Compressive Optic Neuropathies
Compressive Optic Neuropathies With Optic Disc Swelling (Anterior Compressive Optic Neuropathies)
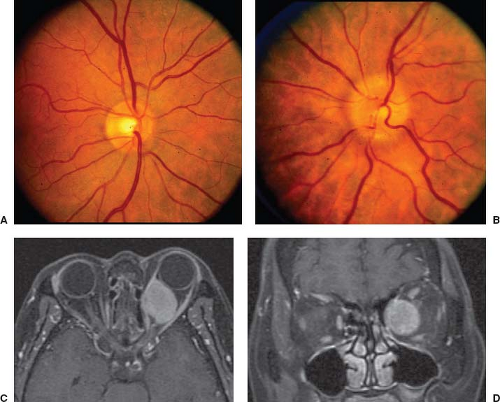
Lesions within the orbit, occasionally within the optic canal, and extremely rarely intracranially, may compress the optic nerve, resulting in optic disc swelling (Fig. 8.1). Such lesions include tumors, infections, inflammations, and even adnexal structures that have become swollen or enlarged by disease. Specific orbital disorders include optic gliomas, meningiomas, hamartomas (e.g., hemangiomas, lymphangiomas), choristomas (e.g., dermoid cysts), and other malignancies (e.g., carcinoma, lymphoma, sarcoma, multiple myeloma), as well as inflammatory orbital pseudotumor and thyroid ophthalmopathy.
In most cases of anterior compressive optic neuropathy, there is progressive visual loss associated with proptosis; however, in many patients, visual acuity remains normal or near normal, and there is almost no external evidence of orbital disease despite obvious disc swelling. This clinical picture occurs particularly in patients with orbital hemangiomas adjacent to the optic nerve and in patients with primary optic nerve sheath meningiomas. In such patients, careful testing of color vision may reveal subtle defects, and there may occasionally be a relative afferent pupillary defect despite retention of normal acuity. The visual field of the affected eye generally shows only enlargement of the blind spot or mild paracentral changes on automated perimetry. When other signs of orbital disease are not present (e.g., proptosis, limitation of ocular motility, orbital congestion), these patients may be thought to have unilateral papilledema from increased intracranial pressure. In such cases the workup may include MRI and MRV and potentially a lumbar puncture with opening pressure measurement. However, it should be noted that orbital disease is the most common cause of unilateral disc swelling without visual loss; hence, in such cases, in particular if there is an absence of signs or symptoms of raised ICP, orbital imaging is essential.
In addition to classical orbital signs, patients with orbital disease may develop various folds or striae that occur at the posterior pole, adjacent to the optic disc (Fig. 8.2). These folds may be horizontal, vertical, or oblique and tend to be coarser than those observed with intracranial pressure syndromes. Transient monocular visual loss can occur in patients with orbital lesions. The visual loss usually occurs only in certain positions of gaze, and vision immediately clears when the direction of gaze is changed. It is assumed that either direct pressure on the optic nerve or interruption of blood supply is responsible for this phenomenon.
Computed tomographic (CT) scanning and magnetic resonance imaging (MRI) offer superb topographic depiction (size, shape, and location) of lesions, and standardized echography provides supplementary information that often helps in refinement of the differential diagnosis. CT scanning is particularly useful for imaging bone, calcium, and metallic foreign bodies (suspicion of the latter being a contraindication for the use of MRI), whereas MRI excels at defining inflammatory and intrinsic disease of the visual pathway and parasellar area. MRI is particularly suited to imaging the intracanalicular optic nerve. With the use of fat-saturation techniques and gadolinium, demarcation of optic nerve sheath meningiomas can be optimized, and better differentiated from infiltrative disorders of the optic nerve.
When inflammatory conditions involve the orbit, such as “idiopathic orbital inflammation (orbital pseudotumor)”
the optic nerve may be compressed with secondary disc swelling. Affected patients usually experience visual loss, pain, proptosis, and congestion, and the presence of an orbital process is rarely in question. Infrequently, meningiomas of the orbital apex produce a similar clinical picture.
the optic nerve may be compressed with secondary disc swelling. Affected patients usually experience visual loss, pain, proptosis, and congestion, and the presence of an orbital process is rarely in question. Infrequently, meningiomas of the orbital apex produce a similar clinical picture.
