Purpose
To examine quantitatively characteristics of the peripapillary retinal nerve fiber layer (RNFL) in preterm children using Fourier-domain optical coherence tomography (FD-OCT).
Design
Prospective cross-sectional study.
Methods
A 3-mm high-resolution FD-OCT peripapillary RNFL circular scan centered on the optic disc was obtained from right eyes of 25 preterm children (10.6 ± 3.7 years old, 8 preterm and 17 with regressed retinopathy of prematurity with normal-appearing posterior poles) and 54 full-term controls (9.8 ± 3.2 years old). Images were analyzed using Spectralis FD-OCT software to obtain average thickness measurements for 6 sectors (temporal superior, temporal, temporal inferior, nasal inferior, nasal, nasal superior), and the global average.
Results
The RNFL global average for preterm children was 8% thinner than for full-term controls. In the preterm group, peripapillary RNFL thickness on the temporal side of the disc was 6% thicker than in full-term controls, while all other peripapillary RNFL sectors were 9% to 13% thinner. In the preterm group, temporal sector peripapillary RNFL thickness was correlated with gestational age (r = −0.47, P < .001), with foveal center total thickness (r = 0.48, P = .008, 1-tailed), and with visual acuity (r = 0.42; P = .026, 1-tailed).
Conclusions
The significantly thinner RNFL global average for preterm children suggests that prematurity is associated with subclinical optic nerve hypoplasia. Significant correlations between temporal sector RNFL thickness and both the foveal thickness and visual acuity suggest that the peripapillary RNFL is related to abnormalities in macular development as a result of preterm birth.
Infants born at less than 32 weeks postconception are at risk for many kinds of eye disorders: retinopathy of prematurity (ROP), amblyopia, strabismus, nystagmus, and optic nerve hypoplasia. Better knowledge of the retinal characteristics of the preterm children would permit more accurate diagnosis and treatment of disorders that are associated with preterm birth.
As measured by optical coherence tomography (OCT), preterm children with or without regressed ROP have greater total foveal thickness than normal controls. We recently reported that birth at <28 weeks postconception was associated with a failure of the inner retinal layers to migrate away from the fovea, resulting in increased thickness of inner foveal layers.
Blood vessels normally begin to develop in the central retina at 24 to 28 weeks postconception. These blood vessels grow from the optic disc toward the central retina dependent on both an oxygen gradient and chemical guidance factors that influence vascular patterning and definition of a foveal avascular zone. There is consensus that growing axons may offer guidance to the developing vasculature and/or axons and vessels may share common guidance signals. Because preterm birth disturbs the normal migration of inner retinal layers away from the fovea, patterning of the perifoveal vasculature may also be affected.
Peripapillary retinal nerve fiber (RNFL) thickness profiles correlate with the location of the main temporal superior and inferior blood vessels. Abnormalities of neurovascular developmental interactions in preterm children may result in abnormal peripapillary RNFL thickness profiles.
Our hypothesis is that abnormalities in the peripapillary RNFL thickness profile result from disruption of neurovascular developmental interactions by preterm birth at <28 weeks. The aim of this study was to examine quantitative characteristics of the peripapillary RNFL in preterm children born at 24 to 32 weeks postconception with and without regressed ROP using Fourier-domain OCT (FD-OCT) to determine whether gestational age at birth is correlated with the type and severity of peripapillary RNFL abnormalities that occur, to examine the relationship between retinal structural abnormalities and visual impairment, and, at the same time, to compare with normal full-term controls.
Methods
Subjects
Twenty-five preterm children were enrolled in the study. Preterm subjects had: 1) a gestational age at birth <32 weeks postconception; 2) either no ROP (N = 8) or a history of regressed ROP (either regression spontaneously or regression following treatment with peripheral ablative surgery, stage 1 to stage 3 acute phase) (N = 17); 3) normal-appearing posterior poles on ophthalmoscopy; 4) ≥5 years of age. Fourteen of the children were born at 24 to 27 weeks postconception and 11 were born at 29 to 31 weeks postconception. They were referred by a pediatric ophthalmologist who conducts ROP screenings in multiple Dallas-area neonatal intensive care units or by a retina specialist who treats ROP. Children with coexisting disease or developmental delay were excluded.
