Purpose
To investigate the prevalence and clinical significance of cystoid macular degeneration in eyes that underwent intravitreal ranibizumab injections for exudative age-related macular degeneration.
Design
Retrospective, interventional case series.
Methods
We reviewed the charts of 56 consecutive patients (19 male, 37 female; mean age ± standard deviation, 80.81 ± 4.8 years) with exudative age-related macular degeneration who received the last intravitreal ranibizumab injection at least 6 months before and were judged to have a fibroatrophic scar without signs of progression by fluorescein angiography or spectral-domain optical coherence tomography. Main outcome measures were the estimated prevalence and clinical significance of cystoid macular degeneration.
Results
Twenty-two eyes showed various combinations of degenerative pseudocysts, whereas 34 eyes did not show any pseudocysts. The 95% confidence interval for the prevalence estimate was 36.98% to 41.02%. Degenerative pseudocysts appeared square-shaped, did not change their overall appearance over time, and were located just below the internal limiting membrane in 11 eyes (50%), in the inner nuclear layer in 16 eyes (72.7%), in the outer nuclear layer in 8 eyes (36.3%), and in all the retinal layers in 6 eyes (27.2%). Best-corrected visual acuity improved in eyes with and without degenerative pseudocysts and decreased significantly in eyes with degenerative pseudocysts ( P = .03). Mean central macular thickness decreased significantly ( P < .001) to 324.1 μm and to 328.2 μm in eyes and without degenerative pseudocysts, respectively.
Conclusions
Cystoid macular degeneration represents a well-distinguished clinical entity that may be detected in exudative age-related macular degeneration eyes showing a posttreatment fibroatrophic scar and should not be considered as a manifestation of choroidal neovascularization activity.
Neovascular age-related macular degeneration (AMD) causes severe and irreversible vision loss if untreated, with loss of 3 lines of visual acuity by 1 year and 4 lines of visual acuity by 2 years. Ranibizumab (Lucentis; Genentech, Inc, South San Francisco, California, USA) is a recombinant, humanized, monoclonal antibody antigen-binding fragment that neutralizes all biologically active forms of vascular endothelial growth factor A. Intravitreal ranibizumab was the first therapy for exudative AMD to show, in 2 phase III clinical studies, an improvement in mean visual acuity. These results were obtained by a fixed-dosing regimen of ranibizumab 0.5 mg or 0.3 mg monthly injected over 24 and 12 months, respectively.
The Prospective OCT Imaging of Patients with Neovascular AMD Treated with intraOcular Ranibizumab study demonstrated a variable dosing regimen, mainly based on optical coherence tomography (OCT) findings, to be as effective as a fixed-dosing regimen in improving visual acuity and OCT findings with a far lower number of injections over 24 months. It is noteworthy that retreatment with ranibizumab (0.5 mg/0.05 mL) was performed if any qualitative increase in the amount of fluid was detected using OCT (during the second year of the study). This highlights how meaningful, to date, is the OCT evaluation of intraretinal or subretinal fluid accumulation in making the retreatment decision on an as-needed basis.
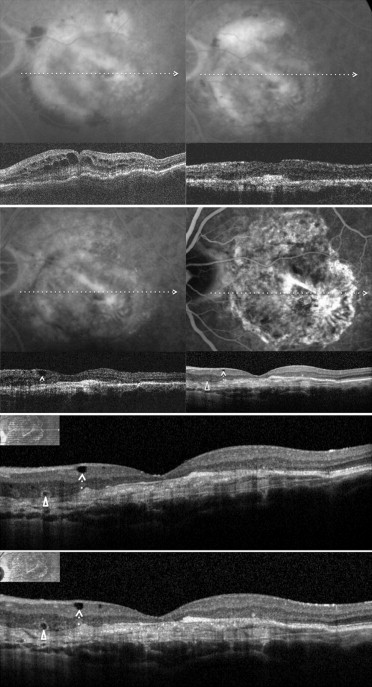
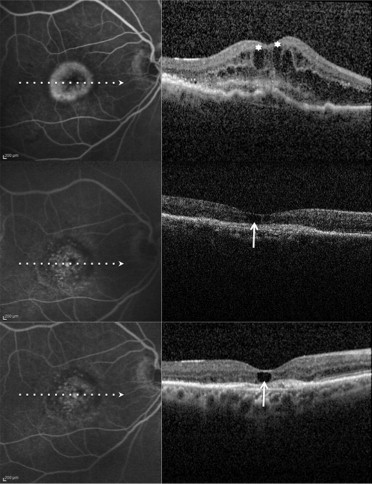
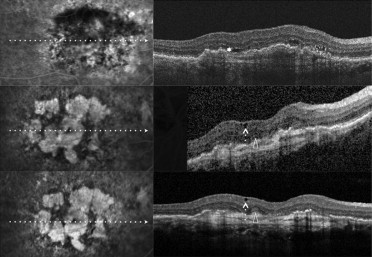
Recently, Cohen and associates, using spectral-domain (SD) OCT, reported on a characteristic and relatively frequent retinal finding of atrophic AMD that they called retinal pseudocysts . In their series, retinal pseudocysts appeared as optically empty spaces, frequently located in the inner nuclear layer. The authors proposed the term pseudocyst rather than the term cyst because these retinal lesions had no obvious wall. Interestingly, fluorescein angiography (FA), which was performed in 80% of eyes, showed no evidence of choroidal neovascularization (CNV), and there was no macular edema in any of the eyes with pseudocysts. They proposed that retinal pseudocysts may correspond to Müller cell degeneration (degenerative pseudocysts), and not to exudation (exudative cysts), and thus their presence should not require prompt treatment. Iida and associates recently proposed the term cystoid macular degeneration to describe a similar entity in central serous chorioretinopathy. Cystoid macular degeneration was characterized by intraretinal cystoid spaces (degenerative pseudocysts) with the absence of any intraretinal hyperfluorescence or leakage on FA. In the past, the term cystoid macular degeneration has been used to describe degeneration of the retina resulting in cystic spaces within the retina, or cystic degeneration (degenerative pseudocysts). It seems reasonable that degenerative pseudocysts (cystoid macular degeneration) may be detected not only in patients affected by atrophic AMD, but also in patients who were undergoing intravitreal ranibizumab injections for exudative AMD and who had a posttreatment fibroatrophic scar. Therefore, even in exudative AMD, degenerative pseudocysts (cystoid macular degeneration) should be recognized and distinguished from cystoid macular edema (CME), because their presence should not require prompt treatment.