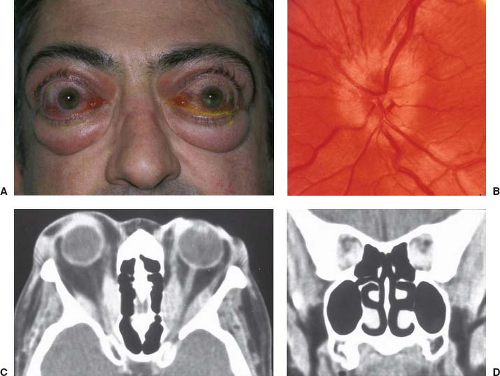
Patients with thyroid eye disease may develop evidence of a compressive optic neuropathy associated with optic disc swelling (Fig. 8.2). In such patients, congestive symptoms almost always precede visual loss resulting from the compression of the optic nerve at the orbital apex (see Fig. 8.2). Presenting visual acuities are typically 20/60 or worse, with central scotomas often combined with arcuate defects. Treatment options for thyroid-associated compressive optic neuropathy include intravenous or oral steroids and orbital decompression. Radiation therapy may also be effective, but its effects are often delayed and therefore more acutely effective treatments with steroids or surgery should be given in conjunction with radiation.
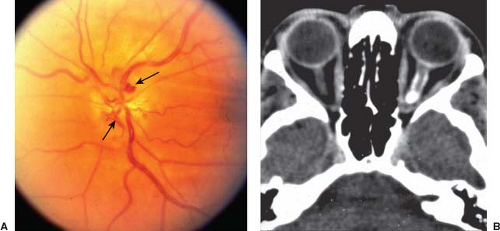
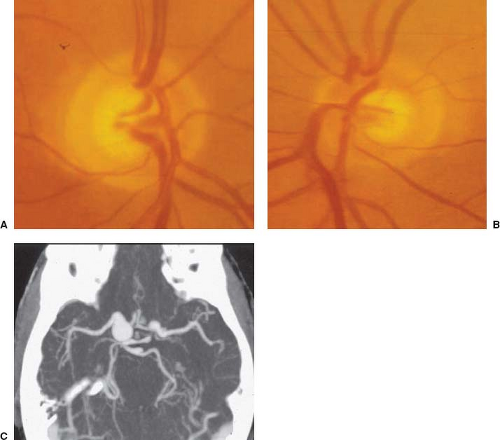
Anterior compressive optic neuropathy may result from primary optic nerve sheath meningioma (ONSM). Optic nerve sheath meningiomas surround the optic nerve and result in impaired axonal transport (leading to disc swelling) and also interfere with the pial blood supply to the optic nerve. The tumor occurs most commonly in middle-aged women and is usually unilateral. Patients present with slowly progressive vision loss, mild proptosis, double vision, transient visual obscurations, and gaze-evoked amaurosis. Examination reveals a color vision defect, afferent pupillary defect, and visual field defects including central scotomas, enlarged blind spots, and generalized constriction. The optic disc may be pale or swollen, and retinochoroidal shunt vessels may be present (Fig. 8.3; see Chapter 4). These tumors may expand rapidly during pregnancy producing a much more dramatic clinical picture in this setting.
Neuroimaging demonstrates focal or diffuse, tubular or fusiform, optic nerve enlargement (Fig. 8.3).
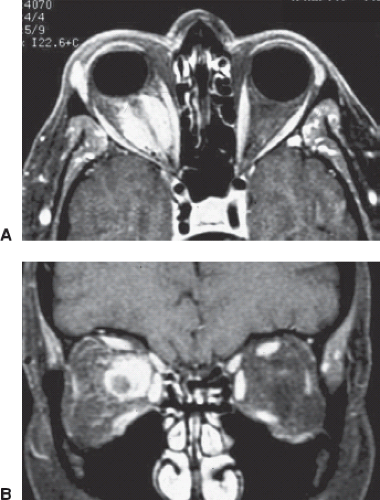
MRI is often diagnostic, showing the tumor to be separated from the optic nerve proper, allowing distinction from gliomas (Fig. 8.4). MRI is particularly good at imaging the intracanalicular optic nerve, and delineating the extent of intracranial extension of the ONSM. On CT, calcification can be seen in one-third of patients as linear bright lines extending over the length of the optic nerve (tram-track sign) (Fig. 8.3). Clinical findings and neuroimaging generally establish the diagnosis and biopsy is almost never needed. In the rare equivocal case, biopsy is only recommended in an eye with very poor vision, after a thorough systemic workup including LP (often serial), empiric treatment including steroids with failure to improve vision, and threat to the contralateral optic nerve via the chiasm.
