Purpose
To clarify the efficacy of combined therapy with intravitreal ranibizumab injections and photodynamic therapy (PDT) in patients with symptomatic retinal angiomatous proliferation.
Design
Retrospective, interventional, consecutive case series.
Methods
We retrospectively reviewed 20 treatment-naïve eyes of 16 patients (8 men, 8 women; age range, 79 to 92 years; mean age, 84.8 years) treated with 3 consecutive monthly intravitreal injections of ranibizumab (0.5 mg/0.05 mL) and PDT and followed up for at least 12 months. PDT was applied 1 or 2 days after the initial injection. Retreatment was performed as a combined therapy of a single intravitreal ranibizumab injection and PDT.
Results
The mean best-corrected visual acuity (BCVA) levels significantly improved from 0.24 at baseline to 0.43 at 12 months ( P < .001). The mean improvement in BCVA at 12 months from baseline was 2.51 lines. The BCVA at 12 months improved in 10 eyes (improved by 3 lines or more) and was stable (defined as a loss of less than 3 lines of vision) in 10 eyes. No patient had a decrease in the BCVA of 3 lines or more during any 12 months. The central retinal thickness decreased significantly from 444 μm at baseline to 143 μm at 12 months ( P < .0001). Complete occlusion of the retinal–retinal anastomosis was achieved in 17 of the 19 eyes at 12 months. The mean numbers of PDT treatments and injections during 12 months, including the treatments in the initial regimen, were 1.8 and 3.8, respectively. No complications or systemic adverse events developed.
Conclusions
Combined intravitreal ranibizumab and PDT for patients with retinal angiomatous proliferation effectively maintained or improved visual acuity and reduced the exudation without adverse events.
Retinal angiomatous proliferation (RAP) was first identified by Yannuzzi and associates. in 2001. RAP has been described as a variant of exudative age-related macular degeneration (AMD) and has been differentiated into 3 stages based on clinical and angiographic observations: stage 1, proliferation of intraretinal capillaries originating from the deep retinal complex (intraretinal neovascularization); stage 2, growth of the retinal vessels into the subretinal space (subretinal neovascularization); and stage 3, clinically or angiographically observed choroidal neovascularization (CNV). Recently, Yannuzzi classified stage 3 with retinal choroidal anastomosis as stage 4. RAP sometimes is referred to as type 3 neovascularization to distinguish it from the Gass CNV type 1 and type 2 anatomic classifications described by Freund and associates.
The proportion of RAP is 15% of newly diagnosed cases of neovascular AMD in white patients and 4.5% in Japanese patients. RAP has a different natural course and response to therapies compared with typical exudative AMD. The risk of neovascularization in the fellow eye is higher in patients with RAP than in those with other forms of neovascular AMD. So far, various treatments for RAP such as conventional laser photocoagulation, transpupillary thermotherapy, surgical ablation, and monotherapy of photodynamic therapy (PDT) with verteporfin (Visudyne; Novartis Pharma AG, Basel, Switzerland) have not been efficacious.
Vascular endothelial growth factor (VEGF) is well known to be closely related to CNV complexes. In animal models, anti-VEGF therapy prevents formation of CNV and decreases leakage from existing CNV. The major clinical trials have reported that ranibizumab (Lucentis; Genentech, Inc, South San Francisco, California, USA) was effective in improving visual acuity (VA) for AMD patients. The treatment of intravitreal ranibizumab has become one of the evidence-based therapies for AMD all over the world. In addition, recent studies have reported that combined therapy of intravitreal ranibizumab with PDT required fewer ranibizumab injections than ranibizumab monotherapy. However, RAP was excluded in the major clinical trials. Recently, anti-VEGF therapy using intravitreal ranibizumab or bevacizumab has been reported to be useful treatment. We also have reported that combined therapy of intravitreal bevacizumab and PDT was effective for treating RAP after 6 and 12 months of follow-up. However, bevacizumab for the treatment of RAP is an off-label use; it is recommended as therapy in combination with ranibizumab for AMD. The purpose of the current study was to clarify the efficacy of combined therapy of intravitreal ranibizumab and PDT for treating patients with RAP over 12 months.