Our aim was to investigate the prevalence and clinical significance of cystoid macular degeneration and the distribution of degenerative pseudocysts in the retinal layers of eyes that underwent last intravitreal ranibizumab injection for exudative AMD at least 6 months before and that were judged to have a fibroatrophic scar without signs of progression by FA or SD OCT.
Methods
We reviewed the charts of all consecutive patients with exudative AMD who underwent intravitreal ranibizumab injections at the University Eye Clinic of Creteil at least 24 months before and who did not receive any intravitreal ranibizumab injection within the previous 6 months. In case of bilateral involvement, only 1 eye was included randomly in the study.
Inclusion criteria were age 50 years or older; best-corrected visual acuity (BCVA), as evaluated by Early Treatment Diabetic Retinopathy Study (ETDRS) charts, of 20/400 or better at presentation; and the presence of subfoveal CNV secondary to AMD (occult CNV [type 1], classic CNV [type 2], and minimally classic CNV [type 1 plus type 2]), as evaluated by fundus biomicroscopy, FA, and indocyanine green angiography (ICGA), showing, at presentation, subretinal fluid, CME, or 1-mm central macular thickness (CMT) of at least 250 μm on OCT. Excluded were patients with fewer than 24 months follow-up, with previous treatment for exudative AMD in the study eye in the previous 6 months, previous surgery in the study eye in the previous 6 months, refractive error of more than −6 diopters, CNV attributable to causes other than AMD, or active intraocular inflammation in the study eye.
Each patient underwent a baseline assessment of BCVA measured at 4 m with standard ETDRS charts and an ophthalmic examination that included slit-lamp biomicroscopy, fundus biomicroscopy, FA, ICGA, and OCT (Stratus OCT, Carl Zeiss-Meditec, Dublin, California, USA; Cirrus HD-OCT, Carl Zeiss-Meditec; Spectralis HRA-OCT, Heidelberg Engineering, Heidelberg, Germany).
Initial treatment consisted of 1 intravitreal injection of 0.5 mg/0.05 mL ranibizumab, administered by retinal specialists according to the usual technique. Briefly, each eye was prepped with 5% povidone–iodine solution and draped, and a lid speculum was placed. Eyes were anesthetized with topical anesthetic, and ranibizumab was injected intravitreally through the inferotemporal pars plana 3.5 or 4 mm posterior to the limbus for pseudophakic or phakic patients, respectively. After the injection, patients were instructed to administer topical antibiotics for 4 days.
After the initial treatment, each patient underwent a detailed medical and ocular history monthly, as well as assessment of BCVA using ETDRS charts and ophthalmic examination, which included slit-lamp biomicroscopy, fundus biomicroscopy, OCT, and FA with or without ICGA. FA with or without ICGA was performed at the discretion of the examiner and not at every postinjection evaluation. However, as a rule, FA was performed in all patients when deciding to discontinue the treatment.
After the initial treatment, intravitreal ranibizumab injection was discontinued in eyes that showed absence of leakage from the CNV on FA, a dry macula (absence of subretinal fluid and CME) on OCT, or both. Eyes that showed a persistent leakage from the CNV on FA, fluid in the macula, or both received an additional injection 1 month after each previous injection. Fibrovascular pigment epithelium detachment without increase in size was not considered to be a sign of exudation.
Patients underwent repeat injections when there was a recurrence in CNV activity defined as either a decrease of BCVA (at least 1 ETDRS line) associated with persistent leakage from the CNV or 1 of the following features: (1) an increase of intraretinal fluid on OCT (increase in CMT by more than 100 μm); (2) a recurrence of subretinal fluid on OCT in a previously dry lesion; (3) a recurrence of CME on OCT in a previously dry lesion; or (4) new macular hemorrhage or new area of CNV. FA and ICGA results were evaluated together with OCT results to assess the accuracy of other clinical data, as well as in case of doubtful findings.