MRI is often diagnostic, showing the tumor to be separated from the optic nerve proper, allowing distinction from gliomas (Fig. 8.4). MRI is particularly good at imaging the intracanalicular optic nerve, and delineating the extent of intracranial extension of the ONSM. On CT, calcification can be seen in one-third of patients as linear bright lines extending over the length of the optic nerve (tram-track sign) (Fig. 8.3). Clinical findings and neuroimaging generally establish the diagnosis and biopsy is almost never needed. In the rare equivocal case, biopsy is only recommended in an eye with very poor vision, after a thorough systemic workup including LP (often serial), empiric treatment including steroids with failure to improve vision, and threat to the contralateral optic nerve via the chiasm.
Natural history data suggest that ONSMs can remain stable or improve over several years. If progression is documented, radiation therapy is the best treatment available. Newer partially fractionated stereotactic delivery protocols may slowly replace conventional fractionated conformal external beam radiotherapy. Surgical excision of these lesions to improve vision is usually not possible because of the tumor involvement of the pial blood supply to the optic nerve. Portions of the tumor are occasionally removed to treat blind or uncomfortable eyes (proptosis), to decompress associated mass effect, or reduce the risk of intracranial extension.
Patients with meningiomas confined entirely to the optic canal may occasionally present with blurred vision and optic disc swelling. The mechanism by which disc swelling occurs in such cases is not clear, but it presumably results from direct compression of the optic nerve with blockage of axonal transport. It is very rare to see optic disc swelling from compression of the intracranial portion of the optic nerve.
Intracranial lesions, particularly sphenoid wing meningiomas, not infrequently extend through the
optic canal, compressing the intracranial, intracanalicular, and intraorbital optic nerve in the process. Optic disc changes, from swelling to pallor, may be minimal in such cases even when the lesion is quite large.
optic canal, compressing the intracranial, intracanalicular, and intraorbital optic nerve in the process. Optic disc changes, from swelling to pallor, may be minimal in such cases even when the lesion is quite large.
Compressive Optic Neuropathies Without Optic Disc Swelling (Retrobulbar Compressive Optic Neuropathies)
The importance of early diagnosis of compressive lesions that affect the retrobulbar portions of the optic nerve and do not cause optic disc swelling cannot be overemphasized. Early decompression of the optic nerves or chiasm may result in significant return of visual function, whereas delayed diagnosis may result in progressive visual failure and irreversible visual loss, neurologic dysfunction, or death. Intracranial, intracanalicular, and occasionally posterior orbital compressive lesions usually do not produce disc swelling or significant neurologic or systemic manifestations. Thus, by the time such lesions cause visible optic pallor, significant damage to the optic nerve has often already occurred, preventing return of visual function even with otherwise successful decompression. The physician managing a patient with unexplained unilateral visual loss must be aware of the characteristic history and early findings in patients with a retrobulbar compressive optic neuropathy.
In cases of prechiasmal optic nerve compression, progressive, painless dimming of vision is noted at an early stage. Although these patients may read the 20/25 or even 20/20 Snellen letters with the affected eye, they do so slowly and with great difficulty compared
with the ease with which they read the 20/15 line with the opposite eye. There is usually some type of visual field defect, particularly when tested with automated perimetry, but the nature of the defect does not in isolation suggest the etiology of the visual loss. Compression of the optic nerve may produce any type of field defect, including an altitudinal, arcuate, hemianopic, central, or cecocentral scotoma. Although a hemianopic field defect or a “junctional” scotoma in the superior temporal field of the opposite eye strongly suggests a compressive lesion, it is the insidious, progressive nature of the symptoms that is most often the critical feature of the compressive process.
with the ease with which they read the 20/15 line with the opposite eye. There is usually some type of visual field defect, particularly when tested with automated perimetry, but the nature of the defect does not in isolation suggest the etiology of the visual loss. Compression of the optic nerve may produce any type of field defect, including an altitudinal, arcuate, hemianopic, central, or cecocentral scotoma. Although a hemianopic field defect or a “junctional” scotoma in the superior temporal field of the opposite eye strongly suggests a compressive lesion, it is the insidious, progressive nature of the symptoms that is most often the critical feature of the compressive process.
