Fig. 29.1
Low diffuse bleb with large surface area is the goal of glaucoma surgery
In this chapter we will review the practical aspects for preventing and managing the complications after glaucoma surgery in general and, specifically, of filtration surgery.
Hypotony and Overfiltration
Hypotony can be caused by excessive aqueous outflow (related to excessive filtration, wound leak, or cyclodialysis cleft) and/or to reduced aqueous production (due to ciliochoroidal detachment, inflammation, inadvertent use of aqueous suppressants, or extensive cyclodestruction). Transient hypotony is very common after glaucoma surgery, but it may lead to other possible complications including flat anterior chamber (Fig. 29.2), corneal edema, Descemet’s membrane folds (Fig. 29.3), choroidal effusions (Fig. 29.4), suprachoroidal hemorrhage, visual loss, cataract, macular and optic disc edema, and chorioretinal folds (predominantly in young myopic patients) [1, 2, 4].
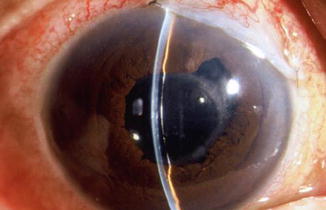
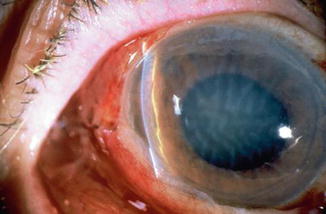
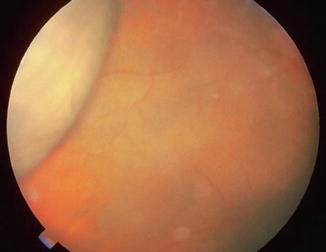

Fig. 29.2
Shallow chamber after trabeculectomy. Note that there is contact between the cornea and the iris but no lens–cornea touch within the pupillary area

Fig. 29.3
Corneal edema and Descemet’s folds are seen in this hypotonous eye

Fig. 29.4
Serous choroidal detachment noted after a trabeculectomy when the IOP was initially 43 mmHg and fell to 12 mmHg. The chamber shallowed and the choroidal detachment was observed in the periphery
The initial management of early postoperative hypotony with a formed anterior chamber is conservative with topical steroids and cycloplegics. Restrictions in activity (bending, weight lifting) and avoidance of Valsalva-positive conditions are recommended (Fig. 29.5). Intervention is indicated in cases when hypotony is associated with other complications such as persistent low intraocular pressure (IOP) with loss of visual acuity and hypotony maculopathy (see below). Treatment should be aimed at correcting the specific cause of hypotony. When there is a flat anterior chamber with lens–corneal touch, immediate surgical intervention is necessary to prevent endothelial damage and cataract formation. Reformation of the anterior chamber with viscoelastic can be done at the slit lamp or under the operating microscope through the paracentesis made intraoperatively. When there are large appositional choroidal effusions and the bleb is threatened, drainage of the fluid is recommended [4].
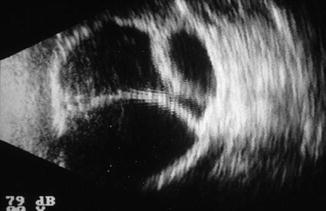

Fig. 29.5
B-scan of large kissing choroidals (serous choroidal effusions)
Most commonly hypotony is due to overfiltration of a filtering bleb. Several treatment options are available. Localized compression of the filtration site with a large bandage contact lens or and Simmons’ shell may be helpful [5, 6]. Several treatments have been used to induce an inflammatory or healing reaction in the filtering bleb which modifies the morphology of the filtering blebs and increase the IOP. These procedures include topical application of chemicals to the bleb surface, cryotherapy, or thermal lasers but are not very popular [7, 8]. Injection of autologous blood into the bleb can reduce overfiltration and resolve bleb leaks [9–11]. Finally, surgical revision is the most efficacious option. Resuturing the scleral flap (occasionally associated with scleral patch grafting when resuturing is not possible) is the most successful option [12–14]. Transconjunctival suturing of the sclera flap is relatively simple and effective [15, 16].
Hypotony Maculopathy
Some patients with intraocular hypotony develop loss of central vision secondary to marked irregular folding of the choroid and retina. The incidence of hypotony maculopathy after glaucoma surgery has increased with the use of antifibrotic agents, specifically mitomycin-C. The maculopathy is most likely to occur in young myopic patients, who may have a sclera more susceptible to swelling and contraction [17–19].
The retina often shows a series of stellate folds around the center of the fovea (Figs. 29.6 and 29.7). The retinal vessels are tortuous and sometimes engorged. There may be swelling of the peripapillary choroid simulating papilledema. Visual loss is due to the marked folding of the central retina. Early detection of this condition is key, since prompt correction of the cause will usually result in visual improvement. In the Tube Versus Trabeculectomy (TVT) study hypotony maculopathy appeared in 5 % cases after trabeculectomy and in 1 % after tube surgery [20].
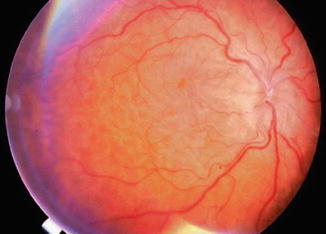
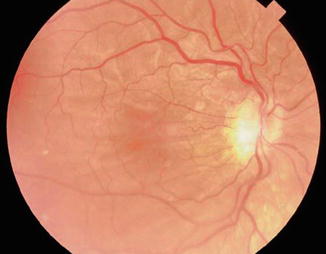

