Fig. 13.1
Design of the operation and intraoperative photographs. Group 2-I (a), group 2-II (b), and group 2-III (c)
13.4.3.2 Second Experiment [27]
A bone flap was created in the same manner. Dogs were divided into three groups. In group 2-I, the bone defect was closed by replacing the original free bone flap without filling the residual gap (Fig. 13.1a). In group 2-II, the gap was filled with the bone substitute material (BSM): collagen-coated b-TCP combined with autologous bone fragments (Fig. 13.1b). In group 2-III, autologous BSCs combined with BSM were used to fill the gap (Fig. 13.1c). The effectiveness of treatment was evaluated 3 and 6 months after operation with 3D-CT and histologic examinations of the operative regions. Bone regeneration was observed in groups 2-II and 2-III, with more extensive formation in group 2-III than in group 2-II (Fig. 13.2c–f). In group 2-I, no bone regeneration was observed (Fig. 13.2a, b). Moreover, in group 2-III at 6 months, new bone was observed connected to the existing cranial bone (Fig. 13.2f). This study showed that BSCs have the potential to promote cranial bone regeneration and confirmed the efficacy of a composite scaffold made of b-TCP combined with autologous bone fragments with fibrin glue.
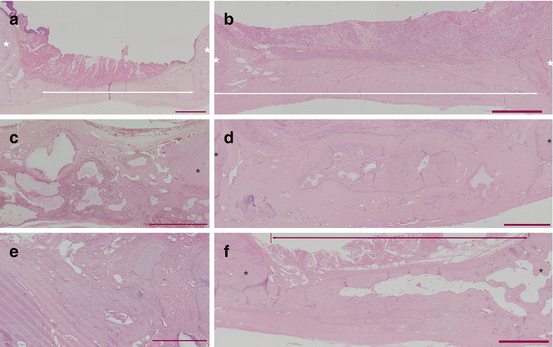

Fig. 13.2
Histology of group 2-I (a, b), 2-II (c, d), and 2-III (e, f). Three-month model (a, c, e). Six-month model (b, d, f). Red bars =1 mm. * original bone
13.4.3.3 Third Experiment [28]
Four experimental models were tested with or without BSM plus BSCs or CA membrane. In group 3-I, the original free bone flap was replaced in the defect (Fig. 13.3a). In group 3-II, after replacing the bone flap, the defect was covered with CA membrane (Fig. 13.3b). In group 3-III, BSM plus BSCs were used as a gap filler (Fig. 13.3c). In group 3-IV, BSM plus BSCs and CA membrane were applied (Fig. 13.3d). Histological examinations were performed 3 and 6 months after the operation. In groups I and II, no bone regeneration was observed but fibrous tissue intrusion was prevented in group II. More bone neogenesis was observed in group III than in group IV at 3 months (P < 0.05) (Fig. 13.4a–d). At 6 months, the regenerated areas were larger than those observed at 3 months, but the differences between groups III and IV were not statistically significant (Fig. 13.4e–g). This experiment showed that, as expected, CA membrane prevents excessive fibrous tissue intrusion and/or dislocation of a bone scaffold. However, use of a CA membrane did not always accelerate cranial bone regeneration. In addition, we found that there were other disadvantages associated with the use of a barrier membrane in cranial bone regeneration.
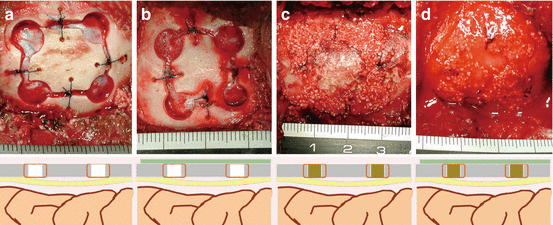
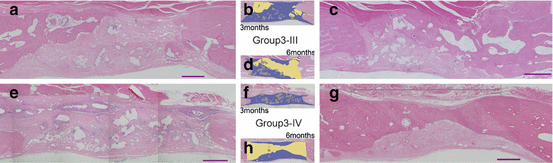

Fig. 13.3
Design of the operation, and intraoperative photographs. Group 3-I (a), group 3-II (b), group 3-III (c), and group 3-IV (d)

Fig. 13.4
Histology of group 3-III (a–d), and group 3-IV (e–h). Three-month model (a, b, e, f). Six-month model (c, d, g, h). The findings for the drilled hole for each model (a, c, e, g). Red bars =1 mm. Corresponding to figures, respectively (b, d, f, h). Yellow areas new bone, gray areas b-TCP, blue areas fibrous tissue, star original bone
Accordingly, even though the barrier membrane acted as intended, levels of important bone regeneration regulatory factors within the barrier might be inadequate. In other words, the supplementary supply of vessels or regulatory factors from the superficial structures may be indispensable in cranial bone regeneration. If other GBR membranes are developed for cranial bone regeneration, these should be chosen to have the ability both to block fibrous tissue intrusion from the superficial layer and to be permeable to regulatory factors and vascularization.
13.4.4 Future Prospects
Collagen-coated b-TCP is a suitable scaffold material for cranial bone regeneration, as its structure needs to be maintained in position until bone regeneration is completed. To improve cranial bone regeneration, it is necessary to prevent fibrous tissue intrusion, while the supplementary supply of vessels or regulatory factors from the superficial structures needs to be maintained. Therefore, a new type of barrier membrane that can block fibrous tissue intrusion without blocking the supply of vessels or regulatory factors should be developed, or other approaches will need to be explored. Further, providing a generous amount of scaffold may compensate for the volume loss of regenerated bone. Changing the shape of the scaffold from a granule type to a block type may solve this problem, but a new problem then arises because it is very difficult to coat collagen into deeply positioned interconnected pores. The scaffolds that can provide suitable environments for cells are indispensable to cell survival and good regeneration. Therefore, further investigation is required with regard to cranial bone regeneration, including better scaffolds and suitable environments.
References
1.
Lew D, Farrell B, Bardach J, Keller J. Repair of craniofacial defects with hydroxyapatite cement. J Oral Maxillofac Surg. 1997;55:1441–51. doi:10.1016/S0278-2391(97)90647-9.CrossRefPubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


