Fig. 8.1
(a) Lateral views showing later development of the pharyngeal arches. (b, c) Ventral or facial views showing the relationship of the first pharyngeal arch to the stomodeum (Cited from Sadler [5])
The first pharyngeal arch plays an important role in formation of the face, the palate, and the superior structures of the oral cavity. Very early during development, the first arch separates into two prominences: the maxillary prominence and the mandibular prominence (Fig. 8.1b). By the end of the fourth week of development, these prominences form four distinct swellings, two maxillary and two mandibular, and are associated with a superior, midline swelling called the frontonasal process. The depression of the surface ectoderm surrounded by these five structures becomes the mouth or stomodeum. The stomodeum is separated from the primordial pharynx by a bilaminar membrane, the oropharyngeal membrane (Fig. 8.1b). At about day 26, the oropharyngeal membrane ruptures to allow the primordial pharynx and foregut to communicate with the amniotic cavity (Fig. 8.1c).
8.1.2.2 Fate of Pharyngeal Arches
The pharyngeal arches contribute extensively to formation of the face, nasal cavities, mouth, larynx, pharynx, and neck. A typical pharyngeal arch contains the following:
An aortic arch
A cartilaginous rod that may contribute to formation of the skeleton
A muscular component
A nerve – innervates all structures derived from the specific pharyngeal arch
The derivatives of pharyngeal arch cartilages are mandible and plate as described above. The horseshoe-shaped primordium of the mandible is formed from ventral portions of the first arch. The cartilage disappears as the mandible develops around the cartilage of the first arch by intramembranous ossification (Fig. 8.2). Other derivatives of the first arch cartilage include the malleus and incus from the dorsal end of the first arch cartilage (Meckel cartilage; Fig. 8.2b) and surrounding ligaments. The remaining arches contribute to provide the supporting skeletal and cartilaginous structures, including the stapes in the middle ear, styloid process and part of hyoid bone from second arch, the remainder of hyoid bone from third arch, laryngeal cartilage from the fourth and sixth arch cartilages (Table 8.1).
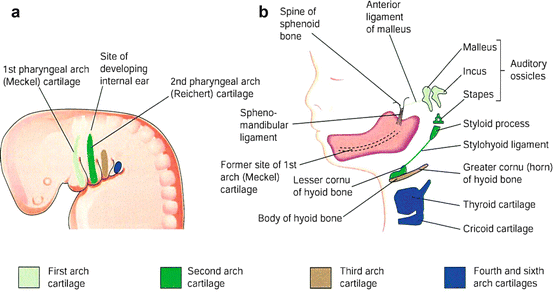

Fig. 8.2
(a) The location of the cartilages in the pharyngeal arches. Lateral view of the head, neck, and thoracic regions of a 4-week embryo. (b) Similar view of a 24-week fetus illustrating the adult derivatives of the arch cartilage (Cited from Moore et al. [6])
Table 8.1
Structures derived from pharyngeal arch components
Arch | Nerve | Muscles | Skeletal Structures | Ligaments |
|---|---|---|---|---|
First (mandibular) | Trigeminal (CN V) | Muscles of mastication Mylohyoid and anterior belly of digastric Tensor tympani Tensor veli palatini | Malleus Incus | Anterior ligament of malleus Sphenomandibular ligament |
Second (hyoid) | Facial (CN VII) | Muscles of facial expression Stapedius Stylohyoid Posterior belly of digastrics | Stapes Styloid process Lesser cornu of hyoid bone Upper part of body of hyoid bone | Stylohyoid ligament |
Third | Glossopharyngeal (CN IX) | Stylopharyngeus | Greater cornu of hyoid bone Lower part of body of hyoid bone | |
Fourth and sixth | Superior laryngeal branch of vagus (CN X) Recurrent laryngeal branch of vagus (CN X) | Cricothyroid Levator veli palatine Constrictors of pharynx Intrinsic muscles of larynx Striated muscles of esophagus | Thyroid cartilage Cricoid cartilage Arytenoid cartilage Corniculate cartilage Cuneiform cartilage |
The musculature derivatives of the first pharyngeal arch are the muscles of mastication and other muscles (Fig. 8.3; Table 8.1). The stapedius muscle, stylohyloid muscle, posterior belly of the digastric muscle, auricular muscles, and muscles of facial expression are derived from the second pharyngeal arch. The stylopharyngeus muscle is derived from the third pharyngeal arch; the cricothyroid, levetor veli palatine, and constrictor muscles of the pharynx are derived from the fourth pharyngeal arch; and the intrinsic muscles of the larynx come from the sixth pharyngeal arch.
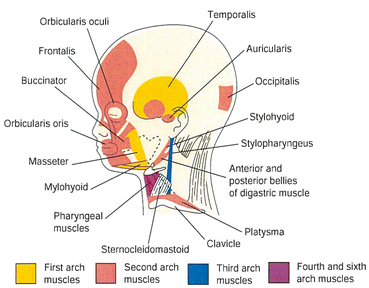