Fig. 29.6
Disc edema with hypotony maculopathy. The border of the optic disc is swollen due to the low intraocular pressure and disruption of axoplasmic flow. The vessels are tortuous and there are chorioretinal folds, with edema

Fig. 29.7
Chorioretinal folds are often noted in hypotonous eyes of long duration. After the IOP is normalized, the folds often regress and vision can return to normal
To prevent postoperative hypotony, intraoperative and postoperative control of outflow drainage is essential. During a glaucoma filtration procedure, the surgeon will try to provide sufficient outflow of aqueous thereby lowering the intraocular pressure (IOP) but avoiding complications, specifically hypotony, flat anterior chamber, choroidal detachment, and pressure spikes. Repeated injections of BSS through the paracentesis or use of an anterior chamber maintainer is recommended to test the outflow after initial suturing of the scleral flap. The flow can then be adjusted as needed [4].
The possibility of removing or cutting a scleral flap suture allows the surgeon to tightly close the scleral flap intraoperatively to decrease the probability of developing profound hypotony and a flat anterior chamber, especially if mitomycin-C has been used. Permanent and/or releasable scleral flap sutures can be cut or adjusted in the postoperative period to enhance the outflow (Figs. 29.8 and 29.9) [21–25].
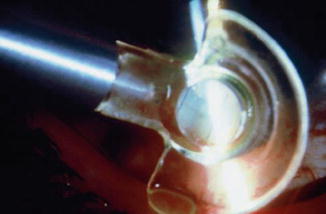
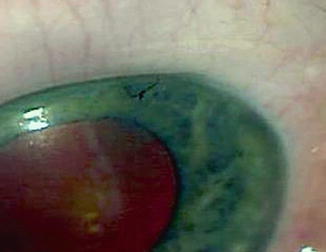

Fig. 29.8
Laser suture lysis with the Hoskins lens

Fig. 29.9
Releasable suture of 10-0 nylon near the limbus helps control flow in the postoperative period. These can be easily removed at the slit lamp
Releasable sutures to close the scleral flap have some practical advantages as the externalized suture is easily removed, effective in cases of hemorrhagic or thickened bleb (that would make difficult suture lysis) and do not require laser equipment [26–28]. There are some potential disadvantages of releasable sutures, including additional operating time, potential discomfort if they are not buried, and ocular infection. If antimetabolites are to be used, there may be a risk of an aqueous leak around the suture site especially if there is a limbal closure (fornix-based flap). The timing for cutting/releasing sutures is important. If antimetabolites are not used, it should be done within the first 2 weeks. Later, fibrosis of the scleral flap may negate any potential increase in outflow. The window of opportunity is expanded when antimetabolites have been associated with the surgery, although the response decreases with a longer interval to suture release [29].
Suture release or lysis should be performed in a conservative stepwise manner. Usually only one suture is released at a time to avoid overfiltration.
Postoperative Suprachoroidal Hemorrhage
Suprachoroidal hemorrhage after surgery is rare but can be devastating. Risk factors for suprachoroidal hemorrhage include glaucoma, aphakia, previous vitrectomy, myopia and postoperative hypotony. Systemic risk factors are arteriosclerosis, high blood pressure, tachycardia, bleeding disorders, and anticoagulant therapy [30–33]. In the TVT study [20], 2 % of patients and 3 % of patients developed suprachoroidal hemorrhage after tube and trabeculectomy surgery, respectively.
Postoperative suprachoroidal hemorrhage usually occurs within the first week after glaucoma surgery and is usually associated with hypotony. The development of a suprachoroidal hemorrhage is typically acute and associated with the sudden onset of severe pain. Often this follows exertion with a Valsalva maneuver. Examination of the anterior segment frequently reveals a shallow anterior chamber (Fig. 29.10) and either a low, normal, or high intraocular pressure depending on the extent of the hemorrhage. On fundus exam a detached and dark choroid is noted. Some cases present with bleeding into the vitreous cavity. Bleeding into the vitreous cavity at the time of the hemorrhage and retinal detachment greatly worsen the visual prognosis.
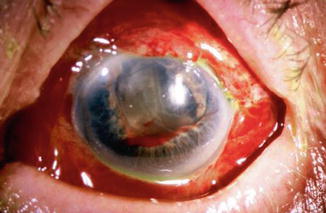

