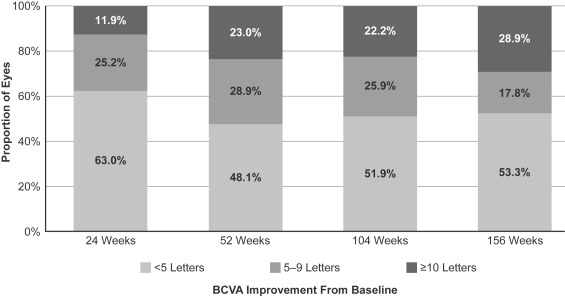
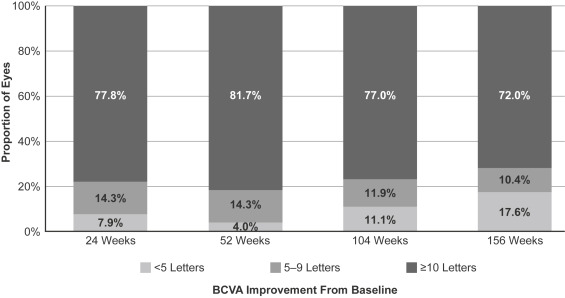
Within the subset of eyes with <5-letter BCVA improvement at 12 weeks, a minority of eyes achieved a BCVA gain of ≥10 ETDRS letters over the course of the study: 23.0% at 52 weeks, rising marginally to 28.9% at 156 weeks ( Figure 2 ). A smaller minority of eyes achieved a BCVA gain of ≥15 ETDRS letters: 6.7% at 52 weeks, increasing to 14.8% at 156 weeks. Approximately one-half of eyes continued to show <5-letter BCVA improvement at these time points. Within the subset of eyes with a pronounced initial BCVA response (≥10-letter improvement at 12 weeks), most eyes maintained this response over the course of the study: 81.7% at 52 weeks and 72.0% at 156 weeks, and approximately one-half of eyes showed ≥15-letter improvement at these time points (61.1% and 47.2% of eyes, respectively) ( Figure 3 ). A small proportion of eyes with ≥10-letter BCVA improvement at 12 weeks experienced subsequent attenuation of response to <5-letter improvement at 52 and 156 weeks.
Intercohort comparisons revealed significant differences in the baseline characteristics of study eyes: eyes with <5-letter improvement in BCVA at 12 weeks were older and had a higher baseline BCVA and a lower baseline CRT than eyes with ≥10-letter improvement in BCVA at 12 weeks ( Table 1 ). In multiple logistic regression analyses there was a negative, statistically significant association between limited BCVA response (<5-letter improvement in BCVA from baseline) at 12 weeks and pronounced BCVA response (≥10-letter improvement in BCVA from baseline) at 52 weeks (odds ratio [OR] 0.20, 95% confidence interval [CI] 0.12–0.36; P < .001) and 156 weeks (OR 0.31, 95% CI 0.19–0.53; P < .001) after adjusting for multiple patient characteristics, including age, sex, prior DME treatment, baseline BCVA and CRT, proportional CRT change (≥20% reduction) at 12 weeks, and cumulative number of ranibizumab injections and laser treatments administered. Of these, age and baseline BCVA were also significantly associated with a pronounced BCVA response (≥10-letter improvement from baseline) at 52 weeks, and baseline BCVA remained significantly associated with BCVA response at study end ( Table 2 ). Additionally, in multiple linear regression analyses BCVA response (BCVA change from baseline) at 12 weeks showed a significant association with BCVA response (BCVA change from baseline) at 52 weeks (coefficient estimate 0.72, standard error [SE] 0.07; P < .001) and 156 weeks (coefficient estimate 0.56, SE 0.10; P < .001) after adjusting for the standard covariates listed above. Age and baseline BCVA also showed significant associations with BCVA response (change from baseline) at 52 weeks, although only baseline BCVA remained significantly associated with BCVA response at study end ( Table 3 ).
| Parameter | BCVA Change From Baseline at 12 Weeks | P Value a | ||
|---|---|---|---|---|
| <5-Letter Improvement N = 135 | 5- to 9-Letter Improvement N = 79 | ≥10-Letter Improvement N = 126 | ||
| Age, mean ± SD (y) | 64.3 ± 9.3 | 61.9 ± 9.9 | 61.7 ± 10.1 | .036 |
| Male sex, n (%) | 78 (57.8) | 38 (48.1) | 75 (59.5) | .775 |
| Baseline BCVA, mean ± SD (letters) | 65.2 ± 12.2 | 64.9 ± 9.8 | 58.8 ± 11.4 | <.001 |
| Baseline CRT, mean ± SD (μm) | 379 ± 112 | 384 ± 107 | 438 ± 136 | <.001 |
| Prior DME treatment, n (%) | 86 (63.7) | 49 (62.0) | 74 (58.7) | .410 |
a Comparison between <5-letter improvement and ≥10-letter improvement categories, based on Student t test for continuous variables and Pearson χ 2 test or Fisher exact test for categorical variables.
| Parameter | Multiple Logistic Regression of ≥10-Letter Improvement in BCVA at 52 and 156 Weeks | |||
|---|---|---|---|---|
| 52 Weeks N = 335 | 156 Weeks N = 335 | |||
| Odds Ratio | P Value a | Odds Ratio | P Value a | |
| Age | 0.96 | .007 | 0.99 | .407 |
| Sex (F/M) | 1.08 | .768 | 0.89 | .659 |
| Baseline BCVA | 0.93 | <.001 | 0.94 | <.001 |
| Baseline CRT | 1.00 | .202 | 1.00 | .779 |
| BCVA CFB <5 letters at week 12 | 0.20 | <.001 | 0.31 | <.001 |
| ≥20% CRT improvement at week 12 | 1.80 | .074 | 1.50 | .174 |
| Cumulative no. RAN injections at week 52 or 156 | 0.94 | .256 | 1.00 | .919 |
| Cumulative no. laser treatments at week 52 or 156 | 0.85 | .167 | 0.92 | .269 |
| Prior DME treatment | 0.64 | .113 | 0.85 | .539 |
| Parameter | Multiple Linear Regression of BCVA Response at 52 and 156 Weeks | |||
|---|---|---|---|---|
| 52 Weeks N = 335 | 156 Weeks N = 335 | |||
| Estimate | P Value a | Estimate | P Value a | |
| Age | −0.13 | .016 | −0.10 | .162 |
| Sex | 0.01 | .991 | 0.66 | .644 |
| Baseline BCVA | −0.22 | <.001 | −0.25 | <.001 |
| Baseline CRT | −0.01 | .061 | −0.00 | .946 |
| BCVA CFB at week 12 | 0.72 | <.001 | 0.56 | <.001 |
| ≥20% CRT improvement at week 12 | 1.59 | .205 | 1.88 | .268 |
| Cumulative no. RAN injections at week 52 or 156 | −0.09 | .658 | 0.04 | .721 |
| Cumulative no. laser treatments at week 52 or 156 | −0.66 | .116 | −0.16 | .684 |
| Prior DME treatment | −2.02 | .054 | −0.16 | .914 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


