Purpose
To evaluate the differential sensitivity of choroidal endothelial, retinal pigment epithelial, and retinal ganglion cells to escalating doses of proton beam radiation and to establish a safe dose range for the management of choroidal neovascularization associated with age-related macular degeneration (AMD).
Design
Laboratory investigation.
Methods
Proliferating simian choroidal endothelial cells (RF/6A), differentiated rat retinal ganglion cells (RGC-5), and serum-starved human retinal pigment epithelial cells (ARPE-19) were exposed to 2, 4, 8, and 12 cobalt gray equivalent of proton beam radiation and cell viability was quantified on day 9. Reactive oxygen species levels were analyzed.
Results
Significant decline of choroidal endothelial cell viability was noted as dose escalated from 4 to 8 cobalt gray equivalent with maximum effect observed at 12 cobalt gray equivalent. RGC-5 and ARPE-19 cell count decreased to 95% and 62.7% at 8 cobalt gray equivalent, respectively. Sub-analysis between 4 and 8 cobalt gray equivalent radiation revealed significant decrease in choroidal endothelial cell viability (43.1% at 7 cobalt gray equivalent and 32.3% at 8 cobalt gray equivalent of radiation). Correspondingly, RGC-5 and ARPE-19 cells did not show decrease in cell count or viability. Reactive oxygen species levels significantly increased in radiation-treated choroidal endothelial cells (8.3%-11.9%).
Conclusions
At 6-8 cobalt gray equivalent proton beam radiation, retinal ganglion and retinal pigment epithelial cells are preserved while choroidal endothelial cells are completely inhibited. This dosage offers optimum therapeutic safety window for treatment using proton beam radiation for exudative AMD.
Age-related macular degeneration (AMD) is the leading cause of late-onset vision loss in people over the age of 65 years in developed countries. The neovascular form of AMD affects 10%-20% of cases and accounts for approximately 80% of cases of severe vision loss. Pathophysiologically, the neovascular form is characterized by choroidal neovascularization (CNV) with choroidal endothelial cell proliferation, a potential target for therapeutic modalities.
Management of CNV using conventional or modified photodynamic therapy has poor differential tissue sensitivity, leading to the destruction of both the stable choroidal endothelial cells and the overlying retinal cells. The loss of stable choroidal endothelial cells leads to ischemia and recurrent proliferation, whereas the degeneration of retinal pigment epithelial (RPE) cells leads to the formation of absolute scotoma in the visual field.
Proliferating endothelial cells present in CNV offer a potential target for radiation-based treatment. Proton radiation, a form of particle radiation therapy, delivers a homogenous 3-dimensional dose to the target lesion with minimal damage to surrounding healthy tissues. Unlike conventional radiation, ideally selected proton radiation dosage should destroy choroidal endothelial cells without damage to RPE cells or retinal ganglion cells. Recently, epimacular brachytherapy has been studied as a novel method to deliver radiation therapy directly to CNV.
In this study, we tried to mimic CNV associated with AMD in an in vitro model, using choroidal endothelial cells (simian), RPE cells (human), and retinal ganglion cells (rat). We evaluated the differential sensitivity of choroidal endothelial cells, RPE cells, and retinal ganglion cells to escalating doses of proton beam radiation to establish a safe dose range that can potentially be applicable in the management of CNV associated with AMD.
Subjects and Methods
Cell Lines
This is an experimental study involving 3 in vitro cell lines—namely, proliferating simian choroidal endothelial cells (RF/6A), differentiated rat retinal ganglion cells (RGC-5), and serum-starved human RPE cells (ARPE-19). These 3 cell lines were cultured in minimal essential medium (Thermo Fisher Scientific, Waltham, Massachusetts, USA), Dulbecco’s modified Eagle’s medium (Life Technologies, Carlsbad, California, USA), and Dulbecco’s modified Eagle’s medium/F12 (Life Technologies), respectively. The media were supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin. Cells were grown at 37°C in logarithmic scale in 75-cm 2 cell culture flasks.