Two important abnormalities that are nearly always present in a patient with retrobulbar compressive optic neuropathy are unilateral dyschromatopsia and an ipsilateral relative afferent pupillary defect. The afferent defect is usually obvious even when visual acuity is minimally reduced and provides the observer with absolute evidence that the visual difficulty is caused by something other than a simple refractive error, incipient cataract, or focal macular disease. Once a relative afferent pupillary defect is detected in a patient with an apparently normal fundus who is complaining of progressive dimming of vision, compression must be ruled out by neuroimaging, preferably gadolinium-enhanced MRI. The evaluating physician should have a low threshold for imaging in such cases, alerting the radiologist as precisely as possible about the region of interest.
The optic disc of a patient with a compressive optic neuropathy may appear normal or show a variable degree of pallor. Asymmetric cupping of the optic disc is not usually a prominent feature, but it may occur and be quite prominent in some patients (Fig. 8.5). In such cases, the visual loss may be thought to have resulted from glaucoma, particularly when there is an arcuate defect, but pallor of the neuroretinal rim indicates the nonglaucomatous nature of the lesion. Optic atrophy in one eye and disc swelling in the other may
result from asymmetric optic nerve compression or from tumor-induced direct optic nerve compression in one eye and papilledema from increased intracranial pressure in the other (Foster Kennedy syndrome) (see Chapter 4).
result from asymmetric optic nerve compression or from tumor-induced direct optic nerve compression in one eye and papilledema from increased intracranial pressure in the other (Foster Kennedy syndrome) (see Chapter 4).
Causes of retrobulbar optic nerve compression include intraorbital and intracranial benign and malignant tumors (most commonly pituitary tumors, meningiomas, and craniopharyngiomas); aneurysms (Fig. 8.5); inflammatory lesions (particularly of the paranasal sinuses); primary bone disease (e.g., osteopetrosis, fibrous dysplasia, craniometaphyseal dysplasia, Paget disease); orbital fractures; dolichoectatic intracranial vessels; congenital and acquired hydrocephalus; thyroid eye disease; and orbital hemorrhage. Meningiomas arising from the planum sphenoidale or olfactory groove, pituitary tumors, and aneurysms are the most commonly identified lesions causing retrobulbar compressive optic neuropathy without disc swelling.
The sudden onset of unilateral visual loss, combined with signs of retrobulbar optic nerve dysfunction, usually suggests a diagnosis of optic neuritis or ischemic optic neuropathy. In rare instances, however, compressive lesions produce acute monocular visual loss, presumably from hemorrhage or from sudden interruption of the vascular supply to the optic nerve. This phenomenon probably occurs most frequently in patients with pituitary apoplexy, but it has also been reported in patients with pituitary tumors and meningiomas during pregnancy and in otherwise healthy patients upon awakening. Sudden onset of monocular visual loss can also occur from a ruptured ophthalmic artery aneurysm, fibrous dysplasia, orbital hemorrhage, subperiosteal abscess, orbital cellulites, and mucocele (particularly of the sphenoid sinus).
Visual Recovery Following Decompression
Restoration of visual function is not only possible following surgical decompression, but it can begin within hours to days after surgery, sometimes with full recovery. “Medical decompression” of the optic nerve with bromocriptine in patients with prolactin-secreting pituitary tumors has yielded similar improvements in visual function. Although it is not surprising that prompt decompression may restore visual function in patients rendered suddenly blind by pituitary apoplexy, it is not intuitively obvious that patients who have lost all light perception for days or weeks can occasionally experience dramatic visual recovery following decompression surgery. There is no correlation between the rapidity of visual loss and the rapidity of visual return. Recent studies using optical coherence tomography have demonstrated utility in the use of this technology in terms of visual prognosis following decompression; preoperative RNFL measurements of over 80 microns portend a better prognosis and may be more helpful than features such as degree of optic atrophy, depth of visual field defect, or color vision or acuity measurements.
It has been theorized that there are three stages of visual recovery after decompression of the anterior visual pathway:
Relief of visual pathway compression is initially followed by rapid recovery of some vision within minutes to hours. This recovery can be likened to the relief of conduction block after an arm or leg “goes to sleep.”
This initial recovery is followed by delayed recovery of additional function over weeks to months. This improvement may be related to progressive remyelination of previously compressed demyelinated axons.
Finally, there is an even longer period of improvement, taking many months to years. The mechanism by which this late recovery occurs is unknown.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree