When basic surgical principles are followed diagnostic salivary endoscopy is a relatively safe operative procedure. Therapeutic sialendoscopy uses such instrumentation as lasers, forceps, baskets, and balloons for endoductal fragmentation, retrieval, and dilatation. Based on experience acquired from more than 300 salivary endoscopy procedures and a review of the current literature, the most relevant operative techniques are presented.
Sialendoscopy, begun in 1988, is now an established procedure for the diagnosis of salivary disease and is a method to control minimally invasive interventions to treat salivary obstructions. In this article a discussion of indications and the different aspects of basic sialendoscopy technique are presented.
Indications
Diagnostic indications include suspicion of obstructive salivary disease (diagnostic endoscopy). Therapeutic indications include the following: treatment of salivary stones (including fragmentation, removal, and stone localization for external approaches); dilatation of strictures and localization of strictures for external approaches; management of chronic sialadenitis by irrigation; and management of recurrent juvenile sialadenitis.
Diagnostic Sialendoscopy
Careful patient history is the first step in ascertaining presence of obstructive salivary disease. Typically, gland swelling is associated with food intake. Inspection and palpation is sometimes helpful; however, ultrasonography is the most important diagnostic procedure. Applying a sialagogue during sonography can help to detect the cause and region of obstruction. In most cases, treatment can be planned with the information obtained by history and sonography. In some cases additional information may be necessary. The most relevant aspects regarding planning of treatment, to which plain sonography is of limited use, are the following:
- 1.
The distinction between a nonechogenic stone and a stricture (qualitative assessment): In the case of a suspected stricture a nonechogenic stone should be excluded using other methods (eg, sialendoscopy).
- 2.
The quantitative assessment of the obstruction using sonography: It is often difficult to measure the three-dimensional size of a stone or to assess the length and number of stenotic areas.
- 3.
The state of the distal duct system in the event that an intraductal approach is planned: It may be useful to assess the duct and its diameter distal of the obstruction to ensure that the duct is wide and straight enough for the instruments, to which plain sonography is poorly suited. These considerations are also important in determining whether the fragments produced by extracorporeal shock wave lithotripsy are easily washed out by the saliva from the duct system. For sonographically controlled procedures it is also important to know if there are kinks and branchings that might complicate approaching the obstruction.
Both sialendoscopy and sialography (sialogram) can be used to ascertain the three aspects listed previously. Sialography gives a better overview of the whole duct system and can give information about areas not reachable by sialendoscopy (eg, behind extreme kinks and strictures). Disadvantages are radiation exposure and false-positive stone detections because of air bubbles. Sialendoscopy allows visualizing the pathology directly. Both, sialendoscopy and fluoroscopy can also be used to control therapeutic interventions so that it is possible to switch from diagnostics to treatment within the same session.
Therapeutic Sialendoscopy
Apart from using the endoscope for the dilatation of strictures within the duct system, sialendoscopy is not a treatment itself but rather a method of visual control for therapeutic procedures.
Fragmentation and extraction of stones
Sialolithiasis is the most frequent reason for salivary duct obstruction. The aim of treatment is to completely remove the stone. Various approaches exist and endoscopic techniques are only one part of the whole spectrum of possibilities ( Table 1 ). It can be advantageous to combine different approaches (multimodal therapy).
| Method | Approach |
|---|---|
| Gland extirpation (not for distal stones) | External |
| Slitting of the duct | Transoral |
| Intraductal therapy Tasks Stone removal Stone fragmentation Duct dilatation Devices Basket, grasper, forceps, suction, balloon, stent, drill, laser Methods of control Blind/tactile Endoscopic Ultrasound Fluoroscopic | Transoral |
| Extracorporeal shock wave lithotripsy | External |
| Minimal incision and combined approaches from the outside for parotid stones | External |
The information obtained during diagnosis determines the choice of treatment. The most important parameters are the patient history of complaints and complications; the position, size, and number of stones; the diameter of the duct between the stone and the papilla; and the surgeon’s experience with the particular techniques.
For the most part, the smallest stones can be removed conservatively. For this, patients are asked to use sialagogues (eg, chewing gum, cherry pits, and so forth) and to massage their glands regularly. This approach is often very helpful as an auxiliary fragment clearing measure for use after other treatment types. If stones are recalcitrant, other treatment options are discussed. Figs. 1 and 2 list other therapeutic techniques to be used depending on the location of stones.


For basic sialendoscopy several parameters should be considered. The normal parotid and submandibular ducts have diameters of about 1.5 mm with bottlenecks up to 0.5 mm at the papillae. The mean diameters of the observed stones may have some variability ranging from about 3 to 8 mm, depending on individual experience. It has been suggested that a stone’s maximum diameter should not be larger than 150% of the anterior ducts and that the absolute diameters should not exceed 3 to 5 mm for Stensen duct and 4 to 7 mm for Wharton duct if it is to be removed without fragmentation. It is sometimes possible, however, to remove stones with even larger maximum diameters if their form is streamlined (eg, if they are highly ellipsoid). The chances for removal are reduced if the stone is not mobile because of adherence to the duct, or if it is located in a diverticulum. In the latter cases there is an increased risk of the basket instrument jamming in the duct.
An advanced application for sialendoscopy for salivary stones includes the localization of the stone by skin transillumination as an aid to external approach.
Dilatation of stenoses and strictures
Relatively few data exist regarding the treatment of salivary duct strictures. It is generally agreed that long strictures have a worse prognosis than short stenoses. Multiple technical options exist to dilate a duct (see Table 1 ). Endoscopically controlled procedures are especially helpful for short, membrane-like stenoses or where stenoses begin at duct branchings. The latter ones can be very difficult to treat with fluoroscopically or sonographically controlled procedures; filiform-like openings might not be identified by these methods. Disadvantages of the endoscopically controlled balloon dilatation are that the diameter of the duct created by the inflated balloon can only be assessed after deflation and that it may also be difficult to determine the position of the tip of the balloon. It can be helpful to combine endoscopy with one of the other imaging techniques (multimodal therapy). A useful technique for difficult stenosis or stricture cases is the placement of a guidewire using endoscopic control. The guidewire is then left in place while the endoscope is removed. Lastly, a balloon or hollow conical dilatator ( Fig. 3 A ) is placed over the guidewire and the dilatation procedure is continued under sonographic or fluoroscopic control.
Management of chronic sialadenitis
Sialendoscopy has been used successfully to treat adult chronic sialadenitis. The mechanism is probably clearance of mucous plugs and dilatation of the duct by irrigation and might be the same as for chronic juvenile parotitis. A less invasive procedure may be performed by just irrigating the duct with a catheter without endoscopy.
Management of recurrent juvenile sialadenitis
Recurrent juvenile parotitis can be treated successfully as with adult chronic sialadenitis using sialendoscopy. Sialography may also be useful (Dr P Katz, personal communication, 2007). The latter normally does not require general anesthesia as is often necessary for sialendoscopy. It is associated with radiation exposure, however, and iodinated contrast material remains long-term in the duct system as a basis for this treatment.
Contraindications to sialendoscopy
Sometimes acute sialadenitis is named as an absolute contraindication because the swollen duct wall is more vulnerable to perforation. Additionally, the endoscopic view is hampered by mucopurulent debris. Uncommonly, conservative treatment including intravenous antibiotics and oral antiphlogistics did not show sufficient effect to avoid a sonographically proved imminent abscess. In these uncommon cases, diagnostic sialendoscopy was then carefully performed to confirm that a sonographically controlled ductal procedure on the stone was possible and curative. Acute sialadenitis is probably a relative contraindication.
Contraindications to sialendoscopy
Sometimes acute sialadenitis is named as an absolute contraindication because the swollen duct wall is more vulnerable to perforation. Additionally, the endoscopic view is hampered by mucopurulent debris. Uncommonly, conservative treatment including intravenous antibiotics and oral antiphlogistics did not show sufficient effect to avoid a sonographically proved imminent abscess. In these uncommon cases, diagnostic sialendoscopy was then carefully performed to confirm that a sonographically controlled ductal procedure on the stone was possible and curative. Acute sialadenitis is probably a relative contraindication.
Side effects
Reported side effects and complications include the following :
- ○
Temporary swelling caused by irrigation (2–3 hours, 100%)
- ○
Wire-basket blockages (6%)
- ○
Canal wall perforations (0.3%–6%)
- ○
Recurrence of symptoms (1%–6%)
- ○
Temporal lingual nerve paresthesia (0.5%)
- ○
Ranula (1%)
- ○
Postoperative infection (2%)
- ○
Ductal strictures (0.3%–3.5%)
General principles for sialendoscopic procedures
Choice of the Appropriate Endoscope
For interventional endoscopies it is often advantageous to start with a fine-caliber diagnostic endoscope. It is less invasive because only a slight dilatation of the papilla is needed and only rarely a papillotomy. Trauma to the duct walls is minimal so that no iatrogenic lesions interfere with the assessment. The narrow diameter and greater flexibility make the procedure easier and allow for the inspection of parts that are not accessible with a larger interventional scope. Important information is gained, such as if a stricture is of short length and more easily treatable with the interventional scope. Another example is that it is often possible to pass by stones with the diagnostic scope by passing between the concretion and the duct wall and measuring the length of the stone and the proximal duct condition. This is important for therapeutic decisions, such as whether a basket can be opened behind the stone.
Preparation of the Optical System and Orientation of the Endoscope
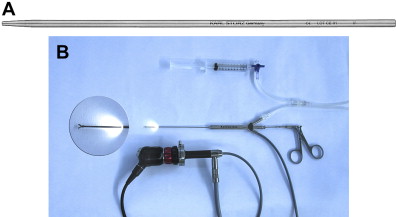
A typical set-up for sialendoscopy includes the connection of the endoscope to a camera system with a monitor, a light source, and an irrigation system ( Fig. 3 B). It is important to know the exact orientation of the endoscope with respect to the picture on the monitor before starting the procedure. For this reason the scope should be passed over a test region with lettering and the orientation can be corrected by repositioning the camera. At the same time acuity, zoom, and white balance can be adjusted. The surgeon should know about the orientation of the working channel relative to the scope’s objective lens. In selected cases it can be advantageous to rotate the position of the scope within one’s hands to a new “zero position” and correct the camera position respectively. This avoids viewing the target upside down on the monitor screen. At the same time it allows defining the position of the instrument leaving the working channel in any desired position relative to the optical axis. This can be of help, for example, when grabbing a stone with a forceps, surpassing a stone with a basket, or entering a stenotic duct with a balloon. Especially with larger instruments the instrument and not the optical axis of the endoscope is centered within the duct system. Foreseeing, choosing, and calculating the direction of this shift can be very helpful.
Anesthesia for Sialendoscopy
Dilatation of the papilla and diagnostic endoscopy are not very painful and often no anesthesia is necessary. The situation is vice versa for interventional sialendoscopy. In most cases an anesthetic irrigation of the duct system is sufficient by using an intravenous cannula or using the rinsing or working channels of an endoscope; xylometazolin 2% or bupivacaine 3% are used to flush the duct system. Additional local anesthesia injections or even regional blockage of nerves are sometimes necessary. Indications for general anesthesia are limited to more complicated problems and also to many children. Some authors recommend initially starting all sialendoscopy procedures under general anesthesia.
Operative Time
The average time needed for sialendoscopy is about 60 minutes, 57 ± 39 minutes for an interventional sialendoscopy for single sialoliths, and 89 ± 42 minutes for multiple sialoliths. It is comparable with the classic gland resection. It has a lower risk of side effects, however, and preserves the gland.
Introducing the Endoscope
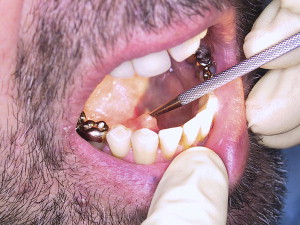
The first step is identification of the papilla. This can sometimes be difficult and time consuming. The use of magnifying loupes or a microscope can be helpful. One way to enhance the visibility of the papilla is to massage the gland with one hand, which pushes saliva to open the papilla and make it visible. Simultaneously, it is possible to introduce a conical dilatator into the opening with the other hand ( Fig. 4 ). This technique can be enhanced by using sialagogues (eg, ascorbic acid, lemon juice). It has recently been suggested to swab the region of the papilla with methylene blue, which might further facilitate the procedure. The papilla of Stensen duct can be found opposite to the second upper molar ( Fig. 5 ). Wharton duct enters the oral cavity at the anterior tip of the sublingual fold (see Fig. 4 ). Sometimes it is difficult to differentiate the Wharton duct papilla from the papillae of the minor sublingual ducts; sometimes it is covered by a mucosal fold. Because the anterior floor of mouth mucosa is quite loose it can be stabilized by grabbing it posterior-superior to the punctum with a toothed forceps. The natural papilla diameters are about 0.5 mm. Most cases need a papilla dilatation done with a conical dilatator. Two different types of dilatators are available: one “dilator” with a sharp tip and the other “bougie” with a blunter tip ( Figs. 6 and 7 ). The pointed one is good for entering a visibly very small papilla and starting dilatation until the blunt one fits. Forceful dilatation with the dilatators should be avoided because it can perforate the duct and create a false passage. The dilatators can also be used to identify a papilla tactilely by moving over the region of its expected position and “dropping” into it. A caution when identifying Wharton papilla is that a minor sublingual duct can be cannulated and a false lumen created. When in doubt a blunt probe should be introduced to check for the length, patency, and direction of the duct ( Fig. 8 ).

A further way to dilate the duct is by using the Solex system (soft lumen expander; Polydiagnost, Pfaffenhofen, Germany) ( Fig. 9 ). After removing the inner obturators a port remains that allows the introduction of the endoscope ( Fig. 10 ). This system is especially helpful in cases in which multiple introductions of the scope might be necessary or if the diameter of the papilla shrinks very quickly after dilatation.