Preparation of the Cells
Cells were grown to confluent densities by seeding them at 1 × 10 5 cells/well, either in 6-well plates or in 25-cm 2 flasks prior to the experiment. Five plates were maintained for 5 different dosage levels. Cell passages 7-15 were used for the study. To mimic in vivo retina with nonproliferative retinal ganglion cells in an in vitro condition, semi-confluent RGC-5 cells were differentiated by applying 100 nm staurosporine as described earlier. Proliferation of ARPE-19 cells was arrested by maintaining them in serum-free media prior to the experiment. However, choroidal endothelial cells were maintained in log-phase of growth.
Experimental Set-up of Modulated Proton Beam and Irradiation of Cells
Schematic representation of the set-up for the delivery of the proton beam to the cell culture is as shown in Figure 1 . The Particle Accelerator Cyclotron (Ion Beam Application Limited, Louvain-la-Neuve, Belgium) at the University of Florida was used to produce 70 MeV protons. Monolayers of confluent RF/6A cells were exposed to the proton beam in 25-mm 2 flasks filled with the media. The flasks were placed such that the growth surface was perpendicular to the incident proton beam. A large field size (25 cm in diameter) of the proton beam was used to ensure uniform distribution of the beam. The error rate of distribution of proton beams was within 2% for the 8-cm length and 4-cm width of the cell plate.
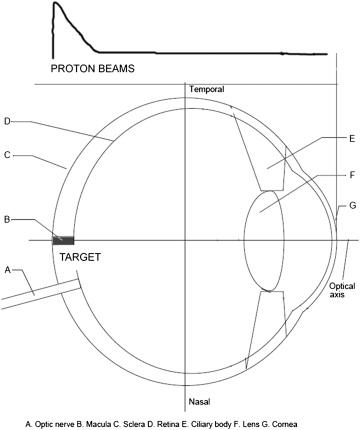
The Cyclotron unit used in the clinical application is a fixed energy system with a set range of 15.1 cm and a modulation width of 10.4 cm. The radiation doses were uniform between depths of 3.9-15.1 cm in water. The cell flasks were positioned between 2 solid water blocks of 10 cm thickness to achieve the desired depth of penetration of the proton beam and to minimize energy loss through Coulomb interactions and scattering within the air. An air gap of 3.5 cm was built to allow cell flasks to be placed between the solid water blocks. The top face of the 2-cm height of the cell container was at the isocenter of the treatment room. The height of the liquid used in the cell container was approximately 2 mm from the bottom of the cell container. A modulator wheel was used to spread out the energy such that it covered the entire cell plate uniformly.
The delivered doses were calculated by the monitor unit program and verified by the measurements with a calibrated ionization chamber. The setting on the reading of the calibrated monitor unit chamber in the proton beam was used to control the actual delivered radiation doses.
As an initial part of our study, we exposed differentiated retinal ganglion cells, nonproliferating ARPE-19, and proliferating choroidal endothelial cells to a single fraction of escalating radiation doses of 2, 4, 8, and 12 cobalt gray equivalent. (A cobalt gray equivalent is the proton dose in grays multiplied by a relative biological effectiveness factor of 1.1). Our data showed a trend of declining choroidal endothelial cell viability after a radiation dosage of 4-8 cobalt gray equivalent. Based on this result, we further performed a sub-analysis of treating cells to increasing doses of proton beam radiation by 1 cobalt gray equivalent increments from 4-8 cobalt gray equivalent in a single fraction and evaluated the viability of treated cells, while unexposed cells served as controls. Following radiation, cells were maintained at 37°C in a humidified atmosphere. Postradiation dose-response studies on these cells were performed on day 9.
Quantification of Cell Growth
We used Vi-CELL XR (Beckman Coulter, Inc, Brea, California, USA), an automated cell viability analyzer, to measure the number of viable cells. This analyzer is based on trypan blue dye exclusion analysis that excludes the dead cells. After postradiation incubation, cells were trypsinized and quantified for each dose on day 9. Experiments were repeated 3 times for concordance.
Evaluation of Radiation-Induced Late Cytotoxicity
Effect of radiation-induced cytotoxicity was assayed using Neutral Red Assay (Sigma-Aldrich Corp, St. Louis, Missouri, USA) 9 days after radiation. In brief, cells were incubated with neutral red (final concentration 0.033%) for 2 hours at room temperature and were subsequently washed with Hank’s buffered saline solution. Further, cells were allowed to air dry for 20 minutes and retained dye was eluted using ice-cold solubilization buffer (1% acetic acid/50% ethanol). Twenty minutes later, 100-μL aliquots were transferred to 96-well plates (Greiner Bio-One North America, Inc, Monroe, North Carolina, USA) for optical density readings at 490 nm using a Multi-detection Microplate Reader (BioTek US, Winooski, Vermont, USA).
