Purpose
To evaluate the diagnostic characteristics of type 2 (classic) choroidal neovascularizations secondary to age-related macular degeneration using spectral domain-optical coherence tomography (SD OCT), indocyanine green angiography (ICGA), and fluorescein angiography (FA).
Design
Observational case series.
Methods
setting: Institutional. study population: Thirteen treatment-naïve eyes with type 2 choroidal neovascularization without an occult component. main outcome measures: Greatest horizontal dimension, based on the anatomic features of the neovascular complex by SD OCT (Spectralis; Heidelberg Engineering), ICGA, and FA; retinal leakage area in late-phase FA and ICGA; and the area of retinal edema in SD OCT. observation procedures: For direct comparison, ICGA and FA images were overlaid manually on infrared plus SD OCT images using VirtualDub and Paint.NET software. Greatest horizontal dimension was measured using Image J software (National Institutes of Health).
Results
The mean greatest horizontal dimension of the neovascular complex and the retinal leakage area consistently were smaller on ICGA compared with the area of retinal edema on SD OCT. According to FA, the greatest horizontal dimension of early, well-demarcated hyperfluorescence was significantly smaller than the neovascular complex on SD OCT. In addition, the greatest horizontal dimension of the retinal leakage area in late-phase FA consistently was smaller than the area of retinal edema on SD OCT.
Conclusions
In classic choroidal neovascularization, ICGA and FA seem to underestimate the extension of the neovascular complex and the associated retinal pathologic features compared with SD OCT imaging.
The combination of currently available diagnostic methods, such as infrared reflectance, fundus angiography (FA), indocyanine green angiography (ICGA), and, more recently, spectral-domain optical coherence tomography (SD OCT), may lead to a better understanding of the pathophysiologic features of macular disease. Neovascular age-related macular degeneration (AMD), in particular, remains the leading cause of severe vision loss in countries with high life expectancy. FA currently is the method of choice for diagnosing and classifying CNV as classic, occult, or combination subtypes. An additional classification characterizes well-defined membranes on FA as type 2 CNV and ill-defined membranes as type 1 CNV according to Gass, with type 2 lesions typically located in the subneuroretinal space and type 1 lesions located beneath the retinal pigment epithelium (RPE). The latter classification is used here. For cases in which an exact lesion characterization is not possible, ICGA can be used as an ancillary angiographic method to identify the entire extension of the CNV lesion because of its ability to image sub-RPE components of CNV. Optical coherence tomography, and in particular SD OCT, with its high-resolution raster scanning capabilities, has revolutionized the diagnostic approach to AMD. SD OCT is able to define the morphologic features of the fibrovascular complex, as well as its exudative consequences of retinal thickening and edema. Additional prognostic factors, based on specific imaging methods, may include the size of the neovascular complex, leakage activity, presence of intraretinal or subretinal fluid, and RPE alterations.
Exact CNV diagnoses and subtype classifications, together with knowledge of prognostic factors, serve as a basis for assessment of disease activity and therapeutic response. The possible importance of diagnostic classifications as prognostic factors has been investigated repeatedly, but the outcome of these investigations are inconclusive because of the paucity of long-term data, differences in trial design, and diversity of reporting styles.
The availability of new imaging techniques mandates detailed analyses and comparisons of images obtained with established methods, such as FA and ICGA, with those of novel techniques, such as OCT. We compared fluorescence patterns and retinal pathologic features according to FA and ICGA with the tomographic characteristics assessed by SD OCT in a homogenous population of eyes with previously untreated type 2 CNV. We selected eyes with type 2 neovascularization to allow a clear distinction between the primary neovascular lesion and secondary leakage, which is not possible with occult lesions.
Methods
Fifty-three consecutive patients who consulted the clinic at the Department of Ophthalmology, Medical University of Vienna, between July 2008 and August 2009 were screened for inclusion. Each patient underwent a complete clinical examination that included slit-lamp biomicroscopy, fundus photography, FA, ICGA, and SD OCT. Type 2 CNV was defined as an area of choroidal hyperfluorescence with well-demarcated boundaries clearly discernible in the early phase of FA, with progressive leakage beyond the initial boundaries of the CNV and an area of early hypercyanescence without marked leakage activity on ICGA. Patients with neovascular maculopathy from pathologic myopia, angioid streaks, infectious inflammatory chorioretinal disease, tumors, hereditary disorders, or trauma were excluded. Thirteen eyes were selected for inclusion.