Fig. 29.10
Delayed suprachoroidal hemorrhage in a patient on chronic antiplatelet therapy
Treatment of postoperative suprachoroidal hemorrhage is directed toward control of the IOP and relief of pain. The majority of these eyes will do well with this conservative management, and surgical drainage is not usually necessary. The indications for drainage through a sclerotomy include intolerable pain, a persistent flat anterior chamber, failure of bleb filtration, and massive choroidal detachments. A waiting period of at least seven to ten days following a suprachoroidal hemorrhage is advised for the fibrinolytic response to liquefy the clot and allow for more effective evacuation of the suprachoroidal space.
Hyphema
In general, hyphema presents at surgery or within the first two or three days following surgery (Fig. 29.11). Hyphema commonly arises from the ciliary body or cut ends of the Schlemm’s canal, though it might also arise from the corneoscleral incision or iris. The probability of having postoperative hyphema is decreased during filtration surgery bleeding by performing the internal fistula as far anterior as possible (i.e., at the cornea). In the CIGTS study bleeding into the anterior chamber was the most common intraoperative complication (8 %) and one of the most common incidents after trabeculectomy (10 %) [34]. In the TVT [20] and AGIS [35] trials the frequency of hyphema was 8 % and 11.4 %, respectively.
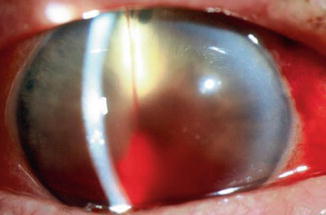

Fig. 29.11
Hyphema photographed in the development stage following filtration surgery. The source of the hyphema was the edge of the trabectomy flap
In the vast majority of cases, no treatment is necessary and the blood will be absorbed within a brief period of time. Cycloplegics, corticosteroids, restriction of activity, and elevation of the head of the bed 30°– 45° (to prevent blood from obstructing a superior sclerostomy) are recommended. Increased IOP can occur, particularly if the filtering site is obstructed by a blood clot, and it should be treated if necessary with aqueous suppressants. Surgical evacuation is considered depending on the level of IOP, size of hyphema, severity of optic nerve damage, likelihood of corneal blood staining, and presence of sickle trait or sickle cell anemia (infarction of the optic nerve can occur at relatively low IOP, and carbonic anhydrase inhibitors are contraindicated). Liquid blood can easily be removed with irrigation. If a clot has formed it can be removed by expression with viscoelastic or with a vitrectomy instrument set at low vacuum.
Malignant Glaucoma
Malignant glaucoma (also called aqueous misdirection) is characterized by a shallowing or flattening of the anterior chamber without pupillary block (i.e., in presence of a patent iridectomy) or choroidal pathology (such as suprachoroidal hemorrhage), commonly with an accompanying rise in IOP. It is more common after surgery for angle-closure glaucoma, in phakic, hyperopic (small) eyes. In the TVT study [20] it occurred in 3 % of patients after tube surgery and 1 % after trabeculectomy, while its incidence in CIGTS was 0.4 % over 5 years [34].
In this condition an increased pressure at the vitreous cavity displaces the lens forward, shallowing the anterior chamber (Fig. 29.12a, b). In malignant glaucoma a relative resistance to the anterior movement of fluid from the vitreous cavity to the anterior chamber probably occurs [36].
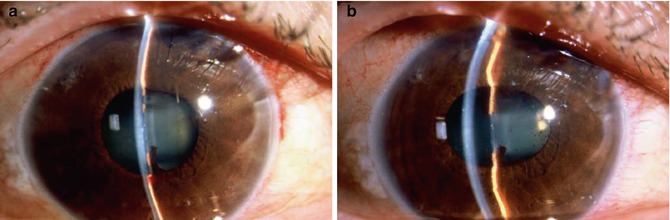

