Purpose
To determine and validate thresholds of anterior chamber angle (ACA) parameters in discriminating open- vs narrow-angle eyes using images from dedicated anterior segment optical coherence tomography (DASOCT).
Design
Reliability analysis.
Methods
Eyes imaged by DASOCT and examined with gonioscopy were reviewed. By gonioscopy, eyes were classified as narrow if posterior trabecular meshwork was not visible and open if the angle was open to scleral spur and beyond. Imaging was performed in the dark with the CASIA SS-1000 (Tomey, Nagoya, Japan). ACA parameters angle opening distance (AOD), trabecular-iris space area (TISA), trabecular-iris circumference volume (TICV), length of iridotrabecular contact (ITC), and extent and area of ITC were calculated. AOD, TISA, and TICV were measured at both 500 μm and 750 μm from the scleral spur landmark. Study eyes were randomly divided into training and testing sets. Thresholds were calculated from the training set. Sensitivity, specificity, and agreement were then calculated against the clinical classification using the testing set.
Results
One hundred and eighty-nine eyes (111 open, 78 narrow, including 8 borderline angles) were included. Using the threshold determined in the training set on the testing set, 9 of 24 ACA parameters had no misclassification of narrow-angle eyes as open (sensitivity = 1.0). Of 9 parameters, the specificity was >0.79. The highest kappa values (kappa = 0.91) was AOD750 inferiorly at 0.31 mm threshold, followed by both TICV500 at 1.92 μL and TICV750 at 4.00 μL with kappa = 0.86.
Conclusion
The parameters with the best discriminative ability for detecting narrow angles were AOD750 inferiorly, TICV500, and TICV750.
The primary angle-closure spectrum of disease occurs as a result of predisposing anatomic relationships between structures in the peripheral angle of the anterior chamber and, therefore, is managed differently than open-angle glaucoma. Consequently, assessment of structures in the anterior chamber angle (ACA) is essential in the diagnosis and management of these disorders.
Although gonioscopy remains the clinical gold standard for grading the ACA, it is a subjective and difficult skill to learn, and assessment of the angle anatomy can be affected by technique. Advances in imaging have resulted in our ability to measure the angle quantitatively. Dedicated anterior segment optical coherence tomography (DASOCT) is one such advance and is a noncontact technique that can be used to image and evaluate the ACA objectively. DASOCT uses a 1310 nm central wavelength, reducing light scatter through tissue such as the sclera and iris, and allowing deeper penetration of the peripheral angle, as compared with an 840- to 870-nm central wavelength, which is used with commercially available devices not dedicated to imaging the anterior segment. The newer generation of Fourier-domain DASOCT uses swept source technology, which provides higher image acquisition speed (30 000 A-scans per second) compared with time-domain DASOCT (2048 A-scans per second). The dramatic improvement in image acquisition speed allows reduction of motion artifacts, increased image resolution, and assessment of the entire 360 degrees of the ACA.
Parameters originally developed using ultrasound biomicroscopy, angle opening distance (AOD) and angle recess area (ARA), can be measured using DASOCT with improved repeatability and reproducibility. Three additional parameters have been developed using DASOCT: trabecular-iris space area (TISA), trabecular-iris circumference volume (TICV), and iridotrabecular contact length (ITC length). The definitions of and the relationship among these parameters can be seen in Figure 1 .