Methods
We retrospectively reviewed 20 consecutive eyes of 16 Japanese patients (8 men, 8 women; age range, 79 to 92 years; mean age ± standard deviation, 84.8 ± 4.8 years) with treatment-naïve RAP who subsequently were treated with combination therapy using intravitreal ranibizumab and PDT at different disease stages. All patients were treated using combined therapy with intravitreal ranibizumab injections and standard fluence PDT from April 2009 through March 2010. Ranibizumab became available for medical use in Japan from March 2009. The patients were followed up for at least 12 months at Fukushima Medical University Hospital. The exclusion criteria were previous treatment for RAP such as laser photocoagulation, submacular surgery, transpupillary thermotherapy, PDT monotherapy, and combined therapy PDT with intravitreal triamcinolone acetonide or bevacizumab; glaucoma; tears in the retinal pigment epithelium; and maculopathies such as diabetic maculopathy, retinal vascular occlusion, or idiopathic macular telangiectasia.
We used the best-corrected visual acuity (BCVA) measured with a Japanese standard decimal VA chart and calculated the mean BCVA using the logarithm of the minimal angle of resolution scale. We used the theoretical value of the Early Treatment Diabetic Retinopathy study chart, which corresponds to each decimal visual acuity character according to the retreatment guidelines for ranibizumab in Japan.
All patients underwent a standardized examination including slit-lamp biomicroscopy with a contact lens, color or red-free fundus photography, fluorescein angiography (FA), and indocyanine green angiography (ICGA) with a fundus camera (TRC-50 FA/IA/IMAGEnet H1024 system; Topcon, Tokyo, Japan) and a confocal scanning laser ophthalmoscope (Heidelberg Retina Angiograph 2; Heidelberg Engineering, Heidelberg, Germany). All OCT examinations were performed using spectral-domain OCT (Heidelberg Spectralis OCT; Heidelberg Engineering). The central retinal thickness (CRT), defined as the distance from the retinal pigment epithelium to the inner limiting membrane at the center of the fovea, was measured at baseline and at 3, 6, 9, and 12 months after treatment (months 3, 6, 9, and 12, respectively) using internal caliper software. FA was performed to determine the lesion type, location, and CNV activity of the RAP lesions, that is, the degree or extent of leakage and the greatest linear dimension. ICGA was performed to diagnose RAP. We identified the retinal–retinal anastomosis by early-phase ICGA images using a confocal scanning laser ophthalmoscope.
All patients had documented visual loss at baseline. The initial treatment included intravitreal ranibizumab injection and PDT 1 or 2 days after the ranibizumab injection, followed by additional intravitreal injections of ranibizumab each month over the next 2 months. Intravitreal ranibizumab was injected 3.5 to 4.0 mm posterior to the corneal limbus into the vitreous cavity using a 30-gauge needle after topical anesthesia was applied. PDT with the standard dose (6 mg/m 2 ) of verteporfin was administered according to the protocol of the Treatment of Age-Related Macular Degeneration with Photodynamic Therapy study. A 689-nm laser system (Carl Zeiss, Dublin, California, USA) was used and 50 J/cm 2 energy was delivered with an 83-second exposure time. The greatest linear dimension was measured based on FA findings. After the initial treatment of combined therapy with PDT and 3 monthly intravitreal ranibizumab injections, follow-up examinations, including evaluation of the OCT images, were carried out every month until month 12. FA and ICGA were performed every 3 months until RAP lesions regressed completely. After achievement of complete regression of RAP lesions, FA and ICGA were performed when new exudative changes or new hemorrhage could be demonstrated by OCT findings or slit-lamp biomicroscopy with a contact lens. In cases with recurrence of RAP, we performed retreatment with combined therapy of a single intravitreal ranibizumab injection and PDT. After the retreatment of combined therapy, additional injections were administered according to the criteria during the second year of the Prospective OCT Imaging of Patients with Neovascular AMD Treated with intraOcular Ranibizumab study, that is, VA loss of at least 5 letters of the Early Treatment Diabetic Retinopathy study score with OCT evidence of fluid at the macula, an increase in the CRT on OCT of at least 100 μm, a new macular hemorrhage, a new area of classic CNV, or evidence of persistent fluid on OCT 1 month after the previous injection.
Statistical analyses were performed using the Student t test to compare the VA and the CRT at months 3, 6, 9, and 12. We used logarithm of the minimal angle of resolution VA scores for statistical analyses.