Fig. 8.3
Lateral view of a 20-week fetus showing the muscles derived from the pharyngeal arches. Myoblasts from the second arch migrate from the neck to the head, then form the muscles of facial expression (Cited from Sadler [5])
A specific cranial nerve (CN) supplies each arch. The dermis and mucous membranes of the head and neck are related to the mesenchyme from the pharyngeal arches, so that these areas are supplied with special visceral afferent nerves. The trigeminal nerve (CN V) is the principal sensory nerve of the head and neck, and its caudal two branches (maxillary and mandibular) innervate the teeth and mucous membranes of the palate, mouth, and tongue (Fig. 8.4). The motor nerve of CN V innervates the muscles of mastication. The second, third, and caudal (fourth to sixth) arches are supplied by the facial nerve (CN VII), the glossopharyngeal nerve (CN IX), and the vagus nerve (CN X), respectively. The second to sixth pharyngeal arches innervate the mucous membranes of the tongue, pharynx, and larynx, respectively.
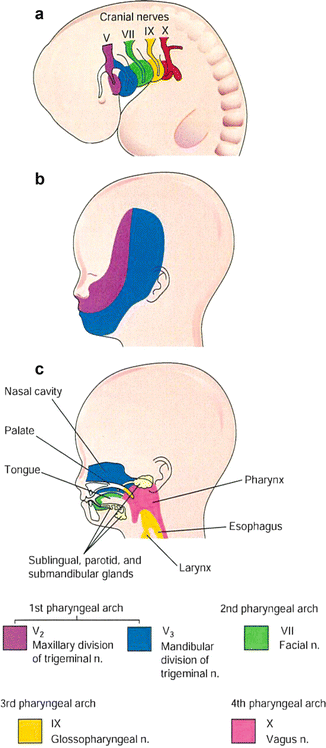

Fig. 8.4
(a) Lateral view of a 4-week embryo showing innervation by the cranial nerves to the pharyngeal arches. (b) Two caudal branches of the first arch nerve (CN V) distribution of a 20-week fetus. (c) The deep distribution of sensory fibers of the nerves to the teeth and mucosa of the tongue, pharynx, nasal cavity, palate, and larynx (Cited from Sadler [5])
The spaces between the arches are clefts externally and pouches internally. Only the first pair of clefts persist as the external acoustic meatus, while the other clefts are obliterated. The expanding first pouch becomes the tympanic cavity and auditory or Eustachian tube. The remainder contributes to glandular tissue of the head and neck, including the palatine tonsillar crypts from the second pouch, the inferior parathyroid glands and thymus from the third pouch, and the superior parathyroid gland from the fourth pouch (Table 8.1). These tissues will migrate to their adult locations.
The tongue is a complex structure consisting of mucosa, muscles, and both general and special sensory innervation. The anterior two-thirds of tongue is formed from mesoderm of the first pharyngeal arch, whereas second to fourth arch mesoderm contributes to the posterior one-third of the tongue, which descends into the oropharynx by 4 years of age. These two tongue regions are separated by a V-shaped cleft called the terminal sulcus. Most of the tongue muscles are derived from myoblasts that migrate from the occipital myotomes, and are innervated by the hypoglossal nerve (CN XII).
8.1.2.3 Molecular Regulation of Mesenchyme Development
Neural crest cells develop from neuroepithelial cells of the surface ectoderm along the edges of the neural plate. Bone morphogenetic protein (BMP) signaling is important in establishing this edge region and then regulates WNT1 expression, which drives neural crest cells to undergo an epithelial–mesenchymal transition and begin migration into the surrounding mesenchyme. Some cranial neural crest cells originate from rhombomeres, which consists of eight segments in the hindbrain, and migrate into specific pharyngeal arches. There are three main streams of neural crest cell migration from rhombomeres: R1 and R2 migrate to the first arch along with crest cells from the caudal midbrain region; R4 migrate to second arc; and R6 and R7 migrate to arches 4 to 6 (Fig. 8.5). These three distinct streams of neural crest cells provide axonal guidance cues for axons from ganglia forming in the head and neck region. Axons from the trigeminal ganglion enter the hindbrain at R2, while those from the geniculate and vestibuloacoustic ganglia enter at R4 and those from the petrosal and nodose ganglia enter at R6 and R7. No axons project to R3 and R5.
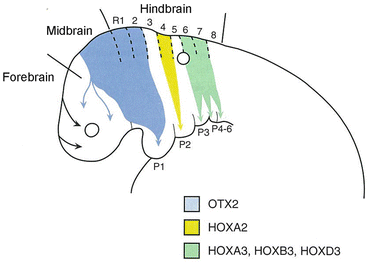