To assess the estimated prevalence and clinical significance of cystoid macular degeneration, we analyzed exudative AMD patients who received the last intravitreal ranibizumab injection at least 6 months before and were judged to be stable on the basis of BCVA (± 1 line/5 letters change), FA with or without ICGA (absence of leakage), and OCT (fibroatrophic scar without signs of progression). Multiple horizontal and vertical raster SD OCT scans were obtained by Spectralis SD OCT. In all these patients, SD OCT examinations were monthly or bimonthly and were performed after discontinuation of intravitreal ranibizumab injection for at least 6 months. The search for retinal degenerative pseudocysts was performed independently by 3 of the authors (G.Q., F.C., and E.H.S.) by analyzing recorded horizontal and vertical scans. In exudative AMD, as in atrophic AMD, there are different kinds of degenerative pseudocysts: unique versus multiple and superficial versus deep. However, despite these differences, all degenerative pseudocysts typically show a more-or-less square shape (at least 1 of their borders appears concave or straight). The same 3 authors also evaluated the location of degenerative pseudocysts: just below the internal limiting membrane, in the inner nuclear layer, in the outer nuclear layer, and in all the retinal layers. In rare cases when interpretation varied between the 3 authors, the SD OCT scans were reviewed as a group to reach consensus.
Also, we assessed the clinical significance of cystoid macular degeneration in terms of outcomes by comparing mean final BCVA and CMT, as well as mean final number of injections between eyes judged to be stable with cystoid macular degeneration and eyes judged to be stable without cystoid macular degeneration. Finally, we investigated the estimated prevalence of outer retinal tubulation (ORT) in eyes with cystoid macular degeneration.
Statistical calculations were performed using the Statistical Package for Social Sciences (version 17.0; SPSS, Inc, Chicago, Illinois, USA). The prevalence of cystoid macular degeneration was estimated by dividing the number of stable eyes with cystoid macular degeneration by the total number of stable eyes. Also, the prevalence of ORT was estimated by dividing the number of stable eyes with ORT by the total number of stable eyes. Comparisons of mean BCVA (ETDRS letters), mean CMT, and mean number of injections were performed using the paired t test. The odds ratios (ORs) and 95% confidence intervals (CIs) were calculated using a logistic regression model to assess influence of cystoid macular degeneration on BCVA and CMT. The 95% CIs of the prevalence estimates was calculated. Correlation between development of ORT and final BCVA was obtained with the Spearman rank correlation. The chosen level of statistical significance was P < .05.
Results
A total of 56 eyes of 56 patients (19 male, 37 female; age range, 67 to 87 years; mean age ± standard deviation, 80.81 ± 4.8 years) who did not receive any intravitreal ranibizumab injection at least 6 months previously and who were judged to be stable on the basis of BCVA (± 1 line/5 letters change), FA with or without ICGA (absence of leakage), and OCT (fibroatrophic scar without signs of progression) were included in the study. Overall, the mean ± standard deviation follow-up period was 29.9 ± 4.7 months, and the mean ± standard deviation number of intravitreal injections was 11.6 ± 3.0. Thirty eyes had a type 1 CNV, 13 eyes had a type 2 CNV, and 13 eyes had a mixed type 1 plus type 2 CNV. All eyes showed presence of exudative cysts in the macular area before treatment.
Among the 56 eyes judged to be stable, 22 eyes (39.3%) showed various combinations of degenerative pseudocysts (just below the internal limiting membrane, in the inner nuclear layer, in the outer nuclear layer, and in all the retinal layers) on the different SD OCT analyzed scans ( Figures 1–4 ), whereas 34 eyes (60.7%) did not show any degenerative pseudocysts ( Table 1 ). The 95% CI for the prevalence estimate for cystoid macular degeneration in eyes judged to be stable was 36.98% to 41.02%.
| Cystoid Macular Degeneration | No Cystoid Macular Degeneration | P Value (between the 2 Groups) | |
|---|---|---|---|
| No. | 22 | 34 | |
| Age (yrs) | 80.81 ± 4.8 ( range, 67 to 87) | 81.29 ± 3.9 ( range, 72 to 88) | .5 |
| BCVA (ETDRS letters) | 43.86 ± 8. | 43.08 ± 7.1 | .5 |
| Duration of macular thickness (mos) | 1.3 ± 0.6 | 1.2 ± 0.8 | .09 |
| CMT (μm) | 437.81 ± 69.3 | 442.5 ± 53.9 | .7 |
| CNV | .5 | ||
| Type 1 | 12 | 18 | |
| Type 2 | 5 | 8 | |
| Type 1 + 2 | 5 | 8 | |
| ORT (%) | 15 (68.2) | 22 (64.7) | .3 |
| Follow-up (mos) | 29.86 ± 5.2 | 29.92 ± 4.4 | .8 |
| Injections (no.) | 11.63 ± 3.3 | 11.58 ± 2.8 | .7 |
| Time to last treatment (mos) | 9.00 ± 2.4 | 9.08 ± 2.0 | .8 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


