Purpose
To document the characteristics of the retinal surface using en face optical coherence tomography (OCT) after internal limiting membrane (ILM) peeling during pars plana vitrectomy (PPV) for myopic macular diseases.
Design
Retrospective, interventional case series.
Methods
We examined 20 eyes of 20 patients with high myopia who underwent PPV with ILM peeling. The patients were followed for at least 6 months postoperatively, and OCT images were examined using spectral-domain OCT. To assess the incidence and patterns of the retinal dimple sign, we reconstructed en face OCT images and quantified the number of dimples during the follow-up period.
Results
We identified 3 retinal dimple signs (ie, foveal-centered pattern, linear pattern, and paravascular pattern). The total numbers of retinal dimples at baseline and 1, 3, and 6 months postoperatively were 0, 31.8 ± 23.7, 47.95 ± 29.5, and 54.6 ± 30.7, respectively. There were significantly more dimples temporal to the macula compared to the other 3 quadrants at 1, 3, and 6 months postoperatively ( P = .025, P = .0021, and P = .0060, respectively). However, the retinal sensitivities assessed by microperimetry were not correlated with the number of dimples.
Conclusion
Development of the retinal dimple sign after PPV and ILM peeling in highly myopic eyes was seen in all cases on en face OCT images. Although retinal dimples are benign, care should be taken after ILM peeling in highly myopic eyes.
Removal of internal limiting membranes (ILM) contributes significantly to anatomic and functional success in various macular diseases, such as macular holes, epiretinal membranes, macular edema, and myopic foveoschisis. However, ILM peeling affects the optic nerve fiber layer and causes an unusual appearance resembling a nerve fiber layer bundle defect. Tadayoni and associates reported this finding, which they called the dissociated optic nerve fiber layer appearance, after pars plana vitrectomy (PPV) was performed to treat idiopathic epiretinal membranes and macular holes.
Spectral-domain optical coherence tomography (SD OCT) devices can acquire B-scans rapidly and facilitate creation of transversal en face OCT C-scans. Using 3-dimensional volume-rendering technologies, investigators have assessed the images obtained from patients who had undergone ILM peeling and reported en face images that showed the dissociated optic nerve fiber layer appearance. Alkabes and associates clearly described the appearance of inner retinal defects as concentric macular dark spots, which they observed in all their cases of idiopathic macular hole after ILM peeling. Subsequently, Spaide also referred to this change as inner retinal dimpling. Each report described closely spaced depressions that appeared to correspond to localized areas of thinning under the retinal nerve fiber layer (RNFL). Moreover, the concentric macular dark spots or retinal dimples did not develop immediately after surgery but emerged from 1 to 3 months postoperatively.
High myopia induces various macular disorders, such as foveoschisis, choroidal neovascularization, and chorioretinal atrophies, and often leads to severe visual loss. Myopic foveoschisis was recognized as a result of the development of OCT. The prevalence of myopic foveoschisis has been reported to range from 8% to 34% in highly myopic eyes. The pathogenesis of foveoschisis remains uncertain; however, based on the foveal status seen on OCT images, eyes with myopic foveoschisis can be divided into 3 types: those with a foveal detachment, retinoschisis, or a macular hole. Vitrectomy with ILM peeling had been thought to be a reasonable therapeutic approach, and many studies have reported good visual outcomes.
Nevertheless, inner retinal changes such as concentric macular dark spots or retinal dimpling after ILM peeling in high myopia have not been fully described. Moreover, Spaide presented a patient in a case series who had central artery occlusion seen intraoperatively. In this case, retinal dimpling also was observed, even after marked rarefaction of the nerve fiber layer, owing to ischemic necrosis. This report prompted us to assess the appearance of retinal dimpling even in eyes with high myopia (foveoschisis, foveal detachment, macular hole), which are thought to have a thinner RNFL.
Patients and Methods
Data Collection
This retrospective study was performed in accordance with the tenets of the Declaration of Helsinki. The institutional review board of the Osaka University Hospital approved the study. All patients provided written informed consent before surgery. The study was an interventional case series of 20 eyes of 20 highly myopic patients who underwent PPV with ILM peeling to treat myopic foveoschisis with or without retinal detachment or a myopic macular hole without retinal detachment from July 1, 2007 through March 31, 2013 at the Department of Ophthalmology, Osaka University Hospital. The inclusion criteria were eyes with myopic foveoschisis or a macular hole whose axial length was not less than 26.5 mm and a follow-up period exceeding 6 months after the initial PPV. Eyes with severe chorioretinal atrophy, such as a round whitish lesion at the fovea; a failed routine ocular examination before and after surgery; poor OCT image quality; a history of other intraocular surgeries except for cataract; and a retinal detachment beyond the vascular arcade were excluded.
