Purpose
To determine whether a fluocinolone acetonide sustained-release intravitreal drug delivery system can be implanted safely at the same time that a glaucoma drainage device is placed for eyes with uveitis and elevated intraocular pressure (IOP) receiving maximum tolerated IOP-lowering therapy.
Design
Retrospective, observational case series.
Methods
Subjects had chronic noninfectious intermediate or posterior uveitis and elevated IOP while receiving maximum tolerated medical therapy. Fluocinolone acetonide implantation and glaucoma tube shunt placement were performed in a single surgical session. The main outcome measures were inflammatory recurrences, visual acuity (VA), use of adjunctive anti-inflammatory therapy, IOP, and adverse events.
Results
Seven eyes of 5 patients were studied. The average number of recurrences 12 months before implantation was 3 episodes per eye; of the 3 eyes followed up for more than 30 months, none had an inflammatory recurrence within 30 months after implantation. The mean Snellen visual acuity 12 months after the combined surgery was 20/114, compared with 20/400 at baseline. Adjunctive steroid use decreased. Average IOP decreased from 27.3 mm Hg at baseline to 14.6 mm Hg 12 months after the combined surgery ( P = .019).
Conclusions
The favorable results observed in all eyes suggest that fluocinolone acetonide implantation can be safely combined with glaucoma tube shunt placement in a single surgical session in eyes with uveitis and elevated IOP receiving maximum tolerated IOP-lowering therapy. Uveitis recurrences decreased, visual acuity improved, and IOP decreased. There were no adverse events during insertion of the fluocinolone acetonide implant and placement of the glaucoma tube shunt.
Elevated intraocular pressure (IOP) is an important secondary complication of uveitis that may result from ocular inflammatory sequelae or from corticosteroids used to treat the inflammation. For example, in one series, secondary glaucoma was found in 18.3% of patients with uveitis. This frequency is dependent on disease chronicity and occurs in 12% of patients with acute uveitis and 26% of those with chronic uveitis.
Uveitis typically is treated with corticosteroids, often with great benefit. However, among the general population, elevated IOP develops in one third of eyes treated with topical corticosteroids, and topical corticosteroids can further contribute to elevated IOP in eyes of patients with uveitis. Other forms of local corticosteroid treatment also can induce IOP elevations. Sustained IOP increases are seen in 22.6% and 34.6% of patients 1 year after a posterior sub-Tenon capsule injection or an intravitreal triamcinolone acetonide injection, respectively.
Elevated IOP is a common and significant complication in patients with uveitis treated with fluocinolone acetonide intravitreal implants (Retisert; Bausch & Lomb, Inc., Rochester, New York, USA). In a 3-year follow-up study of patients with uveitis treated with the 0.59-mg fluocinolone acetonide implant, 71% of implanted eyes had an IOP increase of 10 mm Hg or more compared with baseline IOP, and in 51.2%, 22.5%, and 6.1% of eyes, an IOP of 30, 40, and 50 mm Hg or more, respectively, developed. Thus, previous history of elevated IOP in response to corticosteroid steroid treatment or a history of glaucoma may be relative contraindications to fluocinolone acetonide implant insertion.
Despite the possibility of elevated IOP, the fluocinolone acetonide implant stabilizes or improves visual acuity, decreases uveitic recurrences, and decreases requirements for systemic and local anti-inflammatory therapy in most patients with noninfectious posterior uveitis. Some patients with uveitis are intolerant or not fully responsive to systemic corticosteroid or nonsteroidal immunosuppressive treatment or may require local corticosteroids at an impractical dosing frequency, and also may have a previous history of steroid-induced elevated IOP or glaucoma. Furthermore, glaucoma procedures in eyes with uveitis are fraught with potential complications and have reduced success rates compared with nonuveitic eyes. In these patients without other optimal therapeutic options, we hypothesized that a combined surgical approach that included placement of both a fluocinolone acetonide intravitreal implant and a glaucoma drainage device at the same surgical sitting would offer a useful alternative treatment for this challenging patient population. We anticipated that with this approach, excellent postoperative inflammation control afforded by the fluocinonide acetonide implant may improve glaucoma surgical success rates and may minimize complications incurred by ongoing postoperative inflammation, while reducing the need for adjunctive anti-inflammatory therapy. Furthermore, the combined surgical approach would eliminate anesthetic risks and costs associated with 2 separate procedures performed at different surgical sittings. Herein, we determined for the first time the feasibility and outcomes of such a combined procedure in a series of patients with noninfectious posterior uveitis who required multiple IOP-lowering medications to control elevated IOP.
Methods
Study Population
Patients eligible for the study met the following criteria: a history of chronic recurrent noninfectious posterior uveitis or intermediate uveitis with or without iridocyclitis; incomplete therapeutic response or treatment-limiting side effects to oral, topical, or periocular corticosteroids, immunosuppressive agents, or a combination thereof; elevated IOP that required maximum medical therapy for IOP control or elevated IOP that could not be controlled with maximum medical therapy; visual acuity (VA) of at least light perception; and ability to comprehend informed consent and to comply with follow-up examinations. Patients were excluded if they had a toxoplasmosis scar in the study eye or a peripheral retinal detachment in the area of the planned fluocinolone acetonide implant placement. Our study population consisted of patients who met the above eligibility criteria and who underwent simultaneous insertion of a fluocinolone acetonide implant combined with placement of an Ahmed glaucoma drainage device (New World Medical Inc., Rancho Cucamonga, California, USA).
Study Design and Outcome Measures
All patients were enrolled consecutively and were treated initially at the Duke Eye Center between January 9, 2004, and November 7, 2008. This study was approved by the Duke University Institutional Review Board. Informed consent was obtained from all patients. Surgeries were performed during the same surgical session by a single vitreoretinal surgeon (G.J.J.), who inserted the fluocinolone acetonide implant, and by a glaucoma surgeon experienced in the technical considerations needed in uveitic patients, who placed the Ahmed tube drainage device. In all cases, a Food and Drug Administration-approved fluocinolone acetonide 0.59-mg implant, targeted to release at approximately 0.5 mg/day, was inserted. After surgery, clinic visits occurred on postoperative day 1, week 1, weeks 4 to 6, and then approximately every 3 months thereafter.
Postoperative clinical recurrence in either eye was defined as follows: an increase in the number of anterior chamber (AC) cells by 2 grades, an increase in vitreous haze by 2 grades, an increase in cystoid macular edema, or whenever the investigator believed it was clinically necessary to provide anti-inflammatory treatment. Posterior sub-Tenon capsule triamcinolone acetonide injections were administered to the fluocinolone acetonide-implanted eye or the fellow eye for a 2-grade increase in AC cells. Local or systemic therapy or both with corticosteroids and other immunosuppressants also was initiated for ocular recurrence, and systemic treatment was given if the systemic disease became active.
Data Collection
Baseline and follow-up clinical data and intraoperative data, including all surgical procedures, adverse events, and intraoperative findings, were recorded in an electronic database. Clinical data collected included best-corrected VA measured by the protocol developed for the Early Treatment Diabetic Retinopathy Study, applanation tonometry (either Tono-Pen applanation [Reichert, Inc., Depew, New York, USA] or Goldmann applanation [Haag Streit International, Mason, Ohio USA]), AC cell measurement by slit-lamp biomicroscopy, and indirect ophthalmoscopy. In eyes with media sufficiently clear to allow ocular imaging, retinal thickness and morphologic features were determined by optical coherence tomography. Systemic, periocular, and topical therapies, which were reduced in accordance with the clinical response, were recorded at each visit. Intraoperative complications and postoperative adverse events such as endophthalmitis, retinal detachment, hypotony, suprachoroidal hemorrhage, tube shunt exposure, and vitreous hemorrhage also were recorded.
Preoperative Management
Before surgery, anterior chamber inflammation was treated aggressively with topical corticosteroids, periocular corticosteroid injection, systemic corticosteroids, or immunosuppressive agents alone or in combination to reduce anterior chamber cells to a level of 5 cells or fewer per high-power field, as determined by slit-lamp examination. Eyes were not operated on if there were more than 5 cells per high-power field to minimize hypotony associated with reduced aqueous production caused by ciliary body inflammation. Posterior segment inflammation was allowed before surgery if unaccompanied by anterior chamber cells as described above.
Surgical Technique
The surgery was performed under intravenous sedation with appropriate electrocardiographic, blood pressure, and oxygen saturation monitoring. A retrobulbar block consisting of 0.75% bupivacaine and 4% lidocaine in a 1:1 mixture with hyaluronidase was given for local anesthesia. The operative eye was prepared in a sterile manner, and a wire lid speculum was inserted into the eye. In all but 1 case, the fluocinolone acetonide implant was inserted by the vitreoretinal surgeon before the placement of the Ahmed tube shunt by the glaucoma surgeon. The fluocinolone acetonide sustained drug-delivery implant was inserted as described previously. In eyes that previously had undergone vitrectomy, an infusion line was placed first to prevent globe collapse during subsequent maneuvers. In all cases, the glaucoma tube shunt was placed in the supertemporal quadrant, beneath the Tenon capsule, and was secured to the sclera with 9-0 nylon suture. Six of 7 glaucoma tube shunts were Ahmed S2 models, and the other was an Ahmed model S3. Five of 7 eyes had the Ahmed tube placed in the anterior chamber, the other 2 eyes had the tube placed into the posterior chamber because those eyes had undergone previous vitrectomies. In 6 of 7 eyes, the sclerostomy was created with a 22-guage needle, and in 1 eye, the sclerostomy was created with a 23-guage needle. The tubes were covered with Tutoplast sclera (IOP, Inc., Costa Mesa, California, USA) or pericardium patch grafts, which were secured to the scleral bed with 2 interrupted 7-0 polyglactin 910 sutures. The conjunctival wounds were closed with a running, locking 8-0 polyglactin 910 suture. In eyes with an infusion line, as the infusion line was removed, the preplaced suture around the infusion cannula was tied to maintain a normal IOP and to minimize intraocular bleeding at the sclerotomy site. At the end of the surgery, either 100 or 400 mg cefazolin and 5 mg dexamethasone were injected in the subconjunctival space.