Results
Table 1 shows the characteristics and clinical data of the consecutive 16 patients (20 eyes). All patients were Japanese and were observed for 12 months. Eleven eyes (55%) had stage 2 RAP without a retinal pigment epithelial detachment (PED), 7 (35%) eyes had stage 2 RAP with a PED, and 2 (10%) eyes had stage 3 RAP ( Table 1 ). The mean greatest linear dimension of the entire lesion was 2805 μm.
| Case No. | Age (y) | Gender | Eye | RAP Stage | Baseline | Month 12 a | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VA (logMAR) | CRT (μ) | RRA | GLD (μ) | VA (logMAR) | CRT (μ) | RRA | CME | SRD | PED | Exudative Change | No. Treatments of PDT | No. Treatments of IVR | |||||
| 1 | 88 | F | Left | 2 | 0.5 (0.30) | 357 | Yes | 2276 | 0.5 (0.30) | 130 | No | Di | — | — | No | 1 | 3 |
| 2 | 83 | M | Right | 2 + PED | 0.6 (0.22) | 286 | Yes | 2571 | 0.9 (0.05) | 229 | No | Di | — | NC | No | 1 | 3 |
| 3 | 92 | M | Left | 2 | 0.6 (0.22) | 335 | Yes | 460 | 1.0 (0.00) | 143 | No | — | Di | — | No | 1 | 3 |
| 4 | 83 | M | Left | 2 + PED | 0.2 (0.70) | 574 | Yes | 2233 | 0.7 (0.15) | 165 | No | Di | — | Di | No | 2 | 4 |
| 5 | 82 | M | Right | 2 | 0.06 (1.22) | 292 | No | 1271 | 0.08 (1.10) | 139 | — | Di | — | — | No | 1 | 3 |
| 6 | 82 | M | Left | 2 | 0.2 (0.70) | 554 | Yes | 2733 | 0.4 (0.40) | 114 | No | Di | — | — | No | 2 | 4 |
| 7 | 83 | F | Right | 3 | 0.07 (1.15) | 572 | Yes | 5569 | 0.2 (0.70) | 168 | Yes | Re | Di | De | Yes | 4 | 6 |
| 8 | 83 | F | Left | 2 + PED | 0.15 (0.82) | 438 | Yes | 5565 | 0.4 (0.40) | 98 | No | Di | Di | Di | No | 1 | 3 |
| 9 | 91 | F | Left | 2 + PED | 0.1 (1.00) | 619 | Yes | 6815 | 0.24 (0.62) | 253 | Yes | Re | Di | De | Yes | 2 | 4 |
| 10 | 79 | M | Right | 3 | 0.07 (1.15) | 687 | Yes | 5590 | 0.24 (0.62) | 103 | No | Di | Di | Di | No | 1 | 3 |
| 11 | 79 | M | Right | 2 | 1.0 (0.00) | 298 | Yes | 1225 | 1.0 (0.00) | 143 | No | Di | — | — | No | 2 | 4 |
| 12 | 79 | M | Left | 2 | 0.2 (0.70) | 668 | Yes | 3326 | 0.4 (0.40) | 109 | No | Di | Di | — | No | 2 | 4 |
| 13 | 81 | M | Left | 2 | 0.5 (0.30) | 398 | Yes | 2569 | 0.8 (0.10) | 147 | No | Di | — | — | No | 2 | 4 |
| 14 | 81 | F | Right | 2 | 1.2 (−0.08) | 240 | Yes | 679 | 1.0 (0.00) | 157 | No | Di | — | — | No | 3 | 5 |
| 15 | 81 | F | Left | 2 + PED | 0.08 (1.10) | 658 | Yes | 3033 | 0.4 (0.40) | 111 | No | Di | — | Di | No | 2 | 4 |
| 16 | 79 | F | Left | 2 | 0.1 (1.00) | 365 | Yes | 3036 | 0.07 (1.15) | 106 | No | Di | — | — | No | 2 | 4 |
| 17 | 92 | F | Left | 2 + PED | 0.3 (0.52) | 291 | Yes | 2875 | 0.6 (0.22) | 146 | No | Di | Di | Di | No | 1 | 3 |