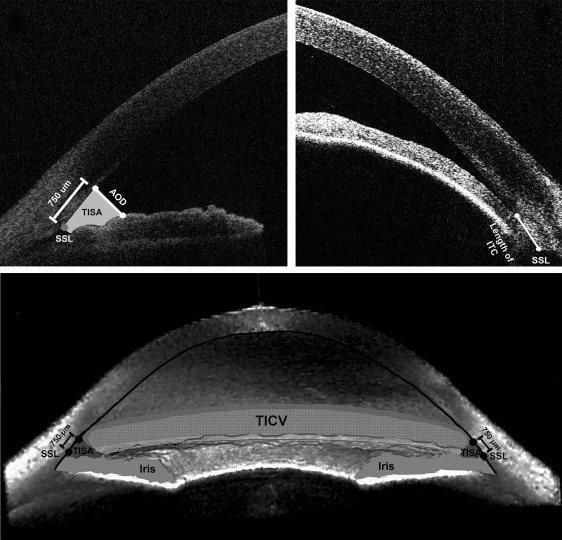
ACA parameters obtained from DASOCT, including AOD, TISA, and TICV, have been variably studied for their repeatability, reproducibility, and agreement and used to evaluate angle anatomy after laser peripheral iridotomy or lens extraction. Previous studies have determined threshold values for AOD, ARA, and TISA obtained from horizontal meridian angles (temporal and nasal) to identify a predisposition to angle closure. Threshold values for the other ACA parameters have not yet been determined. In addition, none of the thresholds have been validated. Before further clinical evaluation of these parameters, validated thresholds to discriminate narrow angles are required. The purpose of this study is to determine the threshold values for ACA parameters by optimizing sensitivity and specificity to identify narrow angles from open angles as well as validating these thresholds using an independent dataset.
Methods
This reliability analysis reviewed 4 prospective cohort studies previously conducted at the Robert Cizik Eye Clinic of the Ruiz Department of Ophthalmology and Visual Science at the McGovern Medical School at The University of Texas Health Science Center in Houston (UTHealth). The University of Texas Health Science Center Committee for the Protection of Human Subjects determined that this study was exempt from review and approved it. All research adhered to the Declaration of Helsinki and was HIPAA compliant, and data collection was in conformity with all relevant laws.
Participants
All participants enrolled in the 4 previous Institutional Review Board–approved prospective studies were 18 years of age and older. All eyes were phakic and underwent DASOCT imaging and gonioscopic examination on the same day or within 2 weeks before imaging. Eyes were excluded if they had (1) anterior segment abnormalities that could affect the angle parameters, such as significant corneal opacity; (2) lid obstruction or eye movement artifact that could not properly be imaged; and (3) been dosed with any medication that may have affected angle anatomy within a month before imaging (ie, pilocarpine or atropine). When both eyes qualified, 1 eye was randomly selected for the study.
Two of these 4 studies recruited participants with narrow angles for imaging before receiving laser iridotomy (26 eyes) or cataract extraction (16 eyes) (Minnal et al, American Glaucoma Society 24th Annual Meeting, February 27-March 2, 2014, Washington, DC). Patients with nanophthalmos (axial length less than 18 mm) were excluded from the cataract extraction study. Another study recruited only phakic, open-angle eyes from patients, visitors, volunteers, and staff (99 eyes). The fourth study recruited participants with both open (12 eyes) and narrow angles (36 eyes) (Rigi et al, American Glaucoma Society 25th Annual Meeting, February 26-March 1, 2015, Coronado, CA). Additionally, eyes that underwent any intraocular procedure within 90 days before imaging were excluded from the fourth study.
Procedures
Demographics (age, race, and sex), type of glaucoma, intraocular pressure (IOP), and number of IOP-lowering medications were recorded. IOP was measured by Goldmann applanation tonometry.
Gonioscopy was performed by 1 of 3 experienced examiners (R.M.F., N.P.B., L.S.B.) using a Posner goniolens in the dark and graded using the Spaeth grading system (grading determined by the deepest visible ACA structure without indentation: A = anterior to the Schwalbe’s line; B = between the Schwalbe’s line and scleral spur; C = scleral spur; D = ciliary body; and E = beyond 0.1 mm of ciliary body). Presence of peripheral anterior synechiae (PAS) was examined with indentation gonioscopy and recorded.
Using the Spaeth grading system, eyes were considered to have open angles if anything beyond the scleral spur was visible (grade D or E); all other eyes were graded as narrow (A or B) based on the deepest structure visible. For angles graded as C where the scleral spur was partially visualized, the classification as narrow or open was based on the clinical decision of whether treatment was required. Criteria clinically invoked to determine if a grade C eye needed treatment were the presence of PAS, steep iris insertion, pigment smudging anterior to trabecular meshwork, or clinical symptoms suggestive of intermittent angle closure.