Measurement of Reactive Oxygen Species
Intracellular reactive oxygen species measurements were made using dihydrorhodamine 123 (AnaSpec EGT, Fremont, California, USA), which is a freely permeable nonfluorescent probe that localizes to mitochondria, where it fluoresces after oxidation by reactive oxygen species. Postradiated cells harvested at different day intervals were loaded with 10 μM dihydrorhodamine 123. On excitation at 485 nm, rhodamine fluoresces at 528 nm, which was quantified using a multidetection microplate reader.
Morphologic Changes in Cells After Radiation
To observe the morphologic changes, control and irradiated cells at various dosages were plated either in 6-well plate or in 25-cm 2 flasks for each dosage. On day 9, each flask was examined separately under a bright-field phase-contrast IX51 microscope (Olympus, Center Valley, Pennsylvania, USA). For each dose studied, 5 randomly chosen photographs were taken at 20× magnification and analyzed.
Statistical Analysis
Experiments were repeated for concordance and data were expressed as mean ± standard deviation. Mean data were normalized with control and presented as a percentage. The comparative significance levels between dosages were analyzed using ANOVA (InStat software; GraphPad Software Inc, La Jolla, California, USA). A P value ≤ .05 was considered statistically significant.
Results
Cell Counts at 2, 4, 8, and 12 Cobalt Gray Equivalent Radiation
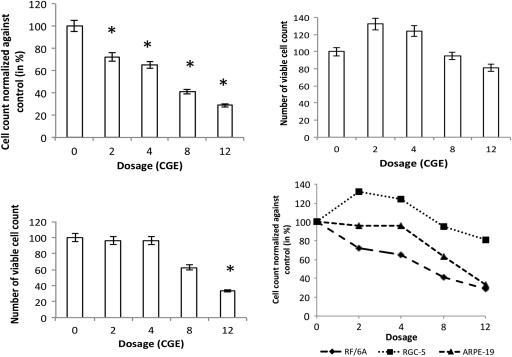
Cell counts were calculated as percentages and normalized against control. Figure 2 shows viable cell counts, in percent, on postradiation incubation day 9. Proliferating RF/6A cell cultures exposed to proton beam showed an increase in cell death as the dose escalated. By day 9, significant difference in cell count compared with control was seen with cells exposed to 2 and 4 cobalt gray equivalent. Cell counts were 72% ± 10.4% at 2 cobalt gray equivalent and 65% ± 12.4% at 4 cobalt gray equivalent doses ( Table ). At higher doses of 8 and 12 cobalt gray equivalent, cell counts showed a further decline to 40.9% ± 9.1% and 28.8% ± 10.9%, respectively ( P = .002).
| Dosages (CGE) | RF/6A (Mean ± SD) | RGC-5 (Mean ± SD) | ARPE-19 (Mean ± SD) |
|---|---|---|---|
| 0 | 135000 ± 15000 | 100000 ± 16000 | 510000 ± 12000 |
| 2 | 97200 ± 10108.8 | 132000 ± 8976 | 490000 ± 8000 |
| 4 | 87750 ± 10881 | 124000 ± 4464 | 490000 ± 5600 |
| 8 | 55215 ± 5024.5 | 95000 ± 5415 | 320000 ± 9500 |
| 12 | 38880 ± 4237.9 | 81000 ± 4293 | 170000 ± 4500 |
RGC-5 cells did not show decrease in cell count compared with control at doses of 2 and 4 cobalt gray equivalent. Cell count increased over a control cell count at 2 cobalt gray equivalent to 132% ± 6.8% and at 4 cobalt gray equivalent to 124% ± 3.6% ( Table ). RGC-5 cells exposed to 8 and 12 cobalt gray equivalent showed a decline in cell counts. They were 95% ± 5.7% at 8 cobalt gray equivalent and 81% ± 5.3% at 12 cobalt gray equivalent doses, respectively, compared with controls. The difference in cell count failed to reach statistical significance at 8 cobalt gray equivalent ( P = .26) but reached significance at 12 cobalt gray equivalent ( P = .00025).