Image Processing
SD OCT, FA, and ICGA images were obtained with the Spectralis HRA+OCT (Heidelberg Engineering, Heidelberg, Germany). A horizontal volume scan, consisting of 49 scans with 25 frames each, was obtained using the infrared+OCT Spectralis tool. Scan length was 6000 μm. FA and ICGA were assessed according to a standard scheme; imaging of 1 minute or less was defined as the early phase, and imaging from 10 to 11 minutes was defined as the late phase.
FA and ICGA images of each eye were superimposed manually on corresponding infrared images of the SD OCT. To ensure correct measurement, retinal vessel landmarks were matched manually; subsequently, raster scans were superimposed over the angiographs to create FA and OCT composites and ICGA and OCT composites. Using this method, early-phase and late-phase angiographic frames were correlated directly with SD OCT images. VirtualDub software version 1.8.6 (available at http://www.virtualdub.org ) was used to select corresponding OCT frames, and Paint.NET software version 3.36 (available at http://www.getpaint.net ) was used for the superimposition of images ( Figure 1 ).
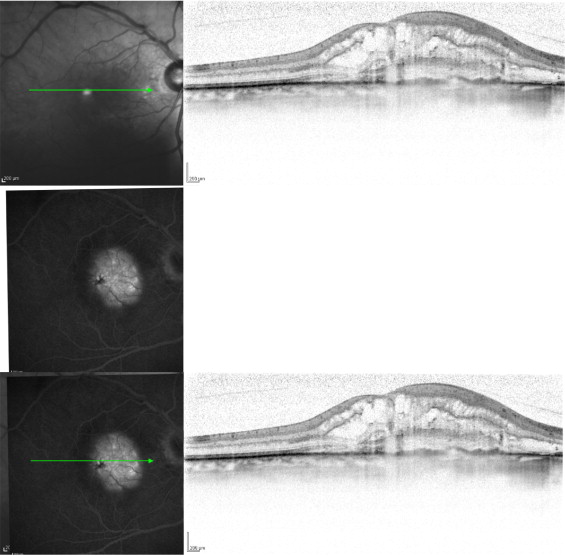
Image Analysis
Each FA image was reviewed by at least 2 experienced readers (C.K., G.D., S.S.). OCT and ICGA images were evaluated by a certified reader at the Vienna Reading Center (G.D.) of the Medical University of Vienna. To insure consistent labeling of angiographic and OCT images, extension of the well-demarcated areas of hyperfluorescence or hypercyanescence during early-phase FA and ICGA was defined as the neovascular complex, and the area of hyperfluorescence or hypercyanescence during late-phase FA and ICGA was defined as the retinal leakage area. After the FA and ICGA images were superimposed on the infrared SD OCT scans, those scans with the greatest horizontal dimension of choroidal hyperfluorescence consistent with the neovascular complex in FA were used for further measurements ( Figure 1 ). The same scan also indicated the greatest horizontal dimension of hypercyanscence in ICGA. In SD OCT, the neovascular complex was defined as the greatest horizontal dimension of the hyperreflective structure anterior to a disrupted RPE band and solid tissue in the subretinal space. The retinal leakage area was defined as an entire extension of exudative pathologic features (i.e., retinal thickening, subretinal fluid, or intraretinal fluid). Pigment epithelium detachment was not observed in any of the included eyes. Furthermore, the greatest horizontal dimension of disrupted RPE was measured and related to fluorescence and cyanescence patterns on the angiographs. Measurements were performed using Image J software (National Institutes of Health, Bethesda, Maryland, USA; available at http://rsbweb.nih.gov ) true to scale. The scale for the FA and the ICGA picture was 11 pixels for 200μm; for the OCT scale, it was 34 pixels for 200 μm based on the assumption that the scale as shown in the bottom right corner of each Heidelberg image is accurate.
Statistical Analysis
Statistical analysis was performed using SPSS statistical software version 15.0 for Windows (SPSS, Inc, Chicago, Illinois, USA) and included ranges, means, and ratios. Paired comparative statistics were determined using the paired t test. P values less than .05 were considered statistically significant.
Results
Neovascular features of type 2 CNV lesions were detectable by ICGA in all eyes. In 8 (61%) of the 13 eyes, the borders of the neovascular complex were well defined. In 5 (39%) eyes, the borders were not well defined. Of the 13 eyes, 7 (54%) were located subfoveally, 3 (23%) were located juxtafoveally, and 3 (23%) were located extrafoveally.
On ICGA, the mean greatest horizontal dimension of the neovascular complex was 1382 ± 654 μm. The greatest horizontal dimension of the neovascular complex by SD OCT was consistently larger, with a mean of 1962 ± 1192 μm ( P = .017; Pearson coefficient, 0.63). The mean ICGA-to-SD OCT ratio was 0.73 ± 0.27 (range, 0.34 to 1.11; Table 1 ). This suggests that ICGA underestimated the size of the neovascular complex in type 2 CNV compared with SD OCT.
| Case No. | FA (Early) GHD (μm) | Ratio of FA to OCT | ICGA (Early) GHD (μm) | Ratio of ICGA to OCT | SD OCT GHD (μm) |
|---|---|---|---|---|---|
| 1 | 1818 | 1.95 | 1030 | 1.11 | 932 |
| 2 | 2128 | 0.90 | 2206 | 0.94 | 2354 |
| 3 | 2206 | 0.79 | 1891 | 0.68 | 2784 |
| 4 | 3903 | 0.89 | 1770 | 0.41 | 4369 |
| 5 | 618 | 0.39 | 545 | 0.34 | 1600 |
| 6 | 1140 | 0.72 | 1745 | 1.11 | 1577 |
| 7 | 1891 | 0.81 | 2327 | 0.95 | 2454 |
| 8 | 1648 | 0.94 | 1455 | 0.83 | 1750 |
| 9 | 1188 | 0.52 | 970 | 0.43 | 2274 |
| 10 | 1479 | 0.76 | 1430 | 0.74 | 1937 |
| 11 | 309 | 1.01 | 163 | 0.53 | 306 |
| 12 | 1036 | 0.79 | 727 | 0.55 | 1318 |
| 13 | 1827 | 0.92 | 1708 | 0.86 | 1977 |
The mean greatest horizontal dimension of the retinal leakage area according to ICGA was 1420 ± 670 μm. The greatest horizontal dimension of the retinal leakage area according to SD OCT was consistently larger, with a mean of 3473 ± 885 μm ( P = .000; Pearson coefficient, 0.65). The mean ICGA-to-SD OCT ratio was 0.40 ± 0.16, and the retinal leakage area between early-phase ICGA and late-phase ICGA was not significant ( P = .15). Thus, it seems that hypercyanescence on ICGA and fluid on SD OCT were not correlated.
In contrast, RPE disruption on SD OCT had a mean greatest horizontal dimension of 1486 ± 693 μm and correlated well with the mean greatest horizontal dimension of the retinal leakage area on ICGA, with a ratio of 0.97 ± 0.07 ( P = .000; Pearson coefficient, 0.985).
On FA, the mean greatest horizontal dimension of the neovascular complex was 1629 ± 888 μm. The greatest horizontal dimension of the neovascular complex on SD OCT was consistently larger, with a mean of 1961 ± 975 μm ( P = .029; Pearson coefficient, 0.87). The mean FA-to-OCT ratio was 0.88 ± 0.37 (range, 0.39 to 1.95; Table 1 ). The mean greatest horizontal dimension of the retinal leakage area was 2276 ± 939 μm. The greatest horizontal dimension of the retinal leakage area by SD OCT was substantially larger, with a mean of 3472 ± 885 μm ( P = .000; Pearson coefficient, 0.70), and the mean FA-to-OCT ratio was 0.65 ± 0.19 (range, 0.32 to 0.94; Table 2 ). Again, the FA-to-SD OCT ratios for both the neovascular complex and the retinal leakage area were less than 1.0, suggesting that FA underestimated the area of pathologic features in type 2 CNV compared with SD OCT.