Fig. 29.12
Aqueous misdirection, historically called malignant glaucoma, is characterized by a flat chamber and increased IOP after pupillary block has been ruled out. (a) Appearance before treatment and (b) after atropine 1 % drops. Note the deepening of the anterior chamber with posterior movement of the lens–iris diaphragm after dilation
Aqueous misdirection usually occurs in the early postoperative period. The anterior chamber is shallow and the intraocular pressure is high. However, with a functioning filtration bleb, the intraocular pressure may not be high. The peripheral iridectomy is patent, and there are no chorioretinal abnormalities.
Malignant glaucoma is initially managed with mydriatic–cycloplegic drops, aqueous suppressants, and hyperosmotic agents. If the condition is relieved (e.g., the anterior chamber has deepened), the hyperosmotic agents are discontinued first, then the aqueous suppressants are reduced or even stopped over several days. Cycloplegic drops should be continued for weeks to months.
In pseudophakic eyes a peripheral hyaloidotomy with the Nd:YAG laser may be efficient and can often be accomplished through an existing peripheral iridectomy [37]. Oftentimes, breaking the hyaloid face with the Nd:YAG laser can be done at the edge of the optic through the posterior capsule. If not successful, zonulo–hyaloido–vitrectomy via the anterior segment has been successfully used in a series of pseudophakic patients [38].
Pars plana vitrectomy should be considered when other therapies fail [39, 40]. A standard 3-port pars plana vitrectomy, removing the anterior vitreous and part of the anterior hyaloid, is done. In phakic patients, the lens can sometimes be spared, but the probability of recurrence is higher. Pars plana tube-shunt insertion with vitrectomy has been recommended to treat patients with aqueous misdirection, especially in cases with angle-closure glaucoma. The implantation of the tube shunt through pars plana can help prevent recurrence of this condition and can help in the long-term control of IOP [40].
Prevention: In high-risk eyes undergoing cataract or filtration surgery, the decompression and shallowing of the anterior chamber should be minimized. Postoperative overfiltration should be avoided with a thick scleral flap sutured tighter and with more sutures than usual. Postoperatively, judicious suture lysis or cutting/pulling releasable sutures and slow tapering of cycloplegics are recommended. A postoperative shallow anterior chamber due to overfiltration should be vigorously treated.
Wipeout
The phenomenon of severe visual loss after surgery, with no obvious cause, is known as “wipeout” or “snuff syndrome” (Fig. 29.13). Wipeout may affect patients who have very severe glaucomatous damage and overall is a very uncommon complication but remains an important concern among glaucoma surgeons. Wipeout was probably more common after full-thickness filtration surgery [41]. However, it is not known whether patients who had been diagnosed with wipeout could have had another undetected cause such as macular edema, hypotonous maculopathy, or inflammation. With modern surgical techniques it is becoming increasingly rare [42–44].
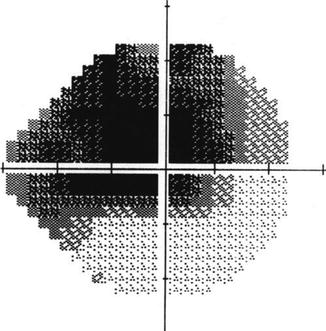