ARPE-19 cells did not show any significant decrease of cell count at lower radiation doses of 2 and 4 cobalt gray equivalent. They were 96.0% ± 1.6% and 96% ± 1.2% at 2 and 4 cobalt gray equivalent dosage, respectively. The difference in cell counts compared with control did not achieve any statistical significance. At higher radiation dose (8 cobalt gray equivalent), ARPE-19 cell count declined to 62.7% ± 3.0%, which further reduced to 33.3% ± 2.7% at 12 cobalt gray equivalent dosage ( P = .0001). This decreased cell count was statistically significant when compared with control ( Figure 2 ).
Effect of Proton Radiation on Cell Viability Using Neutral Red Assay
The spectrometric absorbance were calculated as percentage of viability and normalized against control cells (that are unexposed to proton radiation). Proliferating RF/6A cells after exposure to radiation showed significant decrease in viability at higher doses. Neutral red viability assay results corroborated with dose-dependent cell count using Vi-CELL. On day 9, a significant change in cell viability was noted in cells exposed to 2 and 4 cobalt gray equivalent, compared with control cell viability (85% ± 10.1% and 73% ± 7.9%; P = .022). At 8 cobalt gray equivalent dose, radiation induced a significant reduction in cell viability to 30.1% ± 6.7%, which further declined to 24.2% ± 5.3% at 12 cobalt gray equivalent dose ( Figure 3 ; P < .001).
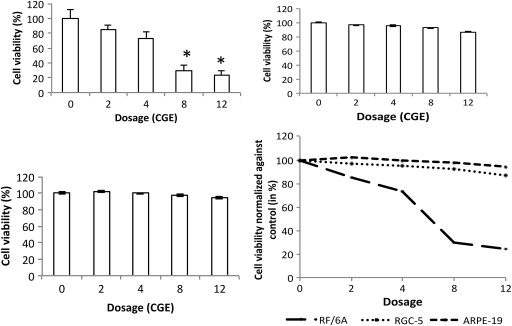
On day 9, compared with nonirradiated cells, differentiated RGC-5 cells exposed to 2 and 4 cobalt gray equivalent dosage did not show significant decrease in cell viability. The observed cell viability was 96.4% ± 7.1% and 95.1% ± 10.1% at 2 and 4 cobalt gray equivalent doses, respectively. At higher doses, the cell viability decreased progressively, but not at a significant rate. Eight and 12 cobalt gray equivalent doses decreased cell viability to 92.5% ± 6.5% and 86.8% ± 6.7%, respectively ( P = .065).
Similar to retinal ganglion cells, ARPE-19 cells did not show any significant variation in viability. On day 9, serum-starved irradiated ARPE-19 cells showed 102.2% ± 7.5% and 99.7% ± 6.5% cell viability at 2 and 4 cobalt gray equivalent, respectively, compared with control. At higher doses, cell viability was 97.3% ± 5.8% at 8 cobalt gray equivalent and 94.3% ± 6.2% at 12 cobalt gray equivalent doses, compared with control. However, the reduction in cell viability was not statistically significant ( P = .07).
Intracellular Reactive Oxygen Species Levels After Radiation
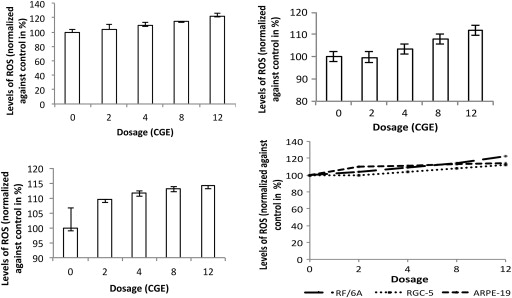
In RF/6A cells, the levels of reactive oxygen species increased in a dose-dependent fashion ( Figure 4 ), but the increase was not statistically significant at 2 and 4 cobalt gray equivalent (4.2% and 8.9%, respectively). At 8 and 12 cobalt gray equivalent, RF/6A cells showed, respectively, 14.4% and 22.2% of increased reactive oxygen species levels compared with controls at day 9. The increased reactive oxygen species levels were significant in RF/6A cells as compared to control ( P = .01).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


