Purpose
To explore the association of angiographic nonperfusion in focal and diffuse recalcitrant diabetic macular edema (DME) in diabetic retinopathy (DR).
Design
A retrospective, observational case series of patients with the diagnosis of recalcitrant DME for at least 2 years placed into 1 of 4 cohorts based on the degree of DR.
Methods
A total of 148 eyes of 76 patients met the inclusion criteria at 1 academic institution. Ultra-widefield fluorescein angiography (FA) images and spectral-domain optical coherence tomography (SD OCT) images were obtained on all patients. Ultra-widefield FA images were graded for quantity of nonperfusion, which was used to calculate ischemic index. Main outcome measures were mean ischemic index, mean change in central macular thickness (CMT), and mean number of macular photocoagulation treatments over the 2-year study period.
Results
The mean ischemic index was 47% (SD 25%; range 0%-99%). The mean ischemic index of eyes within Cohorts 1, 2, 3, and 4 was 0%, 34% (range 16%-51%), 53% (range 32%-89%), and 65% (range 47%-99%), respectively. The mean percentage decrease in CMT in Cohorts 1, 2, 3, and 4 were 25.2%, 19.1%, 11.6%, and 7.2%, respectively. The mean number of macular photocoagulation treatments in Cohorts 1, 2, 3, and 4 was 2.3, 4.8, 5.3, and 5.7, respectively.
Conclusions
Eyes with larger areas of retinal nonperfusion and greater severity of DR were found to have the most recalcitrant DME, as evidenced by a greater number of macular photocoagulation treatments and less reduction in SD OCT CMT compared with eyes without retinal nonperfusion. Areas of untreated retinal nonperfusion may generate biochemical mediators that promote ischemia and recalcitrant DME.
Diabetic macular edema (DME) continues to be a common cause of vision loss in patients with diabetic retinopathy (DR) and decreased vision-related quality of life in working-aged Americans. The prevalence of DME among US diabetics approaches 30% in adults who have had diabetes for 20 years or more, and varies with the stage of diabetic retinopathy. It can occur at any stage of diabetes and can predate the appearance of other findings of diabetic retinopathy. In eyes with mild nonproliferative diabetic retinopathy (NPDR), the prevalence of DME is 3%. This rises to 38% in eyes with moderate to severe NPDR and reaches 71% in eyes with proliferative diabetic retinopathy (PDR).
For nearly 3 decades, the standard therapy and only approved therapy for DME has been focal/grid laser photocoagulation. The Early Treatment Diabetic Retinopathy Study (ETDRS) found a 50% reduction in the likelihood of severe vision loss with focal/grid macular laser. The Diabetic Retinopathy Clinical Research Network more recently reported a 10-letter gain in nearly one-third of patients treated with macular photocoagulation, but reported that 19% experience progressive visual loss. The increasing incidence of sight-threatening DME with limited visual improvement following focal/grid laser treatment has challenged clinical researchers to develop more efficient alternatives for the diagnosis and treatment of DME. Several clinical trials, including READ-2 and RISE and RIDE, have investigated the use of anti–vascular endothelial factor (anti-VEGF) for DME and have shown rapid, sustained visual improvement. Given the favorable results of these trials, ranibizumab was recently FDA-approved for the treatment of DME.
Diagnostic imaging has played an increasing role in eye care in recent years. Under optimal conditions, traditional angiographic methods employing film or digital fundus cameras can capture 50-degree views. With 7 standard fields, a 75-degree view of the fundus can be obtained. Recent advances in scanning laser ophthalmoscope (SLO) technology now allow for consistently reproducible high-resolution angiographic images of the peripheral retina. This development enables panretinal angiographic assessment of retinal vascular perfusion without the need to extrapolate nonsimultaneous angiographic views within a montage. Contact lens–based systems such as the Ocular Staurenghi 230 SLO Retina Lens (Ocular Instruments, Bellevue, Washington, USA) can image out to the 120-degree range. Ultra-widefield fluorescein angiography (FA) using the Optos C200 MA noncontact SLO (Optos PLC, Dunfermine, UK) provides visualization up to 200 degrees.
Researchers (Schwartz SD, et al. IOVS 2005;46:ARVO E-Abstract 4793) have hypothesized the association between peripheral nonperfusion and the presence of neovascularization and macular edema. For patients who have clinically significant DME involving the center of the macula, and high-risk PDR requiring panretinal photocoagulation (PRP), combined focal/grid and PRP laser is recommended by the ETDRS Group and Preferred Practice Pattern (American Academy of Ophthalmology, 2003). Although the ETDRS and other studies provided excellent clinical recommendations, patient management still remains challenging. Despite following current practice patterns, a subset of patients with DME continues to have persistent or recalcitrant DME in spite of several sessions of focal/grid macular photocoagulation or monthly intravitreal injections of VEGF inhibitors and/or intravitreal steroids. A potential explanation for current challenges was nicely reviewed in a recent publication by Wessel and associates showing that peripheral retinal ischemia is significantly correlated with DME in treatment-naïve patients with DR using a clinically practical device.
In this study our main objective was to study patients with focal and diffuse recalcitrant DME and determine the relationship between peripheral nonperfusion assessed with ultra-widefield FA in patients with different severities of DR. We hypothesized that zones of peripheral retinal nonperfusion may generate biochemical mediators such as VEGF and other cytokines that promote recalcitrant DME and cause a suboptimal response to standard therapies.
Methods
Study Population
A retrospective, consecutive, observational case series was carried out at 1 academic institution with the prospective approval of the Illinois Eye Institute Institutional Review Board (IRB), and therefore has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and HIPAA regulations. A chart review was performed on all patients seen with a diagnosis of diabetes mellitus type 2 from September 2, 2010 to March 30, 2012. Approximately 1156 patients diagnosed with diabetes mellitus type 2 were seen in our clinic, of which 76 patients (148 eyes) with recalcitrant DME, who were currently under the care of the University of Chicago Section of Ophthalmology Vitreoretinal Service and met eligibility criteria, were invited to participate in this study. Participants had to be at least 18 years of age or older, have type 2 diabetes, and be diagnosed with recalcitrant DME for at least 2 years. All patients provided informed consent before participation in the study.
Recalcitrant DME was defined as having clinically significant diabetic macular edema (CSDME) with a center macular thickness greater than 300 μm as measured by spectral-domain optical coherence tomography (SD OCT), for a minimum of 2 years duration despite standard therapies. For eyes that met eligibility criteria, the treatment algorithm for CSDME was macular photocoagulation at 3-month intervals only if the study eye had an increase in CMT or decrease in visual acuity ≥5 ETDRS letters. For eyes that met eligibility criteria, the treatment algorithm for PDR was a complete 360 degrees of PRP (between 800 and 1100 spots) at the first visit and then retreatment with PRP at 3-month intervals only if there were any clinical signs of PDR based on ETDRS criteria. Exclusion criteria included diagnosis of ocular disease other than diabetic retinopathy that may produce macular edema (eg, retinal artery or vein occlusion, sickle cell retinopathy, or uveitis) identified by clinical examination, eyes with vitreomacular traction or epiretinal membrane on SD OCT, eyes with tractional retinal detachment identified by clinical examination, eyes that had undergone previous vitreoretinal surgery, eyes that had undergone an anti-VEGF or steroid intravitreal injection within the 2-year study period, eyes with significant media opacities (ie, cataract, vitreous hemorrhage), and eyes with image artifact (eyelids) that precluded photographic evaluation of macular edema and peripheral retinal capillary perfusion status.
Imaging
All subjects underwent ultra-widefield FA using the Optos C200 MA imaging system (Optos PLC) and SD OCT (Cirrus OCT; Carl Zeiss Meditec Inc, Dublin, California, USA) performed at day 1. All ultra-widefield FA images were obtained with the Optos C200 MA (Optos PLC) after standard intravenous infusion of 5 cc of sodium fluorescein 10%. Images were digitally archived and reviewed using the V2 Vantage Review Software (Optos PLC), allowing high-resolution zoom functionality for the review of all images.
SD OCT images were obtained for each patient to confirm the presence of macular thickening. For each patient, horizontal and vertical SD OCT scans of 6 mm length through the fixation point were obtained for evaluation. An average thickness value was obtained after reviewing both scans. CMT was calculated using the calipers feature on the SD OCT instrument, with manual correction as needed.
Data Collection and Definitions
All ultra-widefield FA images were saved as high-quality jpeg files and then transferred to Apple Preview software. One trained masked grader (R.D.P.) selected 1 or more images from each angiogram series during the arteriovenous phase (between 45 seconds and 2 minutes) and graded all 148 angiograms for the presence or absence of retinal nonperfusion of ≥1 disc area, neovascularization (defined as focal leakage ≥2 disc diameters), focal macular edema (defined as late hyperfluorescence ≥500 μm diameter covering less than 75% of the macula), and diffuse macular edema (defined as late hyperfluorescence covering ≥75% of the macula). The best image was chosen based on the largest field of view and the greatest image clarity. Figure 1 illustrates typical findings encountered when grading.