Fig. 29.13
Visual field test. Loss of the central visual field is a rare occurrence following glaucoma filtering surgery and is characterized by decreased central vision (wipeout) that cannot otherwise be explained
There are a few studies that have evaluated the incidence of wipeout after trabeculectomy. The reported incidence ranged from 0 to 0.95 % of surgeries. A retrospective study of 508 trabeculectomies identified four cases of wipeout, all of which had retrobulbar anesthesia [42]. Older patients with high preoperative IOP, advanced visual field defects affecting the central field, split fixation, and postoperative complications would appear to be at increased risk.
Possible Mechanisms and Prevention Strategies of Wipeout
(a)
Direct damage to the optic nerve from anesthetic technique: Local pressure to the optic nerve may result from a hematoma in the optic nerve sheath, a retrobulbar hematoma, or simply from the volume of anesthetic injected [43, 45], even with a low volume of local anesthetic (LA) if the LA were to become trapped between fascial layers to give a “compartment syndrome.” Additional indirect evidence of the possible effect of local anesthetic was noted in a small series of three cases of hyaluronidase-associated orbitopathy, in which the most severe and long-lasting visual loss occurred in the one patient who had glaucoma [46]. Doppler imaging studies have shown that retrobulbar injections can cause a marked reduction in blood flow in the arteries supplying the anterior optic nerve, particularly if epinephrine is included in the LA mixture [47]. This effect is not seen with anterior placement of local anesthetic, for example, by subconjunctival anesthesia [48].
(b)
Pressure spike: Early undiagnosed postoperative IOP spikes could potentially inflict further insult to a very severely damaged optic nerve with end-stage glaucoma. Thus, it seems logical to associate an early and severe pressure spike with the occurrence of wipeout. Pressure spikes may occur if the scleral flap has been sutured very tightly or the fistula is blocked by iris tissue or a blood clot. In patients with advanced glaucomatous damage undergoing trabeculectomy, IOP should be monitored a few hours after surgery and also the following day. Thus, if there is an early IOP spike, it can be treated accordingly.
(c)
Postoperative hypotony: Postoperative profound hypotony has been associated with “wipeout,” and it seems that this complication was more common when full-thickness filtration procedures were the standard surgeries for glaucoma. In the recent Tube Versus Trabeculectomy study (TVT), choroidal effusion was an independent risk factor for unexplained visual acuity loss after surgery [3]. Choroidal effusion is generally considered a relatively mild problem, but in the TVT study results suggested that this complication may not be always benign in nature [3].
Early Bleb Leak
An inadvertent buttonhole in the conjunctiva during a filtering procedure or a wound leak, through the conjunctival incision, can be responsible for an early leaking bleb. Early leaking can flatten the bleb and lead to subconjunctival-to-episcleral fibrosis, which would jeopardize a satisfactory long-term filtration. Bleb leaks are detected with the Seidel test (Fig. 29.14). The incidence of bleb leaks in the AGIS [35], CIGTS [34], and TVT [20] studies ranged from 6 to 11 % but was much higher in both arms of the FFSS study [49] (20–32 %).
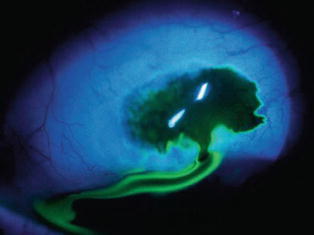

Fig. 29.14
A bleb leak at the limbus can occur early in the postoperative course either from poor wound healing or inadvertent trauma
The usual cause for conjunctival buttonholes is penetration of the tissue by the tip of a sharp instrument (needle, scissors, blade) or the teeth of a forceps. Buttonholes and tears are more likely to occur in cases with extensive conjunctival scarring. To diagnose a buttonhole intraoperatively, the conjunctiva should be carefully examined at the end of the procedure by filling the anterior chamber and raising the filtering bleb. If recognized, the buttonhole should be closed at the time of surgery. If it is located in the center of the conjunctival flap, a “purse string” closure is attempted either internally on the undersurface of the conjunctiva or externally in the flap that has already been reapproximated. A 10-0 or 11-0 nylon on a tapered (“vascular”) needle should be used. When the conjunctival buttonhole or tear occurs at the limbus, it can be sutured directly to the corneal–limbal junction [4].
Early bleb leaks usually heal spontaneously, but discontinuation of topical steroids may accelerate the spontaneous closure. Prophylactic antibiotics are indicated. If there are associated complications such as shallow or flat anterior chamber, active intervention is recommended. Our preferred therapeutic modalities to treat leaking blebs include bandage contact lens, fibrin tissue glue, and surgical revision (see above) [4, 50].
Late Bleb Leak
Spontaneous late bleb leaks are more frequent in avascular, thin blebs, which occur more frequently when antimetabolites are used in the filtering procedure (Fig. 29.15). Leakage of the filtering bleb can be associated with hypotony, shallow–flat anterior chamber, and choroidal detachment and may increase the chances for bleb infection and subsequent endophthalmitis [4].
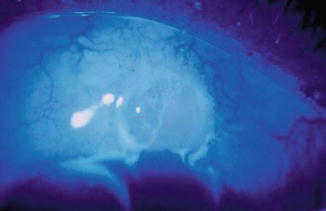

Fig. 29.15
Bleb leak, Seidel (+). Oftentimes, a wound leak may go undetected, especially late in the postoperative period. A Seidel test should be done in avascular, cystic blebs
Bleb leaks can heal spontaneously, and conservative management with prophylactic antibiotics is recommended if there are no associated problems. Patient education regarding symptoms of bleb-related ocular infection is crucial for prompt diagnosis and management. The need and timing for surgical repair of bleb leaks depend on the presence of other complications such as severe hypotony, flat anterior chamber, vision loss, or previous episodes of bleb-related infections [51–57].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