A control group consisted of an age-matched group of 54 healthy children who were born at full term (37−42 weeks of gestational age) ( Table ). These children initially were recruited by letters given to parents of healthy newborns from a local community hospital nursery and have participated as normal controls in a number of studies at the Retina Foundation of the Southwest during infancy and childhood.
| Group | N | Gestational Age (w) | Birth Weight (g), Mean ± SD | Age at Test (y), Mean ± SD (Range) | Sex (F/M) | Spherical Equivalent (D), Mean ± SD (Range) | Acute Stage (0/1/2/3/NA) | Laser (Y/N) |
|---|---|---|---|---|---|---|---|---|
| Preterm | 25 | 27 ± 2.7 | 949.8 ± 440 | 10.6 ± 3.7 (5−19) | 13/12 | −0.6 ± 4.7 (−14.75−11) | (8/3/3/9/2) | 9/16 |
| Full-term control | 54 | 39.5 ± 1.4 | 3572 ± 478 | 9.8 ± 3.2 (5−18) | 25/29 | (−3-1) | NA | NA |
Procedure
Demographics, gestational age at birth, birth weight, acute and cicatricial stages of ROP, treatment for ROP, and cycloplegic refractive error were obtained from medical records ( Table ).
All individuals were scanned by 1 investigator with the Spectralis FD-OCT (Heidelberg Engineering Carlsbad, California, USA) using the eye-tracking feature (ART). Up to three 3-mm high-resolution FD-OCT peripapillary RNFL circular scans centered on the optic disc were obtained from the right eye of each individual; the highest-quality image was selected for analysis. Imaging was obtained in a dim room and an internal fixation target was used in all scans.
On the same day as OCT imaging, visual acuity was measured using a computerized method based on the Early Treatment of Diabetic Retinopathy Study (ETDRS) visual acuity testing protocol, referred to as electronic ETDRS (E-ETDRS).
Peripapillary Retinal Nerve Fiber Layer Circular Scan Analysis
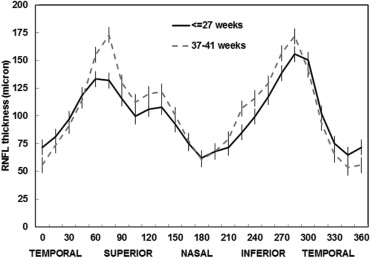
RNFL thickness (from the internal limiting membrane inner margin to RNFL layer outer margin) was automatically segmented using the Spectralis software (ver. 5.3.2.0; Heidelberg Engineering). Segmentation lines were inspected and manually adjusted in cases of error. As shown in Figure 1 , this software provided a thickness profile across the temporal-superior-nasal-inferior-temporal areas of the circular scan, the TSNIT graph. The software also calculated average thickness for each of 6 sectors centered on the optic disc (temporal, superior temporal, temporal inferior, nasal inferior, nasal, nasal superior).
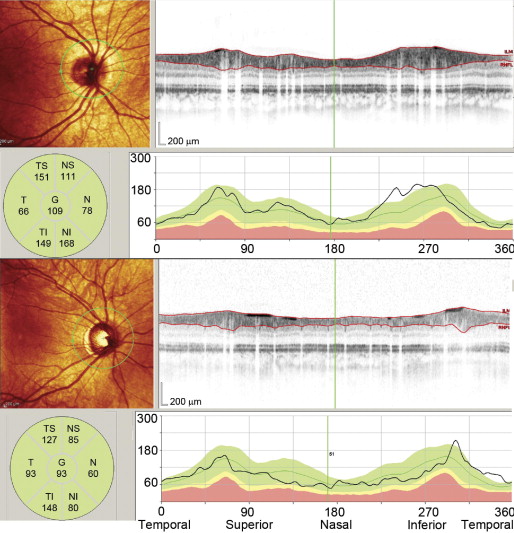
Optic nerve hypoplasia was evaluated using the global average thickness of the RNFL provided by the software.
Data Analysis
The statistical toolboxes in MatLab and STATISTICA (StatSoft Inc, Tulsa, Oklahoma, USA) were used to perform data analysis. Multiple sector comparisons among groups were adjusted using the Bonferroni method.
Results
The characteristics of the children are summarized in the Table ; characteristics include demographics, gestational age at birth, birth weight, age at testing, sex, cycloplegic refractive error (described as spherical equivalent), stage of ROP, and laser treatment for ROP. Visual acuity ranged from 20/16 to 20/80 for the preterm group and 20/16 to 20/20 for the normal control group.
Global Average Thickness of the Peripapillary Retinal Nerve Fiber Layer
The global average thickness of RNFL in preterm children (98.5 ± 10.3 μm) was significantly lower than in normal children (107 ± 9.4 μm) (t test, P = .001) ( Figure 3 ). RNFL global average thickness for preterm children was 8% thinner than for full-term controls. There was no significant correlation between global thickness of RNFL and gestational age or visual acuity.
Distribution Profile of the Peripapillary Retinal Nerve Fiber Layer Thickness
Figure 2 shows the mean distribution of spatial profile of peripapillary RNFL thickness in 360 degrees. The mean distribution profile of preterm children is flatter than that of full-term children. The peaks around superior and inferior sectors are lower and the troughs around temporal sectors are higher in preterm children, while the mean distribution profile in the normal control group is more arcuate.