Fig. 8.5
The three streams of neural crest cell migration from the cranial folds into the face and pharyngeal arches. They provide guidance cues for cranial nerve axon outgrowth (Cited from Sadler [5])
Neural crest cells populating the pharyngeal arches form the skeletal components characteristic of each arch and this process is controlled by pharyngeal pouch endoderm. As pouches form, they express a very characteristic pattern of genes (Fig. 8.6). BMP7 is expressed in the posterior endoderm, FGF8 is expressed in the anterior endoderm, PAX1 expression is restricted to the dorsal-most endoderm of each pouch, and SHH is expressed in the posterior endoderm of the second and third pouches. These expression patterns regulate differentiation and patterning of pharyngeal arch mesenchyme into specific structures. In addition, there are different expression patterns of transcription factors in each mesenchyme and they dictate the fate of development. As for the first arch, HOX genes are not expressed, but OTX2, a homeodomain-containing transcription factor that is also expressed in midbrain, is expressed. The second arch shows HOXA2 expression, while arches 3 to 6 express members of the third paralogous group of HOX genes, HOXA3, HOXB3, and HOXD3 (Fig. 8.6b) [5]. With the influence of such diversity of transcription factors, each arch receives signals emanated from pouch endoderm and forms arch-specific structures.
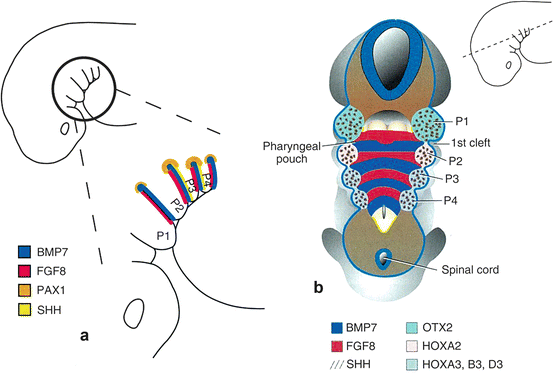

Fig. 8.6
(a) The gene expression patterns in pharyngeal arch endoderm and mesenchyme. Endoderm is responsible for patterning the skeletal derivatives of the arches, but the response of the mesenchyme to these signals is dictated by the genes that the mesenchyme express. (b) Mesenchyme expression patterns. Neural crest cells from the rhombomeres migrate into the arches and establish the patterns (Cited from Sadler [5])
8.2 Regeneration
Surgical resection of tumors causes tissue defects following head and neck surgery. In oropharynx, wide defects of tissue cause various problems, such as disturbance of mastication, deglutition, and articulation, leading to a decreased quality of life. Thus, there is a need for a reconstructive or regenerative approach to restore lost tissues and prevent postoperative complications. In oropharynx, the reconstructive approach is a mainstay of treating surgical defects so far, and there are few references in the literature regarding the regenerative approach. The reconstructive approach using free flaps has advantages in immediate covering of tissue defects without xenobiotic rejection [8]. However, there exist problems such as the stretching ability in free flaps due to the difference of tissue characteristics. Thus, the regenerative approach should also be evolving in oropharynx.
Tissue engineering is an interdisciplinary field that applies the principles of engineering and the life sciences toward tissue regeneration [9]. Three elements have been adopted for tissue regeneration: regulatory factors, cells, and scaffolds. Tissue regeneration will be achieved when these three elements are appropriately provided. At present, there are some reports regarding each element.
Regulatory factors modulate cell behavior such as extracellular matrix production, cell proliferation and differentiation. Growth factors are peptide molecules produced by various cells and representative regulatory factors.
Transforming growth factor (TGF) β3 is one of the TGFβ isoforms that activate nuclear-translocating Smads and other intercellular proteins, leading to altered expression of target genes by first binding to TGFβ type II receptors before pairing together with two type I receptors [10]. Embryonic wounds are known to heal perfectly with no scars [11]. There are far fewer inflammatory cells in embryonic wounds, the inflammatory cells present are less differentiated, and the length of time that inflammatory cells are present is markedly reduced compared with adult wounds [12]. Embryonic wounds express very high levels of TGFβ3 but very low levels of TGFβ1 and TGFβ2 compared to adult wounds [11




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


