Purpose
To describe the incidence and causes of glaucoma after Descemet membrane endothelial keratoplasty (DMEK).
Design
Nonrandomized prospective cohort study at a tertiary referral center.
Methods
The incidence of glaucoma was evaluated in the first 275 consecutive eyes that underwent DMEK for Fuchs endothelial dystrophy (260 eyes) or bullous keratopathy (15 eyes). Glaucoma was defined as a postoperative intraocular pressure (IOP) elevation of ≥24 mm Hg, or ≥10 mm Hg from the preoperative baseline. If possible, the cause of glaucoma was identified, and best-corrected visual acuity (BCVA), endothelial cell density (ECD), and postoperative course were documented, with a mean follow-up of 22 (± 13) months.
Results
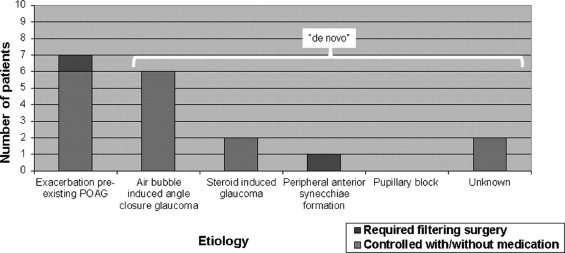
Overall, 18 eyes (6.5%) showed postoperative glaucoma after DMEK. Seven eyes (2.5%) had an exacerbation of a pre-existing glaucoma. Eleven eyes (4%) presented with a de novo IOP elevation, associated with air bubble–induced mechanical angle closure (2%), steroid response (0.7%), or peripheral anterior synechiae (0.4%), or without detectable cause (0.7%). Two eyes (0.7%) required glaucoma surgery after DMEK. At 6 months, all eyes had a BCVA of ≥20/40 (≥0.5), and 81% reached ≥20/25 (≥0.8) (n = 16); mean ECD was 1660 (± 554) cells/mm 2 (n = 15) ( P > .1).
Conclusion
Glaucoma after DMEK may be a relatively frequent complication that could be avoided by reducing the residual postoperative air bubble to 30% in phakic eyes, applying a population-specific steroid regime, and avoiding decentration of the Descemet graft. Eyes with a history of glaucoma may need close IOP monitoring in the first postoperative months, especially in eyes with an angle-supported phakic intraocular lens.
In the past decade, we have introduced various techniques for endothelial keratoplasty, later popularized as deep lamellar endothelial keratoplasty (DLEK) and Descemet stripping (automated) endothelial keratoplasty (DSEK/DSAEK). More recently, we described a technique for the selective transplantation of a donor Descemet membrane, now referred to as Descemet membrane endothelial keratoplasty (DMEK).
With the advent of endothelial keratoplasty, the type of complications and their incidence have shifted compared to those encountered after penetrating keratoplasty. Astigmatism, suture-related problems, and wound dehiscence have been virtually eliminated and the risk of allograft rejection may have been reduced, while graft detachment as a “new” complication has surfaced. However, endothelial keratoplasty may also be associated with a shift in intraocular complications, particularly specific types of glaucoma induced by the presence of an intraocular air bubble in the immediate postoperative phase. Overall, glaucoma has been described to occur in about 30% of cases after endothelial or penetrating keratoplasty, so that it may be one of few severe complications after keratoplasty today, affecting the clinical outcome and/or graft survival in the long term. Since DMEK grafts may require a longer air bubble support to obtain graft adherence to the recipient posterior stroma, the risk of glaucoma after DMEK could be higher than after DSEK/DSAEK, so a better understanding of the various mechanisms may aid in preventing glaucoma, thereby improving the overall success rate of the procedure.
In the present study, we prospectively documented all glaucoma episodes associated with the first 275 DMEK surgeries performed at our center, to determine the causes and incidence of postoperative intraocular pressure (IOP) elevations and to possibly define precautions to minimize the risk of glaucoma after DMEK.
Materials and Methods
This study was a nonrandomized prospective cohort study at a tertiary referral center. For the first consecutive 275 eyes of 229 patients that underwent DMEK for Fuchs endothelial dystrophy (n = 260) or bullous keratopathy (n = 15), the perioperative IOP was documented, with a mean postoperative follow-up time of 22 (± 13) months. Significant IOP elevation was defined as an IOP of ≥24 mm Hg or an elevation of ≥10 mm Hg compared to the preoperative IOP, as measured by Goldmann applanation tonometry. No eyes were excluded from analysis, and none of the eyes had preoperative angle abnormalities, a history of trauma, uveitis, or other relevant comorbidities, other than the pre-existing lens status or the presence of a phakic intraocular lens.
Surgical Procedure
From donor globes obtained less than 24 hours postmortem, corneoscleral buttons were excised and stored by organ culture in modified minimum essential medium (EMEM) at 31 C. After 1 week of culture, endothelial cell morphology and viability were evaluated and the corneoscleral buttons were mounted endothelial side up on a custom-made holder with a suction cup. The Descemet membrane (DM) was stripped from the posterior stroma so that a 9.5-mm-diameter flap of posterior DM with its endothelial monolayer was obtained. Because of the elastic properties of the membrane, a “Descemet roll” formed spontaneously, with the endothelium at the outer side. Each Descemet roll was then stored in organ culture medium until the time of transplantation.
In recipient eyes, a 3.0-mm tunnel incision was made just within the limbus, entering the anterior chamber just at the mark. With an inverted Sinskey hook (D.O.R.C. International, Zuidland, The Netherlands), a circular portion of DM was scored and stripped from the posterior stroma, so that a 9.0-mm-diameter “descemetorrhexis” was created, and the central portion of DM was removed from the eye.
The donor Descemet roll was stained with a 0.06% trypan blue solution (VisionBlue, D.O.R.C. International) and sucked into a custom-made injector (D.O.R.C. International). Using the injector, the donor Descemet roll was inserted into the anterior chamber and the graft was oriented endothelial side down (donor DM facing recipient posterior stroma) by careful, indirect manipulation of the tissue with air and fluid. While maintaining the anterior chamber with fluid and air, the graft was gently spread out over the iris. Then, an air bubble was injected underneath the donor DM to position the tissue onto the recipient posterior stroma. The anterior chamber was completely filled and pressurized with air for 45 to 60 minutes followed by a 50% air-liquid exchange to pressurize the eye. Thus, at termination of the surgery, a 50% air bubble, which did not cover the inferior pupillary margin with the patient in an upright position, was left in the anterior chamber.
Two weeks before surgery, a peripheral yttrium-aluminum-garnet (YAG) iridotomy was routinely made at the 12-o’clock position (because patients were requested to lie flat for several hours, Bell’s phenomenon could render an iridotomy at the 6-o’clock position ineffective). Postoperative medication included 0.5% chloramphenicol for 2 weeks and a routine steroid regime of 0.1% dexamethasone 4 times daily for 4 weeks, followed by fluorometholone 4 times daily, tapered to 1 time daily over a period of 1 year ( Table 1 ). Patients were evaluated at 1 day, 1 week, and 1, 3, 6, 9, and 12 months after the surgery, and at 6-month time intervals thereafter. Best-corrected visual acuity (BCVA), endothelial cell density by specular microscopy (Topcon SP3000P; Topcon Europe Medical BV, Capelle a/d IJssel, The Netherlands), Scheimpflug imaging (Oculus Pentacam HR 70900, Wetzlar, Germany), anterior segment optical coherence tomography (SL-OCT B01; Heidelberg Engineering, Heidelberg, Germany), and slit-lamp photography were performed. All intraoperative and postoperative complications were documented in an SQL database.
| Postoperative Time Interval | Steroid Prescribed |
|---|---|
| 1st month | Dexamethasone 0.1%, 4 times daily |
| 2nd−3rd month | Fluorometholone 0.1%, 4 times daily |
| 4th−6th month | Fluorometholone 0.1%, 3 times daily |
| 7th−9th month | Fluorometholone 0.1%, 2 times daily |
| 10th−12th month | Fluorometholone 0.1%, 1 time daily |
| >12 months | Fluorometholone 0.1%, every other day |
Statistical Analysis
Statistical analysis (SPSS 12.0 statistical package, SPSS, Inc, Chicago, Illinois, USA) was used to compare BCVA ( t test for independent samples [Levine test significance: P > .05]) and endothelial cell density (Mann-Whitney U test) outcomes at 6 months after DMEK between the glaucoma (n = 18) and the normal visual potential group (n = 176). P value less than .05 was considered statistically significant.
Results
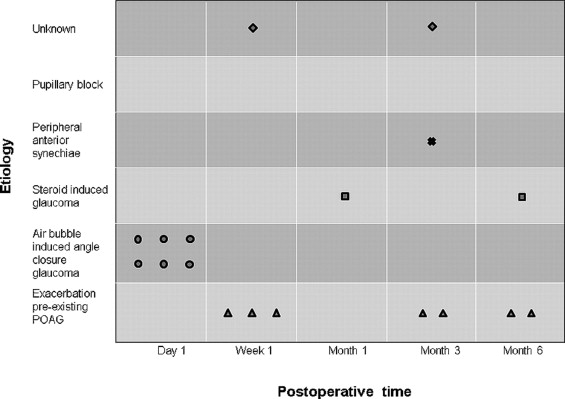
IOP elevation was found in 18 DMEK eyes (18/275; 6.5%) of 18 patients, 14 male and 4 female, 9 phakic and 9 pseudophakic, aged 33 to 81 years ( Table 2 ). Seven of 275 eyes (2.5%) had an exacerbation of a pre-existing glaucoma and 11 eyes (4.0%) presented with a de novo IOP elevation ( Figures 1 and 2 ).
| Patient # (Case #) | Age (y), Sex | OD/OS | Indication for Surgery | ACD (mm) | Glaucoma Type | Onset | IOP (mm Hg) | Initial Treatment | Clinical Course | BCVA at 6 Months | Δ ECD (%) at 6 Months (Absolute ECD in Cells/mm 2 ) | Eventual Status and Remarks |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patients With History of Glaucoma | ||||||||||||
| 1 (6) | 60, M | OS | Fuchs ED (pseudophakic) | n/a | Pre-existing glaucoma; POAG exacerbation | 6th mo postop | 24 | Topical medication | Controlled with topical medication | 20/20 (1.0) | 8% (2270) | Clear transplant |
| 2 (33) | 81, M | OD | Fuchs ED (pseudophakic) | 4.1 | Pre-existing glaucoma; steroid response | 3rd mo postop. | 25 | Steroid tapering; topical medication | Controlled with topical medication | 20/25 (0.8) | 16% (2350) | Clear transplant |
| 3 (75) | 76, M | OD | Fuchs ED (pseudophakic) | 4.4 | Pre-existing glaucoma; POAG exacerbation | 6th mo postop | 40 | Topical and oral medication | Controlled with topical medication | 20/25 (0.8) | 16% (2590) | Clear transplant (DMEK after DSEK) |
| 4 (188) | 74, M | OS | Bullous keratopathy (pseudophakic) | 4.0 | Pre-existing glaucoma; POAG exacerbation | 1st wk postop | 40 | Topical and oral medication | Controlled with topical medication | 20/25 (0.8) a | 32% (2020) | Clear transplant |
| 5 (193) | 78, M | OS | Fuchs ED (pseudophakic) | 4.4 | Pre-existing glaucoma; POAG exacerbation | 3rd mo postop | 25 | Topical medication | Controlled with topical medication | 20/28 (0.7) | 21% (1890) | Clear transplant |
| 6 (271) | 46, F | OD | Bullous keratopathy (phakic) | 2.8 | Pre-existing glaucoma; angle-supported phakic IOL-induced glaucoma | 1st wk postop | 26 | Topical medication | Controlled with topical medication | [20/130 (0.15)] b | 26% (1650) b | Clear transplant, CME |
| 7 (272) | 42, M | OD | Bullous keratopathy (phakic) | 2.9 | Pre-existing glaucoma; angle-supported phakic IOL-induced glaucoma | 1st wk postop | 40 | Topical and oral medication | Filtering surgery 4 months after DMEK | 20/40 (0.5) | 67% (760) b | Clear transplant, anterior synechiae, gonio synechiae, cataract |
| Patients Without History of Glaucoma | ||||||||||||
| 8 (30) | 45, M | OS | Fuchs ED (phakic) | 2.3 | Mechanical angle closure due to air bubble dislocation behind iris | 1st day postop | 30 | Pupillary dilation, topical and oral medication, supine position | No current treatment | 20/20 (1.0) | 47% (1430) | Clear transplant (unrelated mild allograft rejection 18 months after DMEK) |
| 9 (40) | 48, M | OD | Fuchs ED (phakic) | 2.5 | Mechanical angle closure due to air bubble dislocation behind iris | 1st day postop | 36 | Pupillary dilation, topical and oral medication, supine position | No current treatment | 20/20 (1.0) | 48% (1270) | Clear transplant |
| 10 (131) | 33, M | OS | Fuchs ED (phakic) | 3.0 | Mechanical angle closure due to air bubble–induced crystalline lens tilting | Hrs after surgery | 30 | Pupillary dilation, topical and oral medication, supine position | No current treatment | 20/20 (1.0) | 2% (2510) b | Clear transplant |
| 11 (138) | 54, M | OS | Fuchs ED (phakic) | 2.0 | Mechanical angle closure due to air bubble–induced crystalline lens tilting | 1st day postop | 32 | Pupillary dilation, topical and oral medication, supine position | No current treatment | 20/22 (0.9) | 57% (970) | Clear transplant |
| 12 (228) | 50, F | OS | Fuchs ED (phakic) | 2.9 | Mechanical angle closure due to air bubble–induced crystalline lens tilting | Hrs after surgery | >30 | Pupillary dilation, topical and oral medication, supine position | No current treatment | 20/28 (0.7) | 65% (840) | Clear transplant |
| 13 (251) | 50, F | OD | Fuchs ED (phakic) | 2.5 | Mechanical angle closure due to air bubble–induced crystalline lens tilting | Hrs after surgery | 36 | Pupillary dilation, topical and oral medication, supine position | No current treatment | 20/18 (1.2) | 16% (2020) | Clear transplant |
| 14 (87) | 66, M | OD | Fuchs ED (pseudophakic) | 3.4 | Steroid response | 6th mo postop | 26 | Steroid tapering | No current treatment | 20/20 (1.0) | 67% (810) | Clear transplant |
| 15 (154) | 66, M | OD | Fuchs ED (pseudophakic) | 4.3 | Steroid response | 1st mo postop | 28 | Steroid tapering; topical medication | No current treatment | 20/18 (1.2) | 47% (1390) | Clear transplant |
| 16 (163) | 62, M | OD | Fuchs ED (phakic) | 3.0 | Peripheral anterior synechiae formation | 3rd mo postop | 36 | Topical and oral medication | Filtering surgery 5 months after DMEK | 20/20 (1.0) c | 23% (1870) | Clear transplant, peripheral anterior synechiae |
| 17 (143) | 72, M | OS | Fuchs ED (pseudophakic) | 4.2 | Unknown | 3rd mo postop | 25 | Topical medication | Controlled with topical medication | 20/25 (0.8) | 36% (1780) | Clear transplant |
| 18 (166) | 79, F | OD | Fuchs ED (pseudophakic) | 2.8 | Unknown | 1st wk postop | 22 | No treatment | No current treatment | 20/21 (0.95) | 40% (1400) | Clear transplant |
| Average at 6 months after DMEK | 100% (16/16) ≥20/40 (0.5) 81.3% (13/16) ≥20/25 (0.8) | 36% ±19% (1660 ± 554) | ||||||||||
| Compared to 257 (275 − 18) DMEK eyes without glaucoma complication | P > .1 | P > .1 | ||||||||||
a Best spectacle-corrected visual acuity 20/40 (0.5) due to Salzmann nodulus; 20/25 (0.8) with contact lens 8 months after surgery.
b 3-month data are shown because 6-month data unavailable.
c Best spectacle-corrected visual acuity 20/20 (1.0) after secondary filtering surgery and cataract extraction; 20/30 (0.6) at 6 months, before cataract extraction.
The incidence of IOP elevation after DMEK was 25% (7/28) in the group with a history of glaucoma and 4.5% (11/247) in the group without a glaucoma history. One of the eyes in our study had undergone glaucoma surgery prior to DMEK but did not develop an IOP elevation after DMEK. Two of 275 eyes (0.7%; Cases 7 and 16; Table 2 ) required glaucoma surgery after DMEK.
Eyes With History of Glaucoma
In eyes with a history of glaucoma (Cases 1−7; Table 2 ), IOP elevation was attributed to an exacerbation of primary open-angle glaucoma in 4 eyes, to secondary glaucoma in the presence of an angle-supported “phakic intraocular lens” in 2 eyes, and to steroid response in 1 eye. In 6 of these 7 eyes, the IOP normalized with intensified medication and steroid tapering. One of these eyes (Case 7; Table 2 ) with an angle-supported phakic IOL showed progressive, uncontrolled IOP elevations from 3 months after DMEK onwards, and was scheduled for glaucoma surgery.
Eyes With De Novo Glaucoma – Immediate Postoperative Mechanical Angle Closure
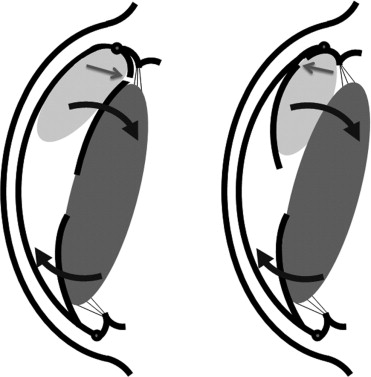
In the 11 eyes with a de novo glaucoma (Cases 8−18; Table 2 ), treatment varied with the assumed mechanism inducing the IOP elevation. In 6 phakic eyes (Cases 8−13; Figure 1 ; Table 2 ), the IOP elevation was attributed to an “air bubble–induced mechanical angle-closure glaucoma.” In 2 of these eyes, the air bubble had moved behind the iris ( Figure 3 ). In all 6 eyes, the situation could be resolved by pupillary dilation, topical 1% apraclonidine and 250 mg acetazolamide orally, and laying the patient down in a supine position for 1 to 2 hours. With the antiglaucoma medication continued for 1 to 2 days, no recurrences were seen.