Instrument details for the CASIA SS-1000 (Tomey, Nagoya, Japan) have been previously reported. Software and image analysis procedures have also been previously described. Briefly, eyes were imaged using 3D mode with the angle analysis scan option in a dark room. The raw image files were imported into the Anterior Chamber Analysis and Interpretation software (ACAI, Houston, Texas, USA). The ACAI software divides 128 2-dimensional (2D) images into 8 panels, 16 images per panel (11.25 degrees between 2 consecutive angles). The images were read by 1 of 2 experienced readers (A.Z.C. or L.A.B.), who were masked to the gonioscopic grading. The readers marked the scleral spur landmarks (SSLs) on each image in the first panel (this panel includes horizontal and vertical meridian images), and then the ACAI software automatically detected corneal and iris edges. If the edges of the cornea and iris were not accurate, the readers manually adjusted the intensity and, if that was not successful, manually adjusted the edge margins. Once the readers had completed and saved the interpreted results of the first panel, ACAI interpolated the SSLs in the remaining panels using the first panel result and detected edges (and assembled a 3-dimensional [3D] reconstruction of the angle).
Anterior Chamber Angle Parameters
After performing a correction for refraction, the ACAI software calculated the following ACA parameters automatically, which are illustrated in Figure 1 . These parameters were grouped into linear, area, and volume parameters. The horizontal image (nasal and temporal) and vertical image (superior and inferior) were used for linear and area parameters, and all images were used for 360-degree parameters.
Linear parameters
Angle Opening Distance (AOD500 and AOD750, mm) at Nasal, Temporal, Superior, and Inferior: the length of a line drawn perpendicular to the cornea starting 500 or 750 μm anterior to the SSL and ending on the anterior surface of the iris.
Iridotrabecular Contact (ITC, mm) Length at Nasal, Temporal, Superior, and Inferior: the length of a line segment from SSL to the most anterior point at which the iris contacts the corneoscleral-aqueous interface. If the ITC point locates peripherally to SSL, the ITC length is defined as 0.
Area parameter
Trabecular-Iris Space Area (TISA500 and TISA750, mm 2 ) at Nasal, Temporal, Superior, and Inferior: TISA500 (or TISA750) is the area bordered centrally by the AOD500 (or AOD750) line, anteriorly by the posterior corneoscleral-aqueous interface, and posteriorly by the anterior surface of the iris. The peripheral border is a line segment starting at the SSL perpendicular to the corneoscleral-aqueous interface and ending on the anterior surface of the iris.
360-degree parameters
Trabecular-Iris Circumference Volume (TICV500 and TICV750, μL): the integrated volume of the peripheral angle taken from TISA500 or TISA750.
Extent of Iridotrabecular Contact (Extent ITC, degrees): degrees of angle presence of ITC (ITC length >0).
Iridotrabecular Contact Area (ITC area, mm 2 ): the integrated area of the peripheral angle taken from ITC length.
The image reading procedure has been validated in several previous studies, and intrareader and interreader correlation coefficients were excellent (>0.9) for TISA750 and AOD750 for all angles. Additionally, we have found that 16 meridians were sufficient to estimate TICV750 with less than 5% mean absolute percent error.
Data Analysis
Data collected were summarized by mean (± standard deviation) for continuous variables or by frequency (%) for discrete variables. The data were divided into a training set and testing set. One hundred and twenty eyes were randomly selected using SAS “procsurveyselect” to form a training set, and the remaining 69 eyes were assigned to a testing set. The training set was used to determine the thresholds, and accuracy was verified via the testing set.
Threshold determination
The training set was further divided into 3 random subsets with 40 eyes in each. Each training subset was then used to compute the area under receiver operating characteristic curve (AUROC) and Youden optimal threshold for each ACA parameter using logistic regression analysis. The Youden optimal threshold provides an equal weight to false-positive and false-negative values and is determined by maximizing (sensitivity + specificity − 1) from the receiver operating characteristic (ROC) curve.