Vitrectomy
A combined 3-port conventional 25 gauge vitrectomy and cataract surgery were performed in all phakic eyes. First, triamcinolone acetonide (0.2 mL of 20 mg/mL) was injected into the vitreous cavity to visualize the vitreous. Vitreous cortex that adhered to the inner retinal surface was removed gently with intraocular forceps or a vitreous cutter; 0.5 mL of 0.5% indocyanine green (ICG) or 0.025% Brilliant Blue G (BBG) was sprayed onto the posterior retina to stain the ILM. Forceps were used to peel ILMs of 3-4 disc diameters. Finally, fluid was exchanged for air and gas tamponade of either 20% sulfur hexafluoride or 14% or 16% perfluoropropane. The ILM was visualized with ICG in the initial 12 eyes (60.0%) and then switched to BBG in the remaining 8 eyes (40.0%). One surgeon (Y.I.) performed all surgeries. Postoperatively, the patients were instructed to maintain a prone position for a couple of days. The area of ILM peeling was evaluated using the captured images obtained during the video recordings of the surgeries in all cases. The disc area in each eye was used as a standard and measured using ImageJ software (NIH image, Bethesda, Maryland, USA).
Examinations
The best-corrected visual acuity (BCVA) measurements, color fundus photography (TRC-50; Topcon, Tokyo, Japan), and SD OCT examinations (Cirrus HD-OCT; Carl Zeiss Meditec, Dublin, California, USA) were performed during all preoperative and postoperative visits. The axial lengths were measured using the IOLMaster (Carl Zeiss Meditec); the refractive errors were measured using an autorefractometer (ARK-530; Nidek, Tokyo, Japan). The retinal dimpling sign was measured on en face SD OCT images preoperatively and 1, 3, and 6 months postoperatively. We looked for associated factors such as the BCVA, axial length (mm), refractive error (diopters [D]), and retinal sensitivity (decibels [dB]).
The retinal sensitivity was assessed in 13 eyes of 13 patients who agreed to undergo this examination using the Microperimeter-1 (MP-1; Nidek) at the final visit. A 4-2 staircase strategy with a Goldmann III size stimulus was used for the retinal sensitivity examination of 76 stimulus locations covering the central 20 degrees of the retina. We divided the 76 stimulus points into 4 groups (ie, the inferior, superior, nasal, and temporal quadrants), and each contained 19 stimulus points. We totaled the sensitivity (dB) of 19 points in each quadrant. Subsequently we evaluated the relationship between the number of dimples and total sensitivity in each quadrant.
En Face Optical Coherence Tomography and Quantification of Dimples
Volumetric dense OCT data were acquired before and after surgery using the Macular Cube 512 × 128 scan pattern on the Cirrus HD-OCT 4000. The 6 × 6-mm 3-dimensional cube data were exported and post-processed to remove acquisition artifacts attributable to ocular movements (GNU Octave; Free Software Foundation, Inc., Boston, Maryland, USA, with proprietary code). The obtained volumetric cube data can extract sharply curved slab data of various thicknesses and at various depths below the detected ILM surface to create appropriate en face images by adding the selected slab intensity data. We confirmed that the curved slab was correctly following the ILM shape and the slab was parallel to the retina at any point. For the current study, en face images representing a 20-μm-thick slab just below the ILM contour provided suitable image quality (compromise between contrast and image noise) for further analysis. Additional en face images obtained from 10-μm-thick slab data at depths of 10, 20, and 30 μm below the ILM surface allowed a qualitative check to ensure that the observed retinal dimples were of limited depth with no imaging artifacts.
We defined the appearance of multiple dark dots along the retinal nerve fibers in the area from which the ILM was peeled as a sign of retinal dimpling. Using ImageJ software, we quantified the number of retinal dimples on the en face images preoperatively and 1, 3, and 6 months postoperatively.
Statistical Analysis
The data were analyzed using JMP version 9.0 (SAS Institute, Cary, North Carolina, USA). One-way analysis of variance, Pearson correlation analysis, the Mann-Whitney U test, and the paired t test were performed as appropriate. P < .05 was considered significant.
Results
Patient Demographic Data
The mean patient age at the time of surgery was 65.7 ± 8.7 years (range, 49–80 years). Twelve eyes (60.0%) were phakic and 8 eyes (40.0%) were pseudophakic. The mean refractive error of the phakic eyes was −15.3 ± 2.54 D (range, −11.0 to −18.0 D); the mean axial length was 29.9 ± 1.50 mm (range, 27.07–32.4 mm). These baseline characteristics were not correlated with the total number of retinal dimples observed 6 months postoperatively ( Table 1 ). The primary preoperative diagnoses were foveoschisis without a retinal detachment in 14 eyes (70.0%), foveoschisis with a retinal detachment in 5 eyes (25.0%), and macular hole in 1 eye (5.0%).
| Categories of Pattern | Number of Eyes | % | |
|---|---|---|---|
| A. | Foveal-centerd pattern | 12 | 60.0 |
| B. | Linear pattern | 9 | 45.0 |
| C. | Paravascular pattern | 8 | 40.0 |
| Outcomes of Combination of Each Pattern | Number of Eyes | % | |
|---|---|---|---|
| A only | 6 | 30.0 | |
| B only | 4 | 20.0 | |
| C only | 2 | 10.0 | |
| A + B | 2 | 10.0 | |
| B + C | 2 | 10.0 | |
| C + A | 3 | 15.0 | |
| A + B + C | 1 | 5.0 | |
| Total | 20 | 100.0 |
Patterns of Retinal Dimpling
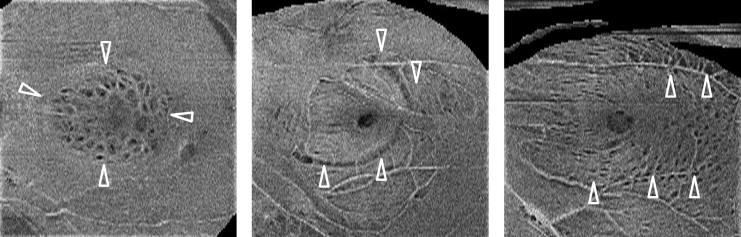
We identified 3 patterns of retinal dimpling after ILM peeling in highly myopic eyes ( Figure 1 ). The foveal-centered pattern (ie, concentrated retinal dimpling) was observed around the macula and limited to about 1-2 disc diameter areas. The linear pattern was characterized by dimples along the retinal nerve fibers (RNF), with dimpling that was more widely spread than the foveal-centered pattern. The paravascular pattern was similar to the linear pattern but developed not along the RNF but adjacent to vessels in the arcade. The foveal-centered pattern, linear pattern, and paravascular pattern were found in 12 eyes (60.0%), 9 eyes (45.0%), and 8 eyes (40.0%), respectively ( Table 2 ). Two or 3 patterns were present in 10 eyes (50.0%) ( Table 2 ).
| Factors | No. of Retinal Dimpling (Average ± SD) | P Value | |
|---|---|---|---|
| Average age (years ± SD) [range] | 65.7 ± 8.7 [49–80] | .316 | |
| Axial length (mm ± SD) [range] | 29.9 ± 1.5 [27.1–32.4] | .657 | |
| Disease, n (%) | .648 | ||
| Foveal schisis | 14 (70.0) | 59.2 ± 33.7 | |
| Foveal detachment | 5 (25.0) | 46.4 ± 22.1 | |
| Macular hole | 1 (5.0) | 30.0 ± 0 | |
| Dye for ILM peeling, n (%) | .0629 | ||
| ICG | 12 (60.0) | 44.3 ± 20.2 | |
| BBG | 8 (40.0) | 70.1 ± 38.2 | |
| Intraocular gas, n (%) | .221 | ||
| SF6 | 13 (65.0) | 48.3 ± 24.2 | |
| Air | 7 (35.0) | 66.3 ± 39.5 | |
| Area of ILM peeling (disc area ± SD) [range] | 15.3 ± 5.9 [5.5–25.1] | .613 | |
| Postoperative visual acuity (logMAR equivalent) (mean ± SD) [range] | 0.28 ± 0.33 (1.10 – −0.17) | .581 | |
| Change in visual acuity (log MAR equivalent) (mean ± SD) [range] | 0.24 ± 0.16 [0.54 – −0.10] | .217 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


