Purpose
To evaluate a novel technique of Descemet membrane endothelial keratoplasty (DMEK) donor insertion using the EndoGlide pull-through device.
Design
Prospective, interventional cases series.
Methods
We performed DMEK using the EndoGlide technique with a protoype donor carrier device (Descemet Mat, or D-Mat) in 30 patients by a single surgeon (including learning curve). Clinical data and donor and recipient characteristics were tracked from our prospective Singapore Corneal Transplant Registry. Main outcome measures were intraoperative complications and the incidence of postoperative graft detachment and primary graft failures within 3 months from surgery.
Results
In our preliminary study of 30 eyes in 30 patients (73% female, mean age 64.5 ± 8.3 years), we observed 3 cases (10%) of partial donor detachment requiring rebubbling and 1 (3%) primary donor failure. Twenty of 30 eyes (66.7%) achieved ≥20/25 3-month best spectacle-corrected visual acuity. Six-month endothelial cell loss reduced from 65% ± 13% to 48% ± 11% ( P = .02) comparing the first and last 15 cases. With the exception of 1 case with excessive fibrin formation, we did not experience any further intraoperative complications related to donor tissue insertion, incomplete tissue unfolding, donor tissue malposition, or incorrect orientation.
Conclusion
Our study suggests that the described technique allows for donor control and insertion in the correct orientation with the endothelium facing down. Further studies are required to confirm if this DMEK donor insertion technique can lead to reduced endothelial cell loss and better graft survival.
Endothelial keratoplasty (EK) is rapidly becoming the surgical treatment of choice for endothelial dysfunction, owing to its advantages over penetrating keratoplasty—such as a more rapid visual recovery and preservation of ocular structural integrity. The first EK procedure, deep lamellar endothelial keratoplasty, was introduced in the 1990s, but its adoption was slow owing to the technical difficulties (ie, the deep manual lamellar dissection of host cornea). Subsequently, Descemet stripping (automated) endothelial keratoplasty (DSEK/DSAEK) was developed and gained popularity as the surgical technique was refined with stripping of the recipient endothelium and Descemet membrane (DM). However, both procedures involve adding corneal stroma with the donor graft, which may result in delayed visual recovery and hyperopic shift.
Descemet membrane endothelial keratoplasty (DMEK) is theoretically the most anatomically precise method of endothelial replacement, as it involves the selective removal and replacement of diseased with donor endothelium with DM. Its potential advantages over current EK procedures include faster and better visual recovery with minimal refractive change, as well as reduced graft rejection. However, the major limiting factor in DMEK is that the scrolled-up donor in the anterior chamber (AC) creates a more significant surgical challenge than DSAEK, and complications related to excessive surgical manipulation required to unscroll and position the donor include inadvertent tissue eversion, excessive endothelial cell damage, and higher rates of primary graft failure, rebubbling, and endothelial cell loss. Development of devices for donor insertion have greatly simplified DSAEK, with glides such as EndoGlide (AngioTech, Reading, Pennsylvania, USA/Network Medical Products, North Yorkshire, UK) and the Busin glide (Asico, Westmont, Illinois, USA), or inserters such as the Endoserter (Ocular Systems, Winston-Salem, North Carolina, USA), whereas DMEK surgical approaches to insertion and donor manipulation have not changed significantly, apart from a pull-through technique recently described for DMEK insertion with a contact lens trifold.
The main challenge in DMEK surgery is that donor DM scrolls with the endothelial layer outermost, and attempts to unscroll the donor the right way up in the AC and position it onto the cornea, without minimal direct handling of the tissue, requires skill and patience, with surgeons essentially just using gentle bursts of balanced salt solution (BSS), the use of small air bubbles in the AC, and indirect massaging of the DMEK donor from the corneal surface. Our approach was to radically alter the concept of DMEK surgery in 2 ways: firstly, to develop a technique that does not allow the donor to naturally scroll onto itself (which then obviates the unscrolling of the donor in the eye); and secondly, to develop a method that allows better manipulation of the delicate DM with minimal touch or damage to the endothelium while in the AC. To achieve both of these objectives, we reviewed exactly how DSAEK donor manipulation differed from DMEK—in DSAEK, the presence of a stromal surface still attached to the DM prevented both scrolling and wrinkling up of the DM, and essentially acted as a stable carrier for the DM and endothelium. When we developed the EndoGlide, we essentially used this stromal carrier to coil the donor tissue endothelium inward, into the insertion chamber (thus minimizing endothelial damage), and directly pulled the tissue into the eye by using forceps, which grasped the stromal edge of the tissue. Our novel approach to DMEK uses this same concept, whereby we reintroduce a stromal carrier (the Descemet Mat, or D-Mat) to support and stabilize the donor DM. This prevented the DM from naturally scrolling endothelial side outward, as well as enabling similar “endothelium-in” coiling into the EndoGlide chamber, without touching the DM. For donor insertion, the EndoGlide is inserted through a similar 4.5-mm scleral tunnel wound, and the curved EndoGlide insertion forceps is used to pull just the DM donor into the AC, leaving the stromal carrier behind in the EndoGlide cartridge. While the surgeon is still holding on to 1 end of the DM, the EndoGlide cartridge is removed, and air is injected into the AC to fully position the donor.