The training procedure was repeated 500 times, and the recommended threshold for each parameter was determined by the mean of 1500 replicates (= 500 times × 3 subsets). In addition, mean AUROC, 95% confidence limits, and means of corresponding sensitivity and specificity were calculated for each parameter.
Verification
The recommended Youden optimal thresholds obtained from the training set were applied to the testing data set, and sensitivity, specificity, and kappa were calculated to validate the thresholds. The accuracy statistics were further evaluated with a bootstrapping procedure. Sixty-nine samples were randomly selected with replacement from the testing set, and accuracy statistics were calculated using the recommended Youden optimal thresholds. The procedure was repeated 500 times, and the mean, minimum, and maximum of accuracy statistics were calculated to assess the accuracy in classifying an eye as either an open or narrow angle.
All statistical analyses were performed using SAS for Window 9.4 (SAS Inc, Cary, North Carolina, USA) and R 3.0.3 with optimal.cutoffs() in optimal.cutoffs package. A P value less than .05 was considered statistically significant. The kappa criteria were <0.2 poor; 0.21–0.40 fair; 0.41–0.60 moderate; 0.61–0.80 good, and >0.80 excellent. It should be noted that this is a statistical classification terminology that may not reflect clinical terminology.
Results
Participants
One hundred and eighty-nine participants were included in the study. One hundred thirty-two (70%) female and 57 (30%) male participants were enrolled, and the average age was 54.0 (± 14.1) years. Ninety-four participants (50%) were white, 44 (23%) black, 27 (14%) Hispanic, and 24 (13%) Asian. One hundred and eleven eyes (59%) were classified as open angles and 78 eyes (41%) were classified as narrow angles on gonioscopy. Eight eyes were graded “C” on gonioscopy, with 4 classified as narrow and 4 classified as open. There was no significant racial difference between the 2 groups ( P = .21). However, open-angle participants were significantly younger (49.1 ± 14.9 years) than narrow-angle participants (60.9 ± 9.2 years) ( P < .001). Sixty-three participants (81%) with gonioscopically narrow angles and 69 (62%) gonioscopically open angles were female ( P = .006) ( Table 1 ).
| Variable | Open-Angle Eyes (N = 111) | Narrow-Angle Eyes (N = 78) | P Value |
|---|---|---|---|
| Age, y (± SD) | 49.1 (± 14.9) | 60.9 (± 9.2) | <.001* |
| Sex, N female (%) | 69 (62%) | 63 (81%) | .006* |
| Race, N (%) | .21 | ||
| White | 60 (54%) | 34 (44%) | |
| Black | 26 (23%) | 18 (23%) | |
| Hispanic | 11 (10%) | 16 (21%) | |
| Asian | 14 (13%) | 10 (13%) | |
| Eye, N right (%) | 50 (45%) | 38 (49%) | .66 |
| IOP, mm Hg (± SD) | 15.0 (± 3.4) | 15.7 (± 3.6) | .17 |
| Number of eyes on IOP-lowering medications (%) | 20 (18%) | 16 (21%) | .71 |
| Glaucoma diagnosis, N (%) a | <.001* | ||
| Normal | 67 (61%) | 0 (0%) | |
| POAG | 14 (13%) | 2 (3%) | |
| POAG suspect | 29 (26%) | 0 (0%) | |
| PACG | 0 (0%) | 9 (12%) | |
| PAC | 0 (0%) | 21 (27%) | |
| PACS | 0 (0%) | 46 (59%) | |
| Spaeth gonioscopic grade, N (%) | <.001* | ||
| A | 0 (0%) | 34 (44%) | |
| B | 0 (0%) | 40 (51%) | |
| C | 4 (4%) | 4 (5%) | |
| D | 45 (41%) | 0 (0%) | |
| E | 62 (56%) | 0 (0%) | |
| Peripheral synechiae, N (%) | 0 (0%) | 20 (25%) | <.001* |
Ocular Characteristics
Treated IOPs were similar between open-angle eyes (15.0 ± 3.4 mm Hg with 20 eyes [18%] on IOP-lowering medications) and narrow-angle eyes (15.7 ± 3.6 mm Hg with 16 eyes [21%] on IOP-lowering medications) ( P = .17). The distribution of type of glaucoma, Spaeth gonioscopic grades, presence of PAS, and ACA parameters are summarized by groups and shown in Tables 1 and 2 . All 24 ACA parameters were significantly different between the 2 groups ( P values < .001).