| 18 | 91 | M | Right | 2 + PED | 0.4 (0.40) | 408 | Yes | 2615 | 0.5 (0.30) | 189 | No | Di | Di | Di | No | 1 | 3 |
| 19 | 87 | F | Right | 2 | 0.5 (0.30) | 508 | Yes | 548 | 0.5 (0.30) | 116 | No | Di | Di | — | No | 2 | 4 |
| 20 | 85 | F | Left | 2 | 0.2 (0.70) | 355 | Yes | 1101 | 0.6 (0.22) | 94 | No | Di | — | — | No | 2 | 4 |
| Mean | 85 | 0.24 (0.62) | 444 | 2805 | 0.43 (0.37) | 143 | 1.8 | 3.8 | |||||||||
| SD | 4.8 | 147 | 1822 | 42 | 0.8 | 0.8 | |||||||||||
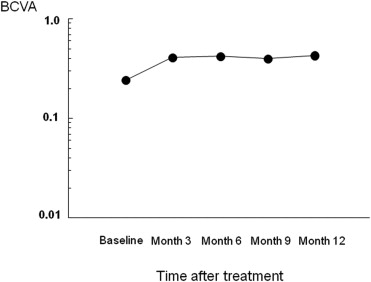
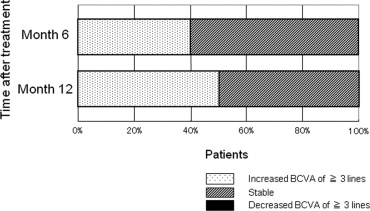
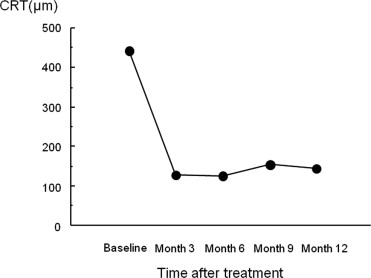
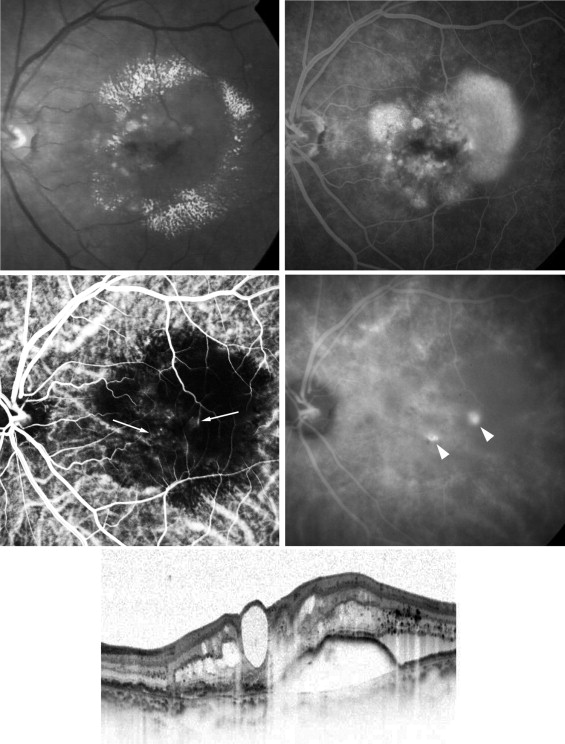
The mean decimal BCVA (logarithm of the minimal angle of resolution equivalent) levels at baseline and months 3, 6, 9, and 12 were 0.24 (0.62), 0.41 (0.38), 0.42 (0.37), 0.40 (0.40), and 0.43 (0.37), respectively ( Figure 1 ) . A significant improvement in the mean BCVA from baseline was seen at 3, 6, 9, and 12 months ( P < .0001, P < .0001, P < .0001, and P < .001, respectively, using the paired t test; Figure 1 ). The mean changes in BCVA at months 3, 6, 9, and 12 were improvements of 2.37, 2.48, 2.20, and 2.51 lines, respectively. Seven (35%) of the 20 eyes had an increase in the BCVA of 3 lines or more, and 13 (65%) eyes had stable VA (defined as a loss of fewer than 3 lines of vision) at month 3. At months 6 and 9, 8 (40%) of the 20 eyes had an increase in the BCVA of 3 lines or more, and 12 (60%) eyes had stable VA ( Figure 2 ) . Ten (50%) of the 20 eyes had an increase in the BCVA of 3 lines or more, and 10 (50%) eyes had stable VA at month 12 ( Figure 2 ). No patient had a decrease in BCVA of 3 lines or more after treatment during the 12 months of follow-up. The CRT significantly decreased from 444 ± 147 μm at baseline to 127 ± 34 μm at 3 months ( P < .0001), 124 ± 45 μm at 6 months ( P < .0001), 153 ± 41 μm at 9 months ( P < .0001), and 143 ± 42 μm at 12 months ( P < .0001; Figure 3 ) . At baseline, 19 of the 20 eyes had cystoid macular edema (CME); 9 of the 20 eyes had a serous retinal detachment; and 9 of the 20 eyes had a PED ( Table 1 ). The CME resolved in all 19 (100%) eyes a mean of 2.5 weeks after baseline. The serous retinal detachment resolved in all 9 (100%) eyes a mean of 4.0 weeks after baseline. The PED resolved in 6 (66.7%) eyes a mean of 6.2 weeks after baseline, decreased in 2 eyes, and remained 1 eye. At month 12, OCT showed the recurrence of CME in 2 eyes and the persistence of PED in 3 eyes ( Table 1 ). Figures 4, 5, 6, and 7 show ocular images demonstrated in patients treated with combined therapy of intravitreal ranibizumab and PDT.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