We therefore conducted a pilot study to evaluate this novel technique of DMEK using the EndoGlide and a prototype DM carrier, which we hoped would simplify the insertion and positioning of the donor graft during the DMEK surgery. As this was a preliminary study, the main aims of this study were to explore the surgical challenges associated with this novel technique and to report the intraoperative obstacles and immediate postoperative complications, such as graft dislocation and detachment. This clinical series also represents the first 30 cases of DMEK and, thus, the learning curve of DMEK surgery by an experienced DSAEK surgeon using the EndoGlide insertion device.
Methods
We evaluated this novel DMEK donor insertion technique in a prospective case series of patients with visually significant corneal edema due to endothelial dysfunction secondary to Fuchs dystrophy at the Singapore National Eye Center (SNEC), as part of the Singapore Corneal Transplant Registry, which is a prospective cohort study that tracks and records all clinical data, surgical complications, and outcomes in patients who undergo corneal transplants at the SNEC. This study was performed with approval from the local Institutional Review Board (CIRB Ref 2011/577/A), conforming to the tenets of the Declaration of Helsinki, and informed consent was obtained from all patients. Data including demographic details, ophthalmic history and postoperative visual acuity with endothelial cell (EC) counts were collected. Preoperative donor and postoperative EC counts were based on specular microscopy (Konan Keratoanalyzer EKA-98; Konan Medical Corp, Hyogo, Japan) performed by independent and experienced ophthalmic technicians. Main outcome measures were intraoperative complications; the incidence of postoperative complications such as graft dislocation or detachment and primary graft failures within 3 months from surgery, as previously defined; and postoperative endothelial cell counts.
Preparation of Prototype Descemet Mat
We manufactured the D-Mat using ultra-pure Poly-2 hydroxyethylmethacrylate (28% water content) approved for use in the manufacture of intraocular lenses. The D-Mat shapes were precision lathed to single-digit nanometer surface finish via Optoform 40 lathe (Precision Technology, Singapore, Singapore). The unique features include the subtle nonparallel curve with tangential peripheral topography to support handling, while still allowing the D-Mat to be rolled up sufficiently into the EndoGlide. The edge was designed as a blunt but smoothly polished form, to minimize the occurrence of edge tearing during handling (dimensions: base curve 8.50 mm, overall diameter 9.00 mm, central thickness 0.05 mm, edge thickness 0.10 mm). All hydration and extraction was conducted in distilled water baths using ultrasonic energy and surfactant to optimize surface cleanliness. The D-Mats were stored in physiological saline, buffers, and tonicity-modifying agents with 0.15% sodium hyaluronate and terminal sterilization was accomplished via steam-based autoclaving before use.
Donor Placement on Descemet Mat
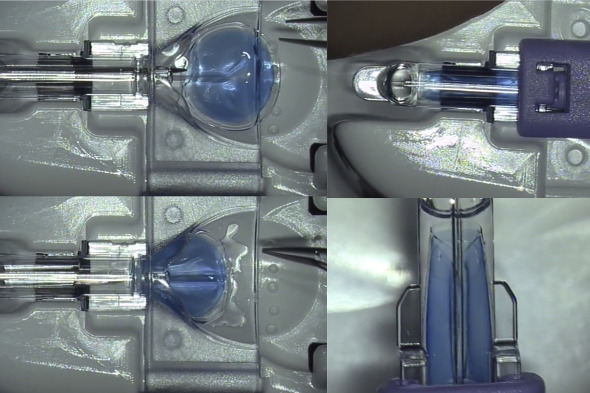
During the process of stripping the DMEK graft and trephining to a diameter of 8.0 mm, care was taken not to allow the DM to scroll on itself, and the DMEK graft was carefully laid back flat onto the donor stroma, endothelium up. Care was taken to ensure that the DMEK graft was well stained with trypan blue. In the presence of a shallow pool of BSS, a D-Mat was then carefully maneuvered beneath the DMEK graft, which would act as a scaffold. The DMEK graft was centralized over the D-Mat, and excess BSS was wicked away so that the graft was in full contact with the D-Mat surface. The larger diameter of the D-Mat also ensured a peripheral rim of the D-Mat, which allowed for handling of the D-Mat and not the DMEK graft, and the D-Mat prevented wrinkling or folding.