| Parameter | Open-Angle Eyes (N = 111) | Narrow-Angle Eyes (N = 78) | P Value |
|---|---|---|---|
| AOD (mm) | |||
| 500 @ Temporal | 0.483 (± 0.200) | 0.141 (± 0.082) | <.001 |
| 500 @ Nasal | 0.491 (± 0.233) | 0.116 (± 0.079) | <.001 |
| 500 @Superior | 0.383 (± 0.182) | 0.061 (± 0.055) | <.001 |
| 500 @ Inferior | 0.457 (± 0.213) | 0.109 (± 0.090) | <.001 |
| 750 @ Temporal | 0.658 (± 0.249) | 0.235 (± 0.105) | <.001 |
| 750 @ Nasal | 0.669 (± 0.296) | 0.233 (± 0.137) | <.001 |
| 750 @ Superior | 0.540 (± 0.224) | 0.144 (± 0.085) | <.001 |
| 750 @ Inferior | 0.644 (± 0.274) | 0.169 (± 0.094) | <.001 |
| TISA (mm 2 ) | |||
| 500 @ Temporal | 0.154 (± 0.070) | 0.033 (± 0.028) | <.001 |
| 500 @ Nasal | 0.159 (± 0.082) | 0.032 (± 0.031) | <.001 |
| 500 @ Superior | 0.107 (± 0.065) | 0.011 (± 0.012) | <.001 |
| 500 @ Inferior | 0.148 (± 0.080) | 0.021 (± 0.019) | <.001 |
| 750 @ Temporal | 0.290 (± 0.118) | 0.078 (± 0.047) | <.001 |
| 750 @ Nasal | 0.297 (± 0.139) | 0.074 (± 0.049) | <.001 |
| 750 @ Superior | 0.220 (± 0.112) | 0.035 (± 0.027) | <.001 |
| 750 @ Inferior | 0.280 (± 0.133) | 0.055 (± 0.039) | <.001 |
| TICV (μL) | |||
| 500 | 4.757 (± 2.277) | 0.794 (± 0.563) | <.001 |
| 750 | 8.930 (± 3.850) | 1.950 (± 1.052) | <.001 |
| ITC length (mm) | |||
| @ Temporal | 0.015 (± 0.048) | 0.182 (± 0.162) | <.001 |
| @ Nasal | 0.007 (± 0.032) | 0.189 (± 0.219) | <.001 |
| @ Superior | 0.061 (± 0.093) | 0.391 (± 0.240) | <.001 |
| @ Inferior | 0.029 (± 0.089) | 0.311 (± 0.269) | <.001 |
| Extent of ITC (degrees) | 45.7 (± 70.9) | 279.1 (± 79.8) | <.001 |
| ITC area (mm 2 ) | 1.13 (± 1.76) | 9.56 (± 5.53) | <.001 |
Area Under the Receiver Operating Characteristic Curve and Thresholds for Anterior Chamber Angle Parameters
Of 120 eyes in the training set, 73 (61%) had gonioscopically open angles and 47 (39%) had gonioscopically narrow angles. Figure 2 (Right) demonstrates the estimated curve for TICV750 using the 40 random samples from the training set. The threshold determination is shown in Figure 2 (Left). In this particular example, the maximum Youden criterion (y-axis) was 0.73, and the corresponding optimal threshold was 4.067 μL for TICV750 with a sensitivity of 0.94, specificity = 0.79, and AUROC = 0.93.