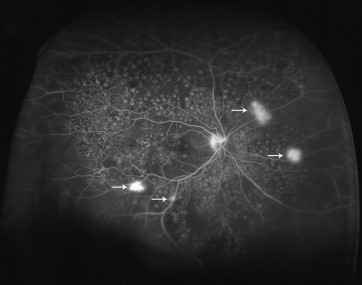
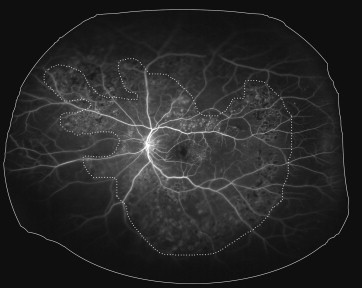
An ischemic index was calculated using previously described methodology. Briefly, the area of capillary nonperfusion seen in the arteriovenous phase image was encircled using the area measurement function and divided by the total image area in pixels ( Figure 2 ). The area of capillary nonperfusion was defined as the area where a dropout of the retinal capillary bed was detected in the ultra-widefield FA image. Retinal vascular leakage was not considered as nonperfusion. Ischemic index has been used in other studies as a marker for retinal nonperfusion.
All eyes were categorized clinically by state of disease, including mild NPDR, moderate to severe NPDR, and PDR according to the international classification proposed by the American Academy of Ophthalmology. Eyes were categorized as quiescent PDR (qPDR) if the patient was clinically diagnosed previously with PDR and had undergone previous PRP and had no clinical signs of proliferative disease for at least 2 years prior to day 1.
SD OCT images were reviewed for all patients to confirm the presence of macular thickening. Only eyes with evidence of angiographic macular leakage and macular thickening documented on SD OCT were considered to have macular edema. Patients were required to have recalcitrant DME with a CMT ≥300 μm by SD OCT and had not received any treatment in the study eye for at least 90 days prior to enrollment.
A chart review was performed for all patients for the following data: sex, age, mean arterial pressure (MAP), hemoglobin A1c, dependence on insulin, and the number of macular photocoagulation and PRP treatments during the 2-year study period. Hemoglobin A1c and MAP measurements were all within a 6-month period from the date of the ultra-widefield FA. SD OCT CMT measurements were recorded at 2 time points: (1) on the day of ultra-widefield FA (day 1) and (2) at a clinic visit at least 2 years prior (baseline). If no macular SD OCT was available exactly 2 years prior to the ultra-widefield FA date, the CMT measurements were taken from a macular SD OCT within 3 months of that date.
All patients referred for evaluation and management of recalcitrant DME and PDR who had undergone laser photocoagulation (focal/grid and/or PRP) within the study period had undergone laser treatment aided with traditional OCT and angiographic techniques, respectively. All patients who had undergone macular photocoagulation treatment followed the modified ETDRS protocol used in the DRCR.net clinical trials.
The term “study period” refers to the time period at least 2 years prior to the date of the ultra-widefield FA. The term “baseline” refers to the first day of the study period. The term “day 1” refers to the day of the angiogram and SD OCT.
Study Design
Four study cohorts were enrolled with eyes that had a diagnosis of recalcitrant DME for at least 2 years. Cohort 1 subjects were diagnosed with mild NPDR and treated with previous macular photocoagulation. Cohort 2 subjects were diagnosed with moderate or severe NPDR and treated with previous macular photocoagulation. Cohort 3 subjects were diagnosed with qPDR and treated with previous macular photocoagulation and PRP. Cohort 4 subjects were diagnosed with active PDR and treated with previous macular photocoagulation with or without previous PRP.
Statistical Analysis
The significance of the differences in the CMT between baseline and at day 1 of the study was analyzed by nonparametric Wilcoxon signed rank test. SPSS version 17.0J for Windows (SPSS Inc, Chicago, Illinois, USA) was used for statistical analyses. A P value of <.05 was considered significant.
Results
Clinical Characteristics of Patients
The clinical characteristics of the 76 patients (41 men and 35 women) are summarized in Table 1 . The mean age of the patients was 48.2 years (range 36-84 years). The mean duration of recalcitrant DME was 32.5 months (range 24-38 months). A total of 158 ultra-widefield fluorescein angiograms from 81 patients initially reviewed. However, 10 angiograms could not be interpreted owing to poor quality, and were therefore excluded. Ultimately, 148 eyes of 76 patients were used in the analysis. Angiographic macular edema was identified in all 148 eyes.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