Statistical Analysis
Statistical analysis was performed using JMP software version 6.0 (SAS Institute, Inc, Cary, North Carolina, USA). The main outcome measures included topical, periocular, intravitreal, and systemic corticosteroid use; systemic immunosuppressive therapy; VA; IOP; cup-to-disc ratio; and adverse events. Descriptive statistics (mean, median, standard deviation [SD], and minimum and maximum values) were computed for continuous baseline and postoperative data (age, preoperative and postoperative distance VAs using logarithm of the minimal angle of resolution [logMAR] units derived from Early Treatment Diabetic Retinopathy Study charts, IOPs in millimeters of mercury [mm Hg], and number of topical and systemic medications). The significance of the change in baseline IOP compared with the 12-month IOP was determined using the Student t test. A paired t test, correcting for the duration of postoperative follow-up, was performed to determine whether there was a statistically significant difference in inflammation recurrence rate before and after the fluocinolone acetonide implant was placed. P values < .05 were considered statistically significant.
Results
Combined fluocinolone acetonide implant insertion and Ahmed glaucoma drainage device placement surgery was performed on 7 eyes of 5 patients. Three eyes of 2 of these patients also underwent an additional third procedure, limbal phacoemulsification with intraocular lens implant, during the same surgical session. Three patients were white, 1 patient was black, and 1 patient was Native American ( Table 1 ). Three of the 5 patients were female. The mean patient age at the time of the combined surgery was 46.1 years (median, 42.6 years; range, 25.2 to 68.1 years). Three patients had idiopathic panuveitis, 1 had sarcoidosis, and 1 had panuveitis presumed to be secondary to Vogt-Koyanagi-Harada syndrome. Three of 5 patients had bilateral disease. All surgeries were performed between November 17, 2005, and January 29, 2009. The mean IOP at the time of the surgery was 27.3 mm Hg (median, 29.5 mm Hg; range, 17 to 41.5 mm Hg). The mean follow-up duration after the combined surgery was 21.9 months (median, 24.7 months; range, 6.0 to 39.4 months). All 7 eyes were followed up for 6 months or longer; 4 of the 7 eyes were followed up for 12 months or longer.
| Patient No. (Eye) | Race | Diagnosis | Gender | Patient Age at Surgery (yrs) |
|---|---|---|---|---|
| 1 | Black | Idiopathic unilateral panuveitis | M | 43 |
| 2 | White | Idiopathic bilateral panuveitis | F | 55 |
| 3 (right) | Native American | Vogt-Koyanagi-Harada syndrome | F | 26 |
| 3 (left) | Native American | Vogt-Koyanagi-Harada syndrome | F | 25 |
| 4 (right) | White | Sarcoidosis | F | 67 |
| 4 (left) | White | Sarcoidosis | F | 68 |
| 5 | White | Idiopathic unilateral panuveitis | M | 39 |
Inflammation
There was a statistically significant reduction in the number of inflammatory recurrences after the surgery, compared with the number of recurrences during the 12 months before surgery ( P = .007; 95% confidence interval, 0.0016 to 0.036). On average, there were 3 recurrences per eye in the 12 months before the fluocinolone acetonide implantation. Of the 4 eyes followed up for less than 30 months, no eye had a recurrence after surgery. Of the 3 eyes followed up for more than 30 months, no eyes had an inflammatory recurrence in the first 30 months after implantation. One eye had an inflammatory recurrence at 33 months after implantation.
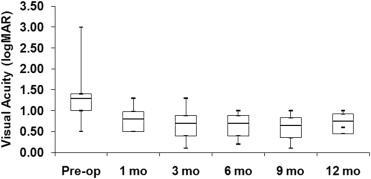
Visual Acuity
The mean baseline VA ± SD for the combined fluocinolone acetonide implanted eyes with glaucoma drainage device was 1.37 ± 0.80 logMAR units (n = 7; median, 1.30; Snellen equivalent, 20/400). This improved to 0.81 ± 0.29 logMAR units (n = 7; median, 0.80; Snellen equivalent, 20/125) at 1 month, 0.67 ± 0.37 logMAR units (n = 7; median, 0.70; Snellen equivalent, 20/100) at 3 months, 0.64 ± 0.28 logMAR units (n = 7; median, 0.70; Snellen equivalent, 20/80) at 6 months, 0.60 ± 0.39 logMAR units (n = 4; median, 0.64; Snellen equivalent, 20/80) at 9 months, and 0.78 ± 0.17 logMAR units (n = 4; median, 0.75; Snellen equivalent, 20/114) at 1 year after the surgery ( Figure 1 ). At all time points, 1, 3, 6, 9, and 12 months of follow-up, there was a greater proportion of eyes that had gained three lines or more of VA ( Figure 2 ). There was a greater proportion of eyes with 3 lines or more of VA improvement than those that had no change or only 1 line of improved VA after the surgery, and no eyes lost vision compared with baseline. All 4 eyes at 12 months of follow-up had a VA between 20/80 to 20/200, whereas 5 of 7 eyes had a VA 20/200 or less before surgery.