AUROC and Youden optimal threshold, along with 95% confidence limits, sensitivity, and specificity obtained from each ACA parameter, are reported in Table 3 . All ACA parameters investigated had an excellent mean AUROC (>0.90), and 20 of 24 parameters had mean AUROC greater than 0.95. The smallest AOD and TISA thresholds were in the superior angle.
| Parameter | AUROC | Mean Threshold [95% Confidence Interval] | Mean Sensitivity/Mean Specificity at Threshold |
|---|---|---|---|
| AOD (mm) | |||
| 500 @ Temporal | 0.967 | 0.28 [0.17, 0.39] | 0.96/0.89 |
| 500 @ Nasal | 0.974 | 0.23 [0.14, 0.32] | 0.94/0.92 |
| 500 @Superior | 0.980 | 0.15 [0.08, 0.21] | 0.98/0.91 |
| 500 @ Inferior | 0.946 | 0.24 [0.21, 0.26] | 1.00/0.90 |
| 750 @ Temporal | 0.969 | 0.37 [0.29, 0.46] | 0.94/0.93 |
| 750 @ Nasal | 0.927 | 0.39 [0.33, 0.46] | 0.97/0.83 |
| 750 @ Superior | 0.980 | 0.24 [0.19, 0.29] | 0.97/0.94 |
| 750 @ Inferior | 0.973 | 0.31 [0.25, 0.37] | 0.99/0.92 |
| TISA (mm 2 ) | |||
| 500 @ Temporal | 0.962 | 0.079 [0.045, 0.113] | 0.95/0.90 |
| 500 @ Nasal | 0.955 | 0.070 [0.037, 0.103] | 0.93/0.91 |
| 500 @ Superior | 0.973 | 0.028 [0.012, 0.043] | 0.99/0.91 |
| 500 @ Inferior | 0.959 | 0.054 [0.045, 0.064] | 1.00/0.91 |
| 750 @ Temporal | 0.967 | 0.162 [0.031, 0.101] | 0.97/0.89 |
| 750 @ Nasal | 0.966 | 0.134 [0.079, 0.190] | 0.93/0.93 |
| 750 @ Superior | 0.983 | 0.073 [0.042, 0.103] | 0.98/0.92 |
| 750 @ Inferior | 0.962 | 0.120 [0.098, 0.142] | 1.00/0.91 |
| TICV (μL) | |||
| 500 | 0.983 | 1.92 [1.26, 2.57] | 0.98/0.93 |
| 750 | 0.984 | 4.00 [2.95, 5.06] | 0.98/0.93 |
| ITC Length (mm) | |||
| @ Temporal | 0.910 | 0.030 [0.000, 0.112] | 0.86/0.88 |
| @ Nasal | 0.926 | 0.017 [0.000, 0.089] | 0.89/0.90 |
| @ Superior | 0.961 | 0.159 [0.067, 0.250] | 0.99/0.89 |
| @ Inferior | 0.950 | 0.054 [0.000, 0.112] | 0.95/0.93 |
| Extent of ITC (degrees) | 0.981 | 133 [83, 184] | 0.99/0.92 |
| ITC Area (mm 2 ) | 0.979 | 3.27 [1.40, 5.15] | 0.99/0.91 |
Thresholds Validation
Of 69 eyes in the testing set, 38 eyes (55%) were gonioscopically open angles and 31 (45%) were gonioscopically narrow. The results of applying the Youden optimal threshold values determined by the training set ( Table 3 ) to the testing set are shown in Table 4 . Ten of 24 ACA parameters (AOD500 at nasal and inferior angles; AOD750 at temporal, superior, and inferior angles; TISA500 and TISA750 at inferior angle; TICV500; TICV750; and ITC area) had excellent agreement (kappa >0.80) between DASOCT classification using the Youden optimal thresholds and the gonioscopic clinical classification of the angle. The highest kappa was 0.91 for AOD750 inferiorly, followed by TICV500 and TICV750 (kappa = 0.86 for both). For TISA parameters, the inferior angle had the best agreement (kappa = 0.83 for both TISA500 and TISA750 at inferior angle). For ITC length, kappas in all 4 quadrants (≤0.65) had relatively lower agreement compared with the other ACA parameters.