Recipient Preparation
A temporal scleral tunnel incision, measuring 4.5 mm wide by 1.5-2.0 mm deep, was made for EndoGlide Ultrathin insertion, in the same fashion as for DSAEK. Before the insertion, an AC maintainer was placed to prevent AC collapse during insertion, 1 clock hour apart from the main scleral wound. After scoring the DM over 360 degrees using a reversed Sinskey hook (Bausch and Lomb, Rochester, New York, USA), descemetorrhexis was performed with the reversed Sinskey hook under air, following an 8.0 mm marking on the corneal surface. A clear corneal paracentesis was made at the nasal limbus directly opposite to the temporal wound; this was for entry of the EndoGlide placement forceps (AngioTech/Network Medical Products).
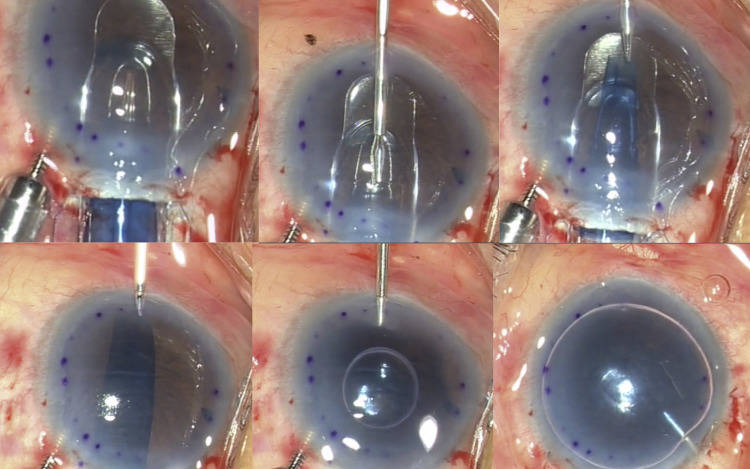
Placement of the Descemet Membrane Endothelial Keratoplasty Graft Into the EndoGlide Capsule
Grasping only the edge of the D-Mat (and not the DMEK graft), the D-Mat along with the DMEK graft were then transferred onto the donor well of the EndoGlide preparation base with endothelial surface facing upward, and a small amount of dispersive ophthalmic viscoelastic device (OVD) was applied on endothelial surface ( Figure 1 ). The EndoGlide loading forceps (AngioTech/Network Medical Products) was then introduced through the anterior opening of the EndoGlide capsule to grasp the leading edge of the D-Mat. The D-Mat and donor endothelium/DM complex was then slowly drawn into the glide capsule, forming a “double-coil” configuration as the edges of the D-Mat encountered the central internal ridge. The double coiling was facilitated by gentle upward strokes of both edges of the D-Mat with a BSS cannula. The D-Mat was drawn forward until the leading edge reached the anterior opening of the glide capsule. The EndoGlide introducer was then inserted into the capsule from behind and locked into place to seal the posterior opening of the capsule, to prevent leakage of BSS or aqueous humor from the EndoGlide during graft insertion. The entire EndoGlide complex was now ready for graft insertion into the AC with the double-coil configuration made by the D-Mat and graft.
EndoGlide Insertion
The EndoGlide was then removed from the preparation base and inverted the right way up for insertion into the AC. The anterior glide surface of the EndoGlide was inserted into the wound until the anterior opening of the capsule was fully within the AC ( Figure 2 ). During the insertion, the collapse of AC was prevented by a slow flow from the AC maintainer and the tight wound seal around the EndoGlide. With the EndoGlide in position, the EndoGlide forceps (AngioTech/Network Medical Products) was inserted through the nasal paracentesis into the AC and onto the glide surface of the EndoGlide ( Figure 2 ). The edge of the clear, transparent D-Mat was grasped and pulled partially into the AC; it was then released and then the blue-dyed DMEK graft was gently but firmly grasped and slowly pulled into the AC. The graft gradually uncoils in the AC in the anatomical position (endothelial surface down), and gentle shaking with the forceps helps achieve this. During this time, the AC maintainer remains on very low flow, so as to ensure a shallow chamber is maintained, but an assistant can vary AC maintainer flow as needed. While the graft was still held with the forceps, the EndoGlide was removed, and a small air bubble (usually 2-3 mm in diameter) was injected beneath the graft using a 30 G needle to float the graft against the recipient stromal surface and prevent descent and scrolling of the graft. The forceps was then released and the suturing of the main scleral wound, the AC maintainer site, and nasal paracentesis site was performed, prior to injecting a complete air fill for a full air tamponade. An inferior peripheral iridectomy was performed in all cases, prior to donor insertion, to prevent postoperative pupillary block.