Purpose
To describe the management of diabetic macular edema during pregnancy with the use of a dexamethasone slow-release intravitreal implant.
Design
Retrospective, observational, consecutive case series.
Methods
The study included 5 pregnant women who presented with diabetic macular edema during pregnancy in the period from 2011 to 2014. Review of charts and photographs comprised best-corrected visual acuity (BCVA), foveal center field thickness assessed by optical coherence tomography, blood pressure, glycated hemoglobin, medications, and changes in such parameters after implant injection.
Results
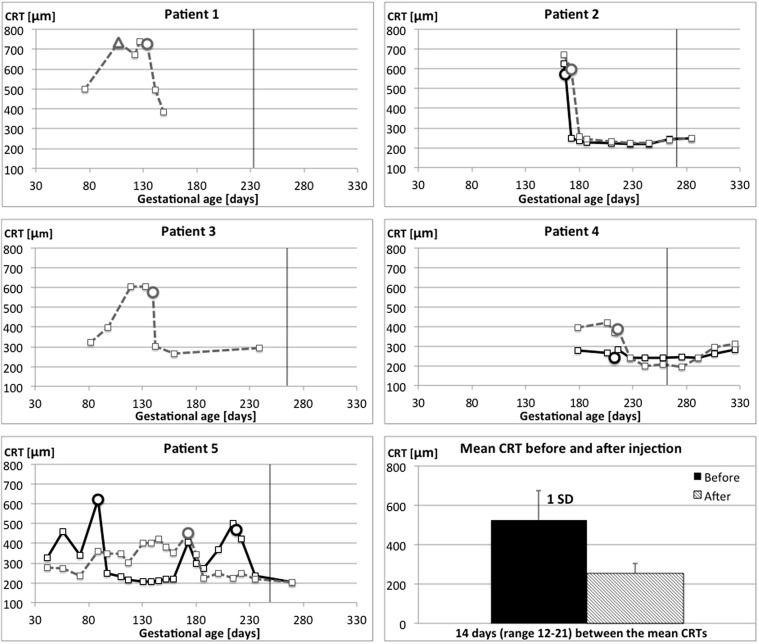
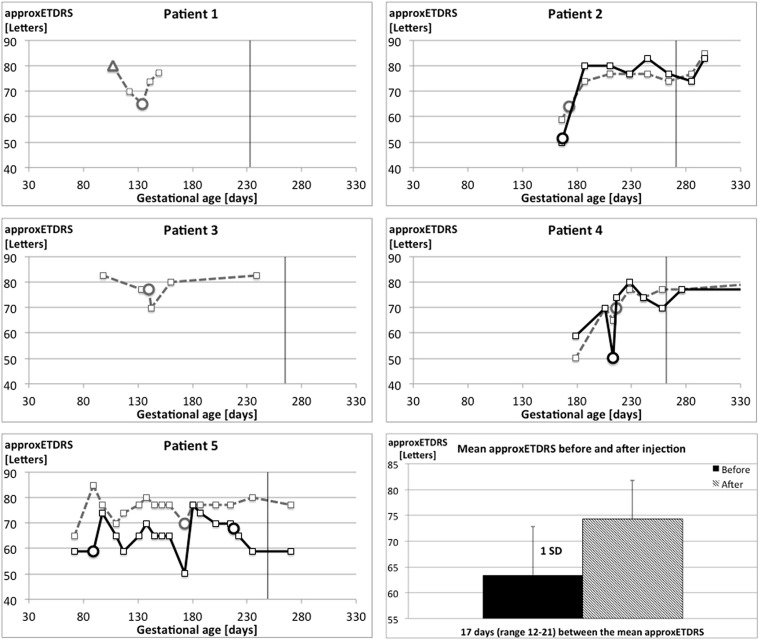
Diabetic macular edema involving the foveal center was observed between gestational weeks 9 and 23 in 10 eyes of 5 patients. Dexamethasone intravitreal implant injection was given 10 times in 9 eyes with a mean preinjection center field retinal thickness of 535 μm (range, 239–727 μm) and a mean preinjection best-corrected visual acuity (BCVA) of 63 approximated Early Treatment Diabetic Retinopathy Study (approxETDRS) letters (range, 50–77 letters). One eye was unavailable for follow-up. In 7 of 8 eyes injection was followed, within 3 weeks, by a greater than 145 μm reduction in foveal center field thickness and in 6 of 8 eyes by an increase in BCVA of 5 or more approxETDRS letters. A mild transient rise in intraocular pressure occurred in 3 out of 8 eyes.
Conclusion
Diabetic macular edema involving the foveal center that presented during pregnancy responded promptly to intravitreal dexamethasone therapy by foveal thickness reduction and visual acuity improvement without clinically significant intraocular pressure increases.
Diabetic macular edema involving the center of the fovea can be associated with considerable early visual acuity reduction and a poor long-term visual prognosis. During pregnancy, diabetic retinopathy may progress to levels that would require treatment, according to criteria commonly used in nonpregnant patients. In nonpregnant patients, the outcome of photocoagulation therapy for diabetic macular edema is inferior to repeated intravitreal injection of vascular endothelial growth factor (VEGF) inhibitors, but concerns about potential fetal side effects and lack of documented experience make the application of specific VEGF inhibition problematic in pregnant women. Case reports of diabetic retinopathy flaring up during pregnancy and waning after delivery also suggest that irreversibly destructive laser photocoagulation may be unnecessary. This prompted us to consider intravitreal glucocorticoid therapy for severe bilateral fovea-involving diabetic macular edema during pregnancy.
Methods
This retrospective, observational, consecutive case series included all patients who presented with diabetic macular edema involving the center of the fovea (Early Treatment Diabetic Retinopathy Study [ETDRS] grid center field thickness >350 μm) during pregnancy between January 1, 2011 and October 31, 2014 and who were treated with intravitreal dexamethasone implant (Ozurdex; Allergan, Inc, Irvine, California, USA). A single additional case was identified as a candidate for injection, but the patient was never treated because of early delivery.
Best-corrected visual acuity (BCVA) was measured using a Snellen chart. For the present analysis, Snellen acuity values were converted to approximated Early Treatment Diabetic Retinopathy Study (approxETDRS) letters using the formula 85 + 50 × log (Snellen fraction) and rounded to the nearest letter. Optical coherence tomography (OCT) was made using a Topcon 3D OCT2000 (Topcon Corporation, Tokyo, Japan) or, in rare cases, a Spectralis/HRA (Heidelberg Engineering AG, Heidelberg, Germany). Intraocular pressure (IOP) was measured using applanation tonometry.
Cases were retrieved from the records of the medical retina clinic to which all cases of suspected diabetic macular edema in pregnant women from the eastern part of Denmark are referred. All pregnant women with known diabetes from this region receive endocrinology and obstetrics care at the Rigshospitalet, where they are being screened for treatment-requiring diabetic retinopathy using fundus photography in early and late pregnancy and, since 2010, also using OCT.
The clinical management of diabetic macular edema presenting during pregnancy was guided by the experience that it is often reversible. This led us to consider first conservative management and then local pharmacotherapy with a dexamethasone intravitreal slow-release implant as means of lessening the immediate burden of visual loss and improving the long-term prognosis for reading vision. The implant procedure was made under controlled aseptic conditions after topical and subconjunctival anesthesia including the application of a povidone-iodide solution on the conjunctiva and periocular skin. Ozurdex was supplied in a prepacked applicator and was injected into the vitreous approximately 3.5 mm posterior of the limbus. Chloramphenicol eye drops were administered immediately after the injection and then 4 times daily for 4 days. Patients were scheduled for follow-up 1 week after the injection. No glucocorticoid eye drops were administered. Complications or side effects included subconjunctival hemorrhage, ocular surface discomfort, postoperative visibility of the implant, and intraocular pressure changes as described below.
Under Danish law, retrospective studies do not require institutional review board approval and request for waivers of approval are rejected if asked for. Retrospective studies do not require patient consent. The study followed the tenets of the Declaration of Helsinki.
Results
We identified 5 cases that were eligible for this retrospective review of cases ( Table 1 ). All pregnancies were desired but had been initiated without prior endocrinology consultation. Injection with intravitreal dexamethasone implant was performed in 9 eyes. Eight eyes were injected once and 1 eye was injected twice. Information was available from 8 follow-up examinations. Changes during the period of investigation in systemic parameters are summarized in Table 2 , whereas changes in retinal thickness and visual acuity are shown graphically in Figures 1 and 2 , respectively.
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | Patient 5 | |
|---|---|---|---|---|---|
| Age a | 32 | 24 | 34 | 35 | 31 |
| Ethnicity | Middle Eastern | Danish | Middle Eastern | Northern European | Danish |
| BMI a (kg/m 2 ) | 26.4 | 29.8 | 27.3 | 17.7 | 24.6 |
| DM type | 2 | 1 | 2 | 1 | 1 |
| DM duration a | 17 years | 22 years | 14 years | 21 years | 24 years |
| DM treatment a | Diet | Lantus 60 IU/day | NovoMix 30 72 IU/day | Lantus, NovoRapid | Lantus 32 IU/day, NovoRapid 10 IU/day |
| Other medications a | Enalapril, simvastatin | None | Losartan, ibuprofen | None | None |
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | Patient 5 | |
|---|---|---|---|---|---|
| Arterial blood pressure (mm Hg) | |||||
| Before pregnancy | – | – | – | – | – |
| 1st trimester | 140/83 a (118–161/73–92) | 126/76 (123–132/75–77) | 120/67 | – | 134/89 (131–136/85–92) |
| 2nd trimester | 126/81 (112–148/68–98) | 121/76 a (109–132/70–79) | 119/68 (102–131/51–81) | – | 140/81 a (126–152/73–86) |
| 3rd trimester | 133/84 b (112–150/58–98) | 116/77 (101–140/70–95) | 117/77 (101–132/67–91) | – | 133/79 (120–153/70–88) |
| Postpartum | 137/85 (120–157/70–96) | 114/78 (101–125/70–83) | – | – | 129/77 (109–137/60–85) |
| HbA 1c (%) | |||||
| Before pregnancy | 5.6 c | 13.5 | 12.2 | 9.4 | 8.1 |
| 1st trimester | 5.6 (5.5–5.6) | 10.2 (8.9–11.5) | 9.5 (8.4–10.5) | – | 8.0 (7.6–8.1) |
| 2nd trimester | 5.4 (5.0–5.7) | 7.2 (6.3–8.8) | 6.7 (6.5–6.8) | – | 6.4 (5.9–7.1) |
| 3rd trimester | 5.7 (5.4–6.0) | 6.3 (6.1–6.4) | 6.4 (6.3–6.4) | 10.7 | 6.4 (6.3–6.6) |
| Postpartum | – | 7.5 (6.7–8.4) | – | – | – |
a Methyldopa treatment begun; continued after delivery.
b Nifedipine treatment begun; continued after delivery.
Case 1
A 32-year-old woman with type 2 diabetes presented with her first pregnancy. Diabetes had been diagnosed at the age of 15 years, when her body weight was 106 kg. Insulin had been used with varying compliance. Elevated carbamide, albuminuria, and hypercholesterolemia approximately 1 year before the pregnancy led to the prescription of enalapril and simvastatin, both of which had been discontinued near the time of conception.
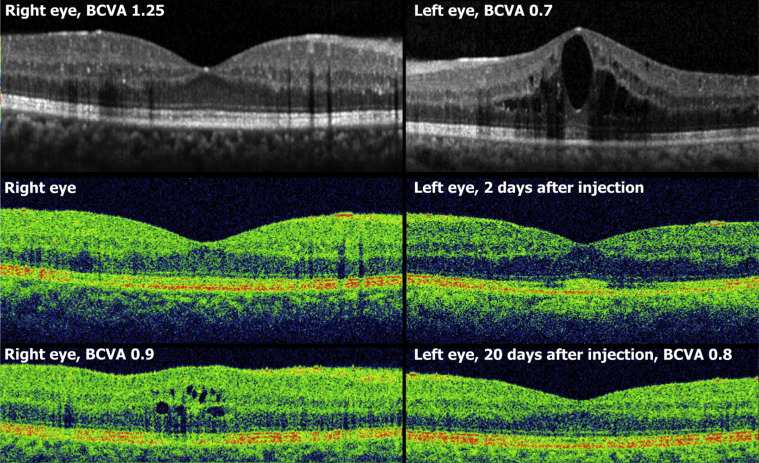
The first photographic screening examination at 10 gestational weeks showed bilateral macular edema. Clinical eye examination at 15 weeks found BCVA 1.2/0.8 in the right and left eye, respectively, center-field retinal thickness (CRT) 622/733 μm, and mild nonproliferative diabetic retinopathy. Modified macular grid photocoagulation was made in the left eye. Two weeks later, at 17 weeks, BCVA was 0.6/0.5, CRT 574/673 μm. At 19 weeks BCVA remained 0.6/0.5, but CRT had increased to 649/727 μm, which prompted injection of a dexamethasone intravitreal implant injected in the left eye. Two weeks later, IOP was 18/18, BCVA 0.8/0.7, and CRT 575/385 μm ( Figure 3 ). Four weeks later, the right eye was injected with a dexamethasone implant. The patient did not present for further eye examinations.
Throughout the pregnancy, HbA 1c remained in the range of 5%–6%. The patient had persistent anemia during, and probably also before, pregnancy, which makes HbA 1c an unreliable index of metabolic control. In week 10 the patient was started on methyldopa and aspirin for hypertension and proteinuria. A healthy girl weighing 1835 g was delivered at 33 completed weeks. The patient failed to report for postnatal eye examinations.
Case 2
A 24-year-old woman with type 1 diabetes for 21 years presented with her first pregnancy. Before pregnancy, BCVA had been 1.0 in both eyes. She was nominally on Lantus 60 IU/day and NovoRapid before meals, but compliance was limited and HbA 1c was 13.5%–14.0% in the year leading up to the pregnancy. Compliance improved during pregnancy and HbA 1c declined rapidly ( Table 2 ). The first fundus photographs, at 8 gestational weeks, showed moderate nonproliferative diabetic retinopathy without diabetic macular edema. In week 13 the patient experienced blurred vision and BCVA was 0.8/1.0. At 23 weeks she was unable to read newspaper print, BCVA was 0.2/0.3, and CRT was 626/670 μm. At 23 weeks and 24 weeks the patient was injected with dexamethasone intravitreal implant in her right and left eye, respectively. One week after the last injection IOP was 18/18, and the macular edema had completely resolved to CRT 237/256 μm. An additional 1 week later BCVA was 0.8/0.6. One month after the last injection BCVA was 0.8/0.7.
At 30 weeks insulin pump treatment was initiated. For the remainder of the pregnancy visual acuity remained stable in both eyes. At 38 weeks, a healthy child was born. Two weeks after delivery BCVA was 0.6/0.7 and there was no edema on OCT. Four months after delivery BCVA was 0.9/1.0 and macular edema was absent. Follow-up OCTs until 6 months after delivery showed no recurrence of edema.
Case 3
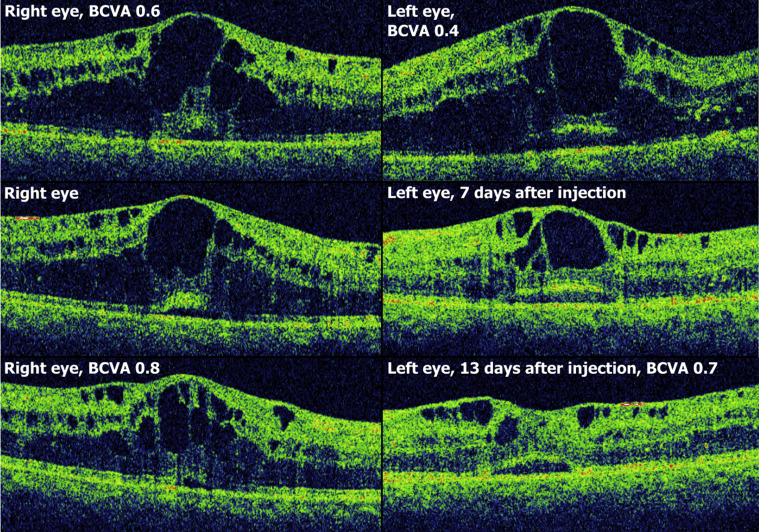
A 34-year-old woman with diabetes type 2 known for 15 years who had previously been diagnosed with microalbuminuria, peripheral neuropathy, and moderate nonproliferative diabetic retinopathy in both eyes and diabetic macular edema in her right eye, for which she had been photocoagulated earlier, presented with an unplanned pregnancy. She had been treated with ibuprofen as needed and losartan, both of which were discontinued after the pregnancy was diagnosed, and Novomix 30 72 IU/day. At gestational week 10 fundus photography showed diabetic macular edema in both eyes, with foveal center involvement in the left eye only. At 14 weeks BCVA was 1.0/0.9 and CRT 308/401 μm. The left eye continued to worsen and at 20 weeks BCVA was 1.25/0.7 and CRT 323/578 μm. Injection of dexamethasone intravitreal implant in the left eye was followed by near-elimination of edema within 3 days. Three weeks after injection, BCVA was 0.9/0.7, IOP 15/15, and CRT 391/266 μm. No treatment was attempted in the right eye ( Figure 4 ). At 34 weeks BCVA was 1.0/0.9 and there was no foveal edema in the left eye. At 37 weeks a healthy girl weighing 3500 g was delivered without complications. The patient was unavailable for further follow